
To date, more than 800,000 US military personnel have undergone successful refractive surgery as part of the Warfighter Refractive Eye Surgery Program (WRESP), an initiative designed to reduce the limitations posed by corrective eyewear in combat arms soldiers. Although a majority of the procedures performed as part of the WRESP have been excimer laser surgery, the EVO Visian ICL (STAAR Surgical) is available in selected US Army WRESP centers to patients who are contraindicated for a laser-based correction procedure such as LASIK and PRK.
BACKGROUND
Both laser- and lens-based solutions to refractive errors can effectively reduce the need for corrective eyewear in the general public and in combat arms soldiers. However, some vision problems after LASIK have been cited in the literature. Namely, low luminance vision and contrast sensitivity can be diminished after surgery. More recently, the use of wavefront technology in excimer laser ablations has resulted in less degradation of low luminance vision and contrast sensitivity;1 yet it has been shown that the EVO Visian ICL induces significantly fewer higher-order aberrations (HOAs) and improves contrast sensitivity when compared with wavefront-guided LASIK.2 Furthermore, the EVO Visian ICL has been found to be safer and more effective than LASIK in treating low and moderate to high myopia.3
To the best of my knowledge, no study has fully compared the quality of night vision in patients after wavefront-optimized LASIK to that in patients who have received a phakic IOL. Therefore, my colleagues and I recently compared night vision performance in patients who had undergone wavefront-optimized LASIK with the WaveLight Allegretto (Alcon) to those implanted with the EVO Visian ICL. In order to test vision in conditions of low luminance, a simulated night vision goggle (NVG) environment was used. Below I share our results.
STUDY DESIGN
A total of 95 eyes (LASIK group, n=48; ICL group, n=47) with -3.00 to -11.50 D myopia and no more than 2.25 D astigmatism were enrolled. All patients had requested refractive surgery, and permission was granted by his or her military commander. Those with primary open-angle glaucoma or cataract were not eligible.
After a screening examination with a WRESP optometrist, all patients attended a group briefing session in which the risks, benefits, and alternatives of refractive surgery were discussed. Upon completion, each patient had an individual consultation with a surgeon. If eligible for laser vision correction, the patient was free to select between LASIK and PRK.
If ineligible due to abnormal topography, thin central corneal thickness, or low residual keratometry readings, the patient was considered for the EVO Visian ICL. Only those with a healthy endothelium were considered candidates for the ICL. In the ICL group, a laser peripheral iridotomy was performed 2 weeks prior to lens implantation. Then, during lens implantation, the primary incision was placed temporally or on the steep axis of refractive cylinder; no astigmatic treatments such as limbal relaxing incisions or bioptics were performed.
Both preoperatively and 1 and 3 months postoperatively, visual performance was assessed with the Rabin Super Vision Test (Precision Vision), a program that simulates the chromaticity and luminance of a NVG display at 25% moonlight. At those same time intervals, total HOAs also were measured with the WaveLight Topolyzer (Alcon). Both sets of tests were administered by a certified ophthalmic technician specifically blinded to the procedure, and the test results were interpreted by an ophthalmologist blinded to the procedure.
RESULTS
Spherical equivalent. The attempted versus achieved spherical equivalent results are presented in Figure 1. When we looked at refractive accuracy, what we found is that, at 3 months postoperatively, 100% of patients enrolled in the study were within ±1.00 D of intended correction. Furthermore, 49% of the ICL group and 40% of the LASIK group were within ±0.13 D (Figure 2). Looking at ±0.50 D, 98% of the ICL group and 92% of the LASIK group were within this range.
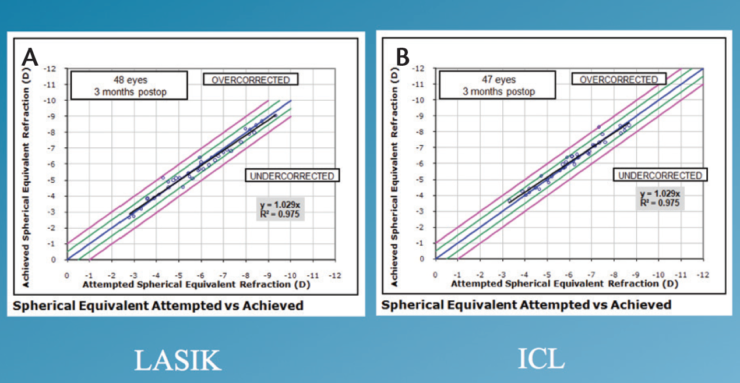
Figure 1. Attempted versus achieved spherical equivalent in the LASIK (A) and ICL (B) groups.
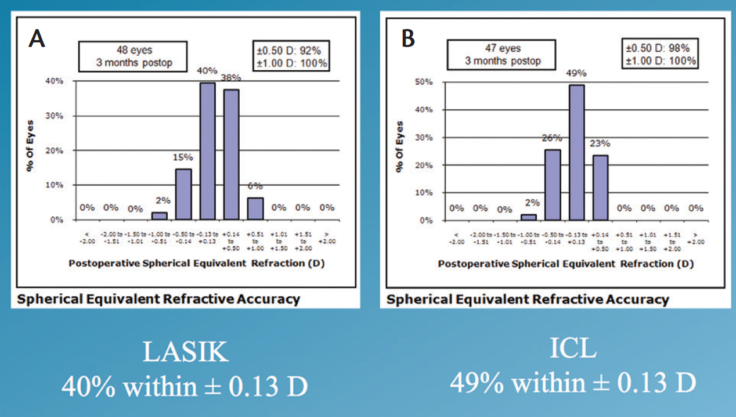
Figure 2. Spherical equivalent in the LASIK (A) and ICL (B) groups.
Visual acuity in normal conditions. Visual acuity (logMAR) and contrast sensitivity were presented on a single chart under both normal light and low luminance levels with the Rabin Super Vision Test. At 3 months postoperatively, 95% of all eyes were 20/20 or better in normal light conditions. When each group was analyzed separately, 96% of the ICL group and 94% of the LASIK group had a UDVA of 20/20 or better (Figure 3).
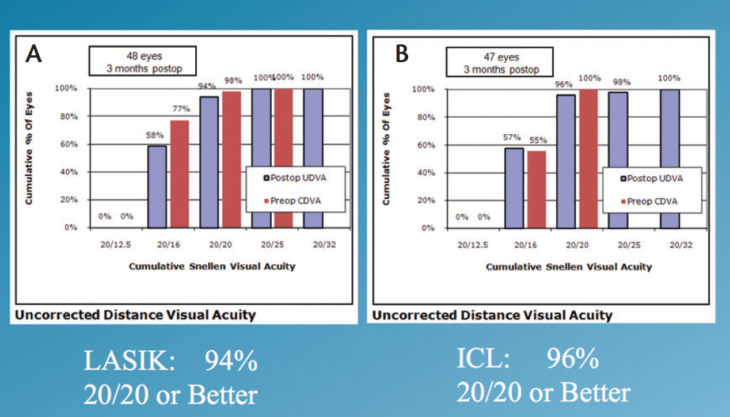
Figure 3. UDVA in the LASIK (A) and ICL (B) groups.
Low luminance visual acuity. At 3 months postoperatively, low luminance BCVA had significantly improved in the ICL group but not in the LASIK group (Figure 4). Furthermore, improvement in contrast sensitivity in low luminance conditions was seen in both groups; however, these improvements were significantly greater in the ICL group (P=.04; Figure 5).
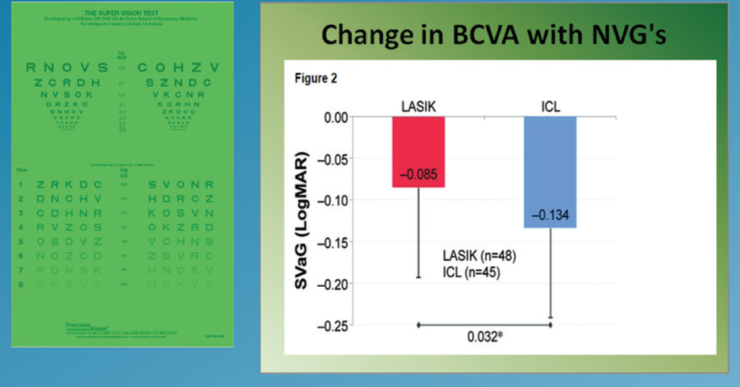
Figure 4. Mean within eye change in BCVA under low luminance levels.
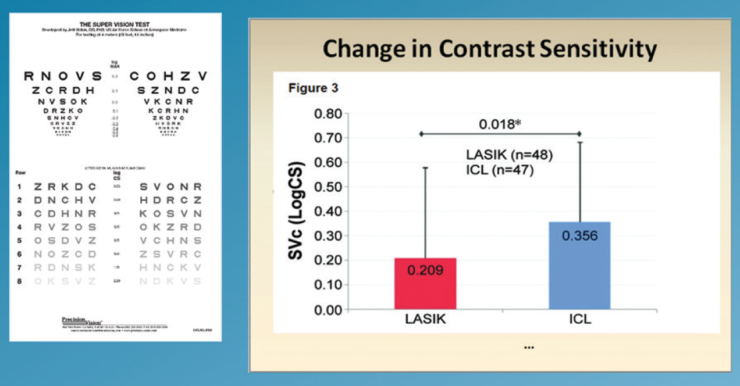
Figure 5. Mean within eye change in contrast sensitivity under low luminance levels.
CONCLUSION
Given that low luminance visual acuity and contrast sensitivity significantly improved in combat arms soldiers after implantation of the EVO Visian ICL and that these outcomes were significantly better than those observed after wavefront-optimized LASIK, phakic IOLs are an interesting option for the correction of refractive errors. This is especially true in patients who require excellent night vision, such as the population included in our study. In addition to visual performance, stability of the ICL is another important consideration included in the risk/benefit analysis for refractive surgery
*Author’s Note: The use of Nightfighter data and information does not imply or constitute US Department of Defense endorsement.
1. Parkhurst GD. A prospective comparison of phakic collamer lenses and wavefront-optimized laser-assisted in situ keratomileusis for correction of myopia. Clin Ophthalmol. 2016;10:1209-1215.
2. Igarashi A, Kamiya K, Shimizu K, Komatsu M. Visual performance after implantable collamer lens implantation and wavefront-guided laser in situ keratomileusis for high myopia. Am J Ophthalmol. 2009;148(1):164-170.
3. Sanders D, Vukich JA. Comparison of implantable collamer lens (ICL) and laser-assisted in situ keratomileusis (LASIK) for low myopia. Cornea. 2006;25(10):1139-1146.
