One of the biggest challenges in refractive cataract surgery is the poor predictability of IOL power in eyes after corneal refractive surgery. These include eyes that have undergone PRK, LASIK, and especially RK that now require cataract surgery. Working in clinics where these procedures have been performed for more than 2 decades, we see more than our fair share of postrefractive surgery patients coming in for cataract surgery.
These patients are demanding, too. They have enjoyed good uncorrected vision for years, and they expect the same freedom from glasses following their cataract surgeries. With current IOL formulas, this is a big enough challenge in normal eyes, let alone previously treated ones. This is how the ClearSight project was born: the need to find a solution through which IOLs could be selected, without formulas, by correctly measuring the optical properties of the eye and then ray tracing them. The initial wish was to find a solution for postrefractive surgery eyes; later, the value of the new technology was appreciated for routine cataract surgery and refractive lens exchange eyes, too, as in all cases excellent refractive outcomes were achievable at the push of a button. (Editor’s Note: For more information on ClearSight, see Accurate Measurements Can Strengthen Cataract Surgery Outcomes.)
This is a hot development space, and many companies are making contributions to the market by combining their anterior segment imaging devices with ocular biometry for measurements of anterior segment dimensions. We recently undertook a comparative analysis to examine what technologies are currently available, and this article recaps some of our findings.

IMPORTANCE OF ACCURATE MEASUREMENT
Today, measuring ocular parameters to derive the inputs for IOL power calculations is based on optical technologies that must provide accuracy and reproducibility. Most published reports on these technologies focus on reproducibility; there is relatively little published on the accuracy of the measurements. Generally, in selecting a biometric instrument, we must consider how accurately it measures the following parameters: curvatures of the anterior and posterior cornea; axial length (AL); anterior chamber depth (ACD); and lens thickness (LT). These are the four major variables used in calculating the power of an IOL to be implanted.
Several technologies are available to measure the anterior surface of the cornea, ranging from reflection-based methods such as keratometry and corneal topography, to Scheimpflug imaging, to anterior segment OCT. Each technology has its own benefits and limitations. Reflection-based methods have the ability to measure with high accuracy; however, limitations in acquiring information on the posterior corneal surface and even ultrastructural information from the cornea have been noted. In contrast, this information is provided both by Scheimpflug measurements and by anterior segment OCT. The current limitation of these two technologies is the accuracy of measurement because of motion artifacts associated with the acquisition of data.
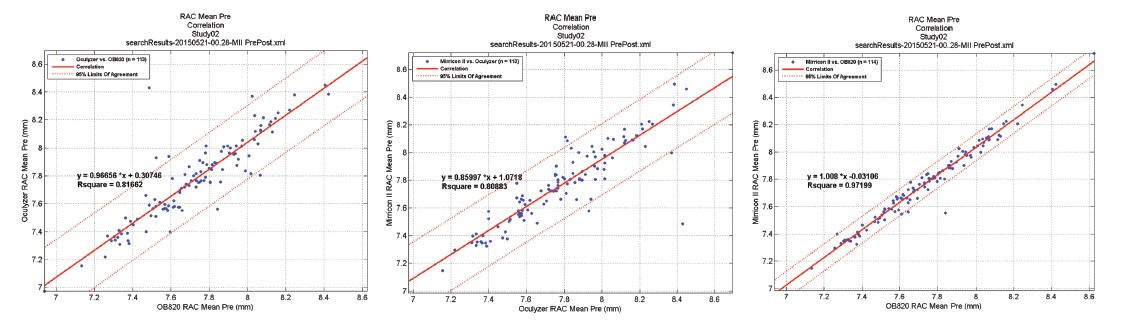
Figure 1. Radius of anterior cornea: Comparison between two reflection-based methods and one Scheimpflug imaging technology.
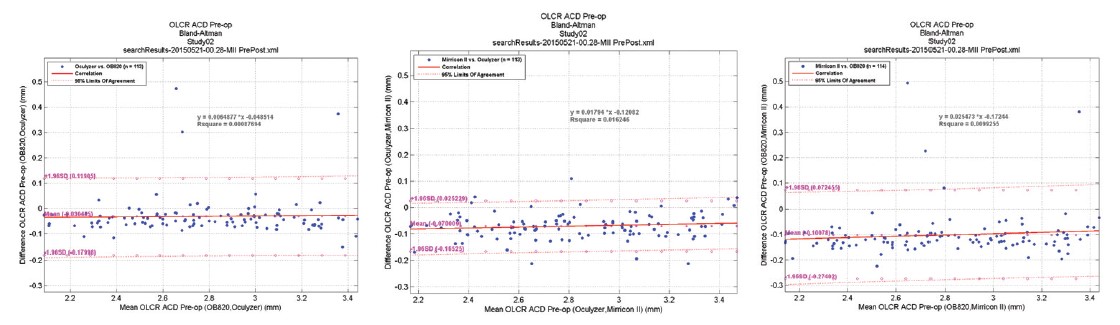
Figure 2. Bland-Altman analysis for preoperative anterior chamber depth between the Lenstar, Mirricon, and a Scheimpflug imaging device.
As an example, Figure 1 shows a comparison of the measured mean anterior corneal curvature for two reflection-based methods and for one Scheimpflug imaging technology. The lack of reproducibility of the mean corneal curvature in human eyes with the Scheimpflug device also raises the question of this technology’s ability to accurately measure the posterior radius of curvature. Similar results have been reported with anterior segment OCT devices.1
In addition to accurate corneal curvature measurements, precise AL measurements are required. Current commercially available devices use optical coherence methods to determine AL. Here, accuracy is mainly driven by three factors: (1) signal-to-noise ratio for peak detection; (2) eye motion during an AL scan; and (3) variability of refractive index of the ocular media. Scanning speed and signal-to-noise ratio are usually linked. Faster scanning allows avoidance of eye motion artifacts, but it can also lead to a reduced signal, mainly in dense cataracts.
An unsolved problem in currently available devices is the limitation of the unknown effective refractive index of the crystalline lens. Most devices use an ultrasonic equivalent to derive the geometric AL from the optical coherence signal. As the refractive index can vary substantially from eye to eye, depending on cataract grade, the accuracy of the AL measurement is affected. The associated error is on the order of about 1%, leading to an uncertainty of about 0.23 mm for a 23-mm long eye. This equals an uncertainty of nearly 0.70 D that will not be recognized by reproducibility measurements as it is a systematic error for each individual eye.
Preoperative ACD and LT have been introduced as parameters that can help effectively predict IOL position.2 Thus, ocular biometry should have the ability to measure these parameters reliably and accurately. A study by Kervick and colleagues at the Mater Private Hospital in Cork, Ireland, demonstrated that, in measurement of ACD, the Lenstar (Haag-Streit) has produced three outliers in ACD measurements compared with the other biometer and Scheimpflug devices. This is seen in the Bland-Altman analysis provided in Figure 2. Such outliers of up to 0.5 mm could lead to a 0.50 D difference in refractive outcome after cataract surgery (personal communication).
Another potential source of error is human factors associated with the manual handling of data generated in IOL power calculation. This is especially true if measurement data must be entered into toric IOL calculators or into the ASCRS website for calculating IOL power after corneal refractive surgery. Such potential errors are hard to quantify and might occur in only a few cases. Optical ray-tracing software has been proposed as a way to minimize such human factors, with software to import the measurements directly from corneal topographers and ocular biometers. However, optical ray tracing can deliver accurate results only if the measurements provided are highly accurate. Also, optical ray tracing does not depend on the A-constant of an IOL, it instead requires information on the geometries of IOLs to allow prediction of the best IOL for each individual eye.
AN OVERVIEW OF OCULAR BIOMETRY DEVICES
Devices currently available and on the horizon in the European Union for measurement of biometry and other anterior segment values are listed in Ocular Biometry Technologies. Below we share our analysis of how these products stack up against each other; in the subsequent pages, each system is described in detail and is paired with comments from an expert surgeon using the technology.
The most familiar devices for IOL power calculation, in their latest incarnations, are the Lenstar LS 900 with the optional T-Cone topography add-on and the IOLMaster 700 (Carl Zeiss Meditec). The Lenstar LS 900 and T-Cone combine noncontact optical low coherence reflectometry (OLCR) and Placido-disc–based topography, whereas the IOLMaster 700 is a swept-source OCT-based biometer. Various versions of these two devices have been shown to provide comparable ocular measurements and refraction prediction results in numerous publications.3-8
Both the Lenstar LS 900 and the IOLMaster 700 provide similar ocular parameters: anterior corneal radii (RAC), central corneal thickness (CCT), ACD, LT, AL, white-to-white distance (WTW), and pupil diameter (PD). These parameters, when successfully measured, are sufficient for IOL power prediction when classical formulas are used. However, they do not provide a full description of the phakic eye as required for ray-tracing IOL power calculation, specifically for unusual eyes. For example, corneal asphericities are important for IOL power prediction in both normal and postrefractive surgery eyes.9 The posterior cornea is another ocular element not measured by either the IOLMaster or Lenstar. Its accurate measurement would significantly improve IOL power calculation, especially for toric IOLs.10-12
At a Glance
• Of the technologies that can measure the corneal anterior surface, each has its own benefits and limitations. Reflection-based methods are highly accurate; however, they have limited ability to acquire information on the posterior corneal surface and even ultrastructural information from the cornea. In contrast, this information is provided by Scheimpflug measurements and by anterior segment OCT; however, the accuracy of measurement with these devices is limited because of motion artifacts associated with the acquisition of data.
• Optical ray-tracing software has been proposed as a way to minimize the manual handling of data.
• Although multiple options in the selection of biometry devices for IOL power calculation are available, no single device measures all the required parameters for ray-tracing IOL power prediction that would work on all eyes for all applications.
A device that might provide the missing ocular elements is the Pentacam (Oculus Optikgeräte), which employs Scheimpflug imaging to measure additional parameters of the anterior eye, such as corneal asphericities and posterior corneal curvature. However, these parameters as measured by the Pentacam have not always been found to be accurate.13 The current version of the Pentacam has an optional software add-on so that it can be used for IOL power calculation;14-16 however, the device does not measure all the required ocular elements, and AL must be measured by a separate biometry device.
For the other devices on the list, there is much less published information regarding accuracy and reliability for IOL power calculation. The Aladdin (Topcon), which includes an optical biometry system and Placido-disc topography, measures the same ocular parameters as the Lenstar and IOLMaster. It has been shown to give results comparable with those of the IOLMaster in terms of ocular parameters and predicted IOL power.17
The Galilei G6 (Ziemer), combining optical A-scan, dual-Scheimpflug imaging, and Placido-disc topography, is reported to measure most ocular elements; however, posterior cornea and corneal asphericities do not appear to be available. Comparisons of earlier Galilei systems with the Pentacam, IOLMaster, and Lenstar have shown comparable results;18-20 however, we identified no publications on the latest device, the Galilei G6, in our literature search.
The OA-2000 (Tomey), which combines Placido-disc topography and Fourier-domain A-scan technology, measures RAC, CCT, ACD, LT, AL, WTW, and PD. No publications were found on refraction prediction and IOL power calculation with this device. On the other hand, the previous version of this instrument, the OA-1000, was shown to provide ocular measurements that correlated well with the IOLMaster.21
The AL-Scan optical biometer (Nidek) provides AL, RAC, ACD, CCT, WTW, and PD, and it demonstrated good repeatability and reproducibility, providing IOL power results comparable with the IOLMaster.20,21 When used in combination with the OPD-Scan III (Nidek), a combined wavefront aberrometer, topographer, autorefractometer, and autokeratometer, it may provide more accurate IOL power prediction. However, the use of both devices is required to attain this improvement.
The Cassini (i-Optics) is a multicolored spot reflection topographer, and it measures total corneal astigmatism. It has been shown to give repeatable measurements24 and has demonstrated comparable corneal power measurements with the Lenstar and Pentacam.25,26 Unfortunately, we identified no publications on the accuracy of predicted refraction. This device must be combined with an optical biometer to provide complete data for IOL power calculation.
No IOL power prediction accuracy has been reported for the Argos (Movu), which is not commercially available yet. This device is a swept-source OCT system, and it measures AL, CCT, ACD, LT, WTW, PD, and RAC. As with most of the devices discussed above, it does not measure asphericities of the posterior cornea.
As can be seen, there are multiple options in the selection of biometry devices for IOL power calculation. However, no single device measures all the required parameters for ray-tracing IOL power prediction that would work on all eyes for all applications, including normal, postrefractive surgery, and toric IOL planning. Such a device would require true measurements of all ocular elements, without assumptions and fudge factors, in order to obtain the optimal individualized IOL power for each eye.
THE VALUE OF ACCURATE OCULAR BIOMETRY
By Arthur B. Cummings, MB ChB, FCS(SA), MMed(Ophth), FRCS(Edin); Diana Bogusevschi, PhD; Andrew Nolan, PhD; Ronan Byrne, MBA; Maria Galligan, BSc; and Michael Mrochen, PhD
Whether the eyes of the patient in our office are normal, short, long, or postrefractive surgery, the objective for the surgeon remains the same: to provide the best possible refractive outcome. To do this well can take significant experience, time, and effort utilizing the available devices and formulas. Even so, the challenge of dealing with the individual eye as opposed to the average one can result in refractive surprises for some patients.
By accurately measuring all the ocular parameters, we are able to obtain the complete geometry of the eye. This removes possible sources of error and allows the biometry to do the work without effort from the surgeon in arriving at the correct IOL power prediction for the individual patient. Thus, the task can reliably be delegated to a technician who can do the work with the simple push of a button. It also removes the risk of human error and allows us to deal with difficult cases such as postrefractive surgery eyes with a higher degree of confidence than heretofore.
Another exciting consequence of having more accurate biometry is the ability to offer more premium lens solutions. For example, choosing the right axis and power for a toric IOL based on currently available ocular biometry can be a challenge. Besides the accuracy and reliability of the measurements, we have to consider potential human errors occurring due to manual planning, which can lead to refractive outliers. Intraoperative aberrometry has been proposed to overcome this problem; the primary advantage is limited to axis alignment during surgery, not so much the IOL power selection. Improving the accuracy of IOL power and axis prediction would allow us to correct lower amounts of astigmatism with sufficient accuracy, leading to the ability to serve more patients with toric IOLs.
Multifocal IOLs also require precise prediction of IOL power and final lens position. The optical concepts of these IOLs are based on creating two or three focal planes or expanding the depth of focus of the IOL. These lenses are designed to have a contrast peak in the through-focus curve at a predetermined defocus—namely, at emmetropia. Any residual refractive error will shift this throughfocus curve from its intended position, and this can lead to visual symptoms and patient complaints. Consequently, the confidence of surgeons in these lenses is generally low, especially for higher refractive adds. Improving the accuracy of prediction of the IOL position would potentially increase surgeon confidence and drive the use of multifocal IOLs into more routine use.
SUMMARY
Improving IOL power predictability is important for patients, surgeons, and clinics. Patient demands increase with every good outcome achieved, and good results help build your practice.
When four old friends play golf together and one is routinely asked to find or follow the ball because his eyesight is so good after cataract surgery, that is a practice-building result. The more that surgeons become aware of this and take advantage of new biometric technologies to produce good refractive outcomes, the more they will achieve results like these. We must all strive to improve our refractive outcomes, and industry today is providing us with a wide range of options to do just that.
ClearSight Innovations has been working to address the issues raised in this article, and the prototype Mirricon device measures all the ocular parameters needed for IOL power calculation without reliance on formulas. A prospective 100-eye clinical trial has been completed, and presentation and publication of the results will be forthcoming. n
1. Lee YW, Choi CY, Yoon GY. Comparison of dual rotating Scheimpflug-Placido, swept-source optical coherence tomography, and Placido-scanning-slit systems. J Cataract Refract Surg. 2015;41(5):1018-1029.
2. Olsen T, Hoffmann P. C constant: New concept for ray tracing-assisted intraocular lens power calculation. J Cataract Refract Surg. 2014;40(5):764-773.
3. Rohrer K, Frueh BE, Wälti R, Clemetson IA, Tappeiner C, Goldblum D. Comparison and evaluation of ocular biometry using a new noncontact optical low-coherence reflectometer. Ophthalmology. 2009;116(11):2087-2092.
4. Cruysberg LPJ, Doors M, Verbakel F, Berendschot TTJM, De Brabander J, Nuijts RMMA. Evaluation of the Lenstar LS 900 non-contact biometer. Br J Ophthalmol. 2010;94:106-110.
5. Olsen T. Improved IOL power calculation using Lenstar measurements. Cataract & Refractive Surgery Today Europe. October 2010.
6. Reitblat O, Assia EI, Kleinmann G, Levy A, Barrett GD, Abulafia A. Accuracy of predicted refraction with multifocal intraocular lenses using two biometry measurement devices and multiple intraocular lens power calulation formulas. Clin Experiment Ophthalmol. 2015;43(4):328-334.
7. Jasvinder S, Khang TF, Sarinder KK, Loo VP, Subrayan V. Agreement analysis of Lenstar with other techniques of biometry. Eye (Lond). 2011;25(6):717-724.
8. Mylonas G, Sacu S, Buehl W, Ritter M, Georgopoulos M, Schmidt-Erfurth U. Performance of three biometry devices in patients with different grades of age-related cataract. Acta Ophthalmol. 2011;89(3):e237-241.
9. Savini G, Hoffer KJ, Barboni P. Influence of corneal asphericity on the refractive outcome of intraocular lens implantation in cataract surgery. J Cataract Refract Surg. 2015;41(4)785-789.
10. Savini G, Næser K. An analysis of the factors influencing the residual refractive astigmatism after cataract surgery with toric intraocular lenses. Invest Ophthalmol Vis Sci, 2015;56(2):827-835.
11. Preussner PR, Hoffmann P, Wahl J. Impact of posterior corneal surface on toric intraocular lens (IOL) calculation [published online ahead of print September 26, 2014]. Curr Eye Res.
12. Alpins N, Ong JK, Stamatelatos G. Corneal topographic astigmatism (CorT) to quantify total corneal astigmatism. J Refract Surg. 2015;31(3):182-186.
13. Piñero DP, Alió JL, Alesón A, Escaf M, Miranda M. Pentacam posterior and anterior corneal aberrations in normal and keratoconic eyes. Clin Exp Optom. 2009;92(3):297-303.
14. Sayed KM, Alsamman AH. Interchangeability between Pentacam and IOLMaster in phakic intraocular lens calculation. Eur J Ophthalmol. 2015;25(3):202-207.
15. Karunaratne N. Comparison of the Pentacam equivalent keratometry measurement in intraocular lens power calculations. Clin Experiment Ophthalmol. 2013;41(9):825-834.
16. Kim EC, Cho K, Hwang HS, Hwang KY, Kim MS. Intraocular lens prediction accuracy after corneal refractive surgery using K values from 3 devices. J Cataract Refract Surg. 2013;39(11):1640-1646.
17. Mandal P, Berrow EJ, Naroo SA, et al. Validity and repeatability of the Aladdin ocular biometer. Br J Ophthalmol. 2014;98(2):256-258.
18. Salouti R, Nowroozzadeh MH, Zamani M, Ghoreyshi M, Salouti R. Comparison of anterior chamber depth measurements using Galilei, HR Pentacam, and Orbscan II. Optometry. 2010;81(1):35-39.
19. Zhang L, Sy ME, Mai H, Yu F, Hamilton DR. Effect of posterior corneal astigmatism on refractive outcomes after toric intraocular lens implantation. J Cataract Refract Surg. 2015;41(1):84-89.
20. Koch DD, Jenkins RB, Weikert MP, Yeu E, Wang L. Correcting astigmatism with toric intraocular lenses: effect of posterior corneal astigmatism. J Cataract Refract Surg. 2013;39(12):1803-1809.
21. Goebels SC, Seitz B, Langenbucher A. Comparison of the new biometer OA-1000 with IOLMaster and Tomey AL-3000. Curr Eye Res. 2013;38(9):910-916.
22. Yagci R, Güler E, Kulak AE, Erdogan BD, Balci M, Hepsen F. Repeatability and reproducibility of a new optical biometer in normal and keratoconic eyes. J Cataract Refract Surg. 2015;41(1):171-177.
23. Huang J, Savini G, Li J, et al. Evaluation of a new optical biometry device for measurements of ocular components and its comparison with IOLMaster. Br J Ophthalmol. 2014;98(9):1277-1281.
24. Kanellopoulos AJ, Asimellis G. Distribution and repeatability of corneal astigmatism measurements (magnitude and axis) evaluated with color light emitting diode reflection topography [published online ahead of print June 6, 2015]. Cornea.
25. Klijn S, Reus NJ, Sicam VA. Evaluation of keratometry with a novel color-LED corneal topographer. J Refract Surg. 2015;31(4):249-256.
26. Hidalgo IR, Rozema JJ, Dhubhghaill SN, Zakaria N, Koppen C, Tassignon MJ. Repeatability and inter-device agreement for three different methods of keratometry: Placido, Scheimpflug, and color LED corneal topography. J Refract Surg. 2015;31(3):176-181.
Diana Bogusevschi, PhD; and Andrew Nolan, PhD
• Senior Scientists, ClearSight Innovations
• diana.bogusevschi@clearsightinnovations.com; andrew.nolan@clearsightinnovations.com
• Financial disclosure: Employees (ClearSight Innovations)
Ronan Byrne, MBA
• CEO, ClearSight Innovations
• ronan.byrne@clearsightinnovations.com
• Financial disclosure: Employee (ClearSight Innovations)
Arthur B. Cummings, MB ChB, FCS(SA), MMed(Ophth), FRCS(Edin)
• Consultant Ophthalmologist, Wellington Eye Clinic and Beacon Hospital, Dublin, Ireland
• Associate Chief Medical Editor, CRST Europe
• abc@wellingtoneyeclinic.com
• Financial disclosure: Consultant (Alcon/WaveLight), Chief Medical Officer (ClearSight Innovations)
Maria Galligan, BSc
• Clinical Trial Coordinator, ClearSight Innovations
• maria.galligan@clearsightinnovations.com
• Financial disclosure: Employee (ClearSight Innovations)
Michael Mrochen, PhD
• Chief Technical Officer and Cofounder, ClearSight Innovations
• Chief Executive Officer, IROC Science, Zurich, Switzerland
• michael.mrochen@irocscience.com
• Financial disclosure: Consultant (Alcon/WaveLight, Avedro, IROC Innocross)


