
In the medical industry and profession, simplicity usually means lower costs, reduced risks, and increased usability. When procedures are made simpler, everybody benefits: the patient, the treating ophthalmologist, and, potentially, the ophthalmic industry as well.
The possibility of performing CXL at the slit lamp, rather than in the operating room, is an example of how simplicity may induce a paradigm shift both clinically and commercially. The goal is to reduce the minimum infrastructure necessary to perform a CXL procedure and, thus, potentially open the technique to use by ophthalmologists who are not necessarily specialized anterior segment surgeons.
AT A GLANCE
• The possibility of performing CXL at the slit lamp, rather than in the OR, is an example of how simplicity may induce a paradigm shift both clinically and commercially.
• Recent research suggests that riboflavin distribution is position-independent for the first 60 minutes after instillation, a time span largely sufficient to perform CXL.
• Omitting the requirement of having to perform CXL in the aseptic environment of the OR can reduce the time and related costs for both the surgeon and the patient, can allow a clinician or vision health care specialist without easy access to an OR to perform CXL, and could increase the number of potential customers for CXL.
Currently, the most common treatment protocol for CXL for ectatic eye diseases is to remove the corneal epithelium, apply riboflavin drops, wait for saturation, and then administer UV-A light in an operating room setting with the patient supine.1,2 If we wish to perform CXL at the slit lamp, three elements, listed in the graphic directly below, must be achieved. This article addresses the questions related to these elements and goes on to explore other issues related to performing CXL at the slit lamp.
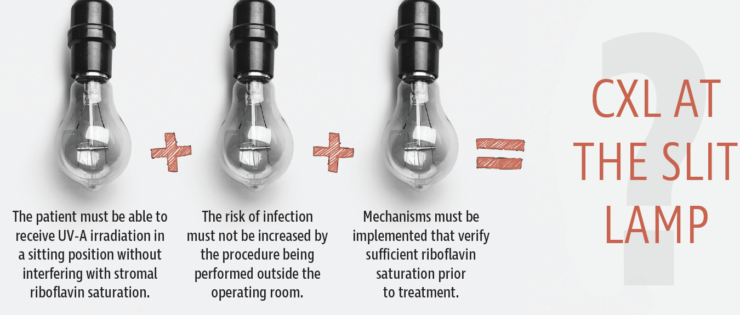
ANSWERING THE QUESTIONS

Does the patient lose riboflavin saturation when sitting in an upright position? Sufficient riboflavin saturation is an essential element to achieve the maximal biomechanical effect of CXL. A number of conditions must be considered to achieve maximal saturation, including the appropriate osmolarity of the riboflavin solution, the removal or nonremoval of the epithelium, and the duration of riboflavin instillation. Another important question is whether the patient’s position during treatment affects the saturation levels needed for maximal effect. Recent research suggests that riboflavin distribution is indeed position-independent for the first 60 minutes after instillation, a time span largely sufficient to perform CXL.3

Is an operating room location necessary to safely perform CXL? Another misconception is that CXL treatment must be performed in an operating room (OR) because of the risk of infection. In this regard, it is important to note that every CXL procedure provides not only a biomechanical stiffening effect, but also several additional complex effects, including a strong antimicrobial effect, disrupting the cellular membranes of both bacteria and fungi and blocking replication via DNA-RNA intercalation. In other words, every CXL treatment, whether it is performed for keratoconus or for infection, stiffens the cornea and also leaves the cornea disinfected, so to speak, at the end of treatment. Therefore, it does not necessarily have to be performed in a sterile environment.

Figure 1. The C-Eye device is designed to perform CXL at the slit lamp.
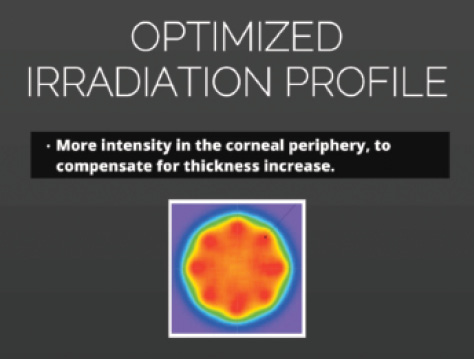
Figure 2. The optimized irradiation profile of the C-Eye device.
Omitting the requirement of having to perform CXL in the aseptic environment of the OR can significantly reduce the time and related costs for both the surgeon and the patient. Additionally, a clinician or vision health care specialist without easy access to an OR can perform CXL using this approach, which significantly increases the usability of the method and, consequently, the number of potential customers for CXL.
Further, transferring a septic patient into an aseptic environment to perform the antiseptic CXL procedure potentially creates added risk for other patients who will subsequently be operated on in the same environment (ie, for cataract surgery). Performing CXL at the slit lamp avoids this potential risk.

Is there a feedback mechanism that can verify proper riboflavin saturation? Having an easy and quick means to verify that there is enough riboflavin saturation before UV-A administration, such as a fluorescence sensor, could also increase the success rate of the treatment. Furthermore, if this saturation sensor indicated that not enough riboflavin had been absorbed, the clinician could review a checklist of reasons why the cornea might not be fully saturated. Next, the clinician could correct the problem or problems, and only then administer the UV-A light for the treatment.
A measurement tool is currently being integrated into the C-Eye device (EMAGine), designed for CXL at the slit lamp (Figure 1). The device is CE-Mark pending, and EMAGine has been granted patents in Japan and South Africa, with pending patent applications in nine countries and EU members states. Additionally, it has an optimized irradiation profile that provides more intensity of treatment in the corneal periphery in order to compensate for the thickness increase at the slit lamp (Figure 2). For more information on using the C-Eye to perform CXL at the slit lamp, see How Does It Work?

I already have a UV-A lamp for CXL, so do I need a device that treats at the slit lamp? If CXL is used only for treating keratoconus and ectasia, performing CXL at the slit lamp might not lower infrastructure costs enough or save sufficient time to justify the purchase of new equipment. On the other hand, the availability of CXL treatment at the slit lamp could have a major impact if its utility is expanded to the treatment of infectious keratitis.
A procedure known as photoactived chromophore for keratitis CXL (PACK-CXL) uses the same basic materials and methods as traditional CXL, but its aim is radically different: the killing of harmful pathogens. With an estimated several millions of cases globally per year, corneal infections have been called a silent epidemic.4,5
When a patient presents with a corneal infiltrate or infection, the clinician is faced with three major challenges, especially when practicing in a remote location:
• Properly diagnosing the origin of the infiltrate/infection;
• Dealing with a low expectation for patient compliance; and
• Accounting for the costs related to follow-up visits.
In most cases, diagnosis of corneal infections is based on the clinician’s judgments and assumptions, as there are no simple means to confirm the nature of the underlying microorganism at the time of examination. Because early treatment reduces the spread of infection, the clinician must make these educated guesses and begin treatment immediately.
CXL at the Slit Lamp: How Does It Work?
• Apply topical anesthetics
• If performing PACK-CXL, remove debris at the slit lamp with a dry sponge
• If performing CXL, perform abrasion at the slit lamp
• Move the patient to a reclining chair, where riboflavin installation occurs
• Mount the C-Eye device and sterile cap
• Irradiate at the slit lamp
There are two possible ways to overcome this diagnostic dilemma: (1) improve the devices or means available for point-of-care diagnosis, or (2) develop a treatment modality that is not pathogen-specific. PACK-CXL represents such a treatment modality and has been shown to be effective in killing multiple types of harmful pathogens, including antibiotic-resistant organisms.6-9 The riboflavin formulations that are currently commercially available can already help significantly in reducing the number of eyes and the amount of vision lost to infections, especially in remote locations. Other chromophores that will further increase the killing rates will become available in the future.

If antimicrobial drugs are readily available, why should I perform PACK-CXL? In unfortunate cases in developed and developing countries alike, some infections are either mixed (bacterial-fungal) or caused by antibiotic-resistant bacteria. These cases severely complicate treatment: Either antimicrobial drugs are unable to kill both pathogens at once, or the drugs do not work at all because of resistance. No matter what drugs are administered, therefore, the outcome in the worst cases might be irreversible damage to the eye and vision of the patient.10,11 PACK-CXL is effective against both bacteria and fungi, and it kills bacteria irrespective of antibiotic resistance.
Costs may be an issue, however. Although antifungal and antibiotic pharmaceutical drugs may be readily available at relatively low cost, in some situations the number of doctor’s visits required for adequate follow-up is the limiting factor. For many patients, their funds are barely sufficient to pay for travel costs and a single visit. These patients may be lost to follow-up, and, often, delayed consequences may ensue.
PACK-CXL treatment provides another advantage over antimicrobial agents as well. It strengthens the cornea against enzymatic digestion by collagenases, and this may help to keep the size of scar formation to a minimum.
CONCLUSION
There are two main aims in introducing and promoting the concept of CXL at the slit lamp:
No. 1: To improve access to treatment for keratoconus, corneal ectasia, and corneal infections; and
No. 2: To improve patient care by reducing costs while increasing the safety and efficacy of the treatment.
Performing CXL and PACK-CXL at the slit lamp may provide a means to achieve both of these aims.
1. Raiskup-Wolf F, Hoyer A, Spoerl E, Pillunat LE. Collagen crosslinking with riboflavin and ultraviolet-A light in keratoconus: long-term results. J Cataract Refract Surg. 2008;34(5):796-801.
2. Wollensak G, Spoerl E, Seiler T. Riboflavin/ultraviolet-A-induced collagen crosslinking for the treatment of keratoconus. Am J Ophthalmol. 2003;135(5):620-627.
3. Salmon B, Richoz O, Tabibian D, Kling S, Wuarin R, Hafezi F. CXL at the slit lamp: no clinically relevant changes in corneal riboflavin distribution during upright UV Irradiation. J Refract Surg. In press.
4. Gonzales CA, Srinivasan M, Whitcher JP, Smolin G. Incidence of corneal ulceration in Madurai district, South India. Ophthalmic Epidemiol. 1996;3(3):159-166.
5. Whitcher JP, Srinivasan M. Corneal ulceration in the developing world--a silent epidemic. Br J Ophthalmol. 1997;81(8):622-623.
6. Kymionis GD, Kouroupaki AI, Liakopoulos DA, Arandjelovic IR, Tsoulnaras KI. Multiorganism, drug-resistant keratitis treated by corneal crosslinking [published online ahead of print January 22, 2016]. Eur J Ophthalmol.
7. Alshehri JM, Caballero-Lima D, Hillarby MC, et al. Evaluation of corneal cross-linking for treatment of fungal keratitis: Using confocal laser scanning microscopy on an ex vivo human corneal model. Invest Ophthalmol Vis Sci. 2016;57(14):6367-6373.
8. Bamdad S, Malekhosseini H, Khosravi A. Ultraviolet A/riboflavin collagen cross-linking for treatment of moderate bacterial corneal ulcers. Cornea. 2015;34(4):402-406.
9. Richoz O, Kling S, Hoogewoud F, Hammer A, et al. Antibacterial efficacy of accelerated photoactivated chromophore for keratitis-corneal collagen cross-linking (PACK-CXL). J Refract Surg. 2014;30(12):850-854.
10. Jhanji V, Sharma N, Satpathy G, Titiyal J. Fourth-generation fluoroquinolone-resistant bacterial keratitis. J Cataract Refract Surg. 2007;33(8):1488-1489.
11. Mamalis N. The increasing problem of antibiotic resistance. J Cataract Refract Surg. 2007;33(11):1831-1832.


