
As specialists in anterior segment surgery, we cataract and refractive surgeons do not tend to pay a great deal of attention to the posterior segment of the eye. However, it is important to keep in mind that the eye as a whole works as a complex optical system. There are several causes of different retinal conditions that we must keep in mind when planning for cataract surgery. Beyond retinal tears, multiple retina-related factors should be taken into account.
Diabetic retinopathy (DR) is the leading cause of vision loss in adults aged 20 to 74 years,1,2 and around 95 million people worldwide are affected by vision-threatening DR.2,3 Age-related macular degeneration (AMD) is the leading cause of visual disability in the industrialized world and the third leading cause of visual disability worldwide.4-6
Not every case of retinal disease shows specific symptoms, especially when it is masked by additional loss of vision due to an opacified crystalline lens. This article identifies several key tenets to be aware of when screening patients for cataract surgery and discusses how to handle retinal conditions that may be encountered.

PREOPERATIVE SCREENING FOR RETINAL DISEASE
A key factor in the screening process for retinal disease is anamnesis, or medical history. Close attention should be paid for a possible history of systemic vascular disease, diabetes, hypertension, rheumatic disorders, or autoimmune diseases. What we can determine before the patient’s physical exam enables us to later look more precisely into possible findings.
AT A GLANCE
• Ophthalmic surgeons of all subspecialties should keep in mind that the eye works as a complex optical system.
• Not every case of retinal disease shows specific symptoms, especially if it is masked by an additional loss of vision due to an opacified crystalline lens.
• Because the focus of cataract surgery is to enhance the intraocular optical system with the goal of correcting refractive errors, it is important to ensure that the perceptive part of the eye—the retina—is able to process the light information it will receive postoperatively.
The question of whether a patient’s visual acuity is in proportion to the findings of the examination must be raised and answered. Obvious changes of the fundus are often easy to identify but may still require a closer look. The differentiation between wet and dry AMD, early DR, and even a macular pucker or macular hole can easily be overlooked through an opacified lens. Modern diagnostic devices such as OCT and wide-field fundus photography can provide better accuracy in diagnosing possible retinal changes that could limit cataract surgery outcomes.
It is important to realize that, regardless of the demands of our daily routine and efforts to work more efficiently, our attention should always be centered on the patient and any possible diseases that could affect his or her visual acuity.

BUCKLING AND IOL CALCULATION
It is not only acute and chronic retinal diseases that deserve our attention. Treatment of retinal tears or small retinal detachments with scleral buckling surgery can have a significant impact on IOL calculation. When cataract surgery is combined with scleral buckling, the axial length of the globe can be expected to increase. Similarly, the axial length could decrease if an older buckle is to be removed during or after cataract extraction. Either increasing or shortening the axial length will induce a change in the refraction, and this must be taken into account.
However, scleral buckling does not influence only the axial length of the eye: Hayashi et al7 described significant corneal shape changes, too. Thus, not only spherical but also astigmatic values can be affected. In this time when cataract surgeons use many different types of aspheric IOLs for standard cataract surgery, we must also consider potential changes in higher-order aberrations (HOAs) and possible tilt or decentration problems caused by a more anterior scleral buckle.

PREVENTING POSTOPERATIVE CME
The literature reports the general risk of cystoid macular edema (CME) after uneventful cataract surgery to be 1% to 5%; this risk increases to around 20% in complicated cases.8 However, a case does not need to be complicated to carry an increased risk for postoperative CME development. In patients with DR, venous occlusive disease, and even macular pucker, the risk for postoperative CME can increase dramatically.
New NSAIDs such as nepafenac (Ilevro; Alcon) have proven effective in reducing CME development in at-risk populations. Approaches that include simultaneous use of intravitreal dexamethasone also seem promising for preventing CME in these patients. For patients with diabetic macular edema who are scheduled to undergo cataract surgery but are receiving intravitreal injections, use of a more long-term steroid delivery system such as the dexamethasone intravitreal implant (Ozurdex; Allergan) or the fluocinolone acetonide implant 0.19 mg (Iluvien; Alimera Sciences) can be beneficial. If the agent is still in effect during the scheduled lens extraction, the steroid will not only prevent CME development but also reduce any inflammatory effect of the surgery itself.
If obvious fundus changes such as neovascularization are seen, pretreatment with an anti-VEGF agent or classical laser coagulation of the affected retinal area can be useful before cataract surgery. Especially in eyes with severe iris neovascularization, additional anti-VEGF injections in the anterior chamber can reduce the risk of intraoperative complications.

CATARACT SURGERY IN AMD
An often discussed and controversial issue is the relationship between cataract surgery and the progression of AMD. Several study authors have documented an acceleration of AMD progression after cataract surgery,9-11 although others could not verify this correlation.11-14 Several recent articles underline the claim that cataract surgery in patients with AMD serves to improve both visual acuity and quality of life.16-18
In patients with dry AMD, the German Ophthalmological Society recommends cataract surgery without prophylactic intravitreal injection of any anti-VEGF agent. The need for use of a blue-light-filtering IOL is also a controversial topic in this area. So far, no data have shown a protective ability of yellow IOLs to reduce the risk of AMD development or progression.
In patients with wet AMD (Figure 1), recommendations for the timing of surgery vary. Although some surgeons believe that OCT must demonstrate a dry macula preoperatively, it is proven that visual function increases after cataract surgery in patients with wet AMD. I believe that both diseases, AMD and cataract, must be treated effectively.
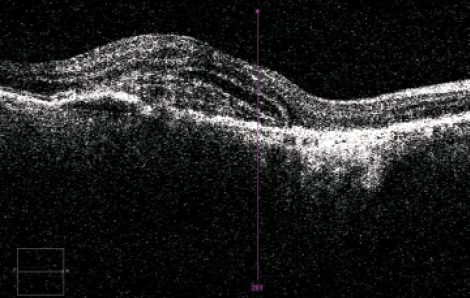
Figure 1. OCT in a patient with wet AMD.
In our clinics, we prefer that the patient have a stabilized AMD status with no increase of fluid on OCT. In patients with newly diagnosed AMD, we prefer to give at least two intravitreal injections, followed by reevaluation of the OCT images. If the disease has stabilized or improved, we schedule the cataract extraction before the next anti-VEGF injection. In patients who have received long-term intravitreal treatments and whose OCT findings are stabilized, we also schedule the cataract procedure between two anti-VEGF injections.

VITREOMACULAR TRACTION
Although uncommon, additional vitreomacular traction (VMT) syndrome can still be seen in cataract patients. Often asymptomatic, VMT syndrome can be associated with peripheral retinal tears. If vitreoretinal adhesions (Figure 2) are observed, an extensive retinal fundus examination should be performed to rule out possible causes of future retinal detachment. If these can be excluded, pretreatment of the VMT is not always necessary. Mirshahi et al19 showed a 21% induction of vitreoretinal detachment 1 week after cataract surgery and 31% at 4 weeks after cataract surgery. If a vitreoretinal detachment cannot be achieved, further treatment options are available.
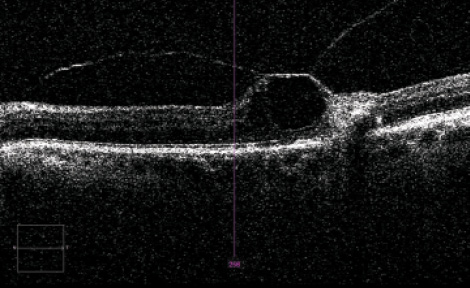
Figure 2. VMT with interrupted intraretinal layers.
CONCLUSION
Ophthalmologists must be aware of all the diseases and conditions that can affect our patients, even if they fall outside our subspecialty. Considering that the primary focus of cataract surgery is to enhance the intraocular optical system with the aim of correcting refractive errors, we must ensure that the perceptive part of the eye—the retina—is able to process the light information that will be transmitted through the IOLs we implant. Small changes in our daily examination, planning, and operative routines can have a big impact on our patients achieving their best possible outcomes.
1. Cheung N, Mitchell P, Wong TV. Diabetic retinopathy. Lancet. 2010;376(9735):124-136.
2. Lee R, Wong TY, Sabanayagam C. Epidemiology of diabetic retinopathy, diabetic macular edema and related vision loss. Eye Vis (Lond). 2015;2:17.
3. Yau JW, Rogers SL, Kawasaki R, et al; Meta-Analysis for Eye Disease (META-EYE) Study Group. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care. 2012;35(3):556-564.
4. Pascolini D, Mariotti SP. Global estimates of visual impairment: 2010. Br J Ophthalmol. 2012;96(5):614-618.
5. Pennington KL, DeAngelis MM. Epidemiology of age-related macular degeneration (AMD): associations with cardiovascular disease phenotypes and lipid factors. Eye Vis (Lond). 2016;3:34.
6. Wong WL, Su X, Li X, et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob Health. 2014;2(2):e106-e116.
7. Hayashi H, Hayashi K, Nakao F, Hayashi F. Corneal shape changes after scleral buckling surgery. Ophthalmology. 1997;104(5):831-837.
8. Kessel L, Tendal B, Jørgensen KJ, et al. Post-cataract prevention of inflammation and macular edema by steroid and nonsteroidal anti-inflammatory eye drops: a systematic review. Ophthalmology. 2014;121(10):1915-1924.
9. Freeman EE, Munoz B, West SK, Tielsch JM, Schein OD. Is there an association between cataract surgery and age-related macular degeneration? Data from three population-based studies. Am J Ophthalmol. 2003;135(6):849-856.
10. Kaiserman I, Kaiserman N, Elhayany A, Vinker S. Cataract surgery is associated with a higher rate of photodynamic therapy for age-related macular degeneration. Ophthalmology. 2007;114(2):278-282.
11. Pollack A, Marcovich A, Bukelman A, Oliver M. Age-related macular degeneration after extracapsular cataract extraction with intraocular lens implantation. Ophthalmology. 1996;103(10):1546-1554.
12. Baatz H, Darawsha R, Ackermann H, et al. Phacoemulsification does not induce neovascular age-related macular degeneration. Invest Ophthalmol Vis Sci. 2008;49(3):1079-1083.
13. Dong LM, Stark WJ, Jefferys JL, et al. Progression of age-related macular degeneration after cataract surgery. Arch Ophthalmol. 2009;127(11):1412-1419.
14. Sutter FK, Menghini M, Barthelmes D, et al. Is pseudophakia a risk factor for neovascular age-related macular degeneration? Invest Ophthalmol Vis Sci. 2007;48(4):1472-1475.
15. Hooper CY, Lamoureux EL, Lim L, et al. Cataract surgery in high-risk age-related macular degeneration: a randomized controlled trial. Clin Exp Ophthalmol. 2009;37(6):570-576.
16. Huynh N, Nicholson BP, Agron E, et al; Age-Related Eye Disease Study 2 Research Group. Visual acuity after cataract surgery in patients with age-related macular degeneration: age-related eye disease study 2 report number 5. Ophthalmology. 2014;121(6):1229-12236.
17. Monestam E, Lundqvist B. Long-term visual outcome after cataract surgery: comparison of healthy eyes and eyes with age-related macular degeneration. J Cataract Refract Surg. 2012;38(3):409-414.
18. Rosenfeld PJ, Shapiro H, Ehrlich JS, Wong P; MARINA and ANCHOR Study Groups. Cataract surgery in ranibizumab-treated patients with neovascular age-related macular degeneration from the phase 3 ANCHOR and MARINA trials. Am J Ophthalmol. 2011;152(5):793-798.
19. Mirshahi A, Hohn F, Lorenz K, Hattenbach LO. Incidence of posterior vitreous detachment after cataract surgery. J Cataract Refract Surg. 2009;35(6):987-991.


