
Dry eye disease (DED) involves several ocular tissues, including the lacrimal glands, cornea, conjunctiva, lid structures, and a complex feedback system of sensory and motor nerves that connect these tissues.1 Disease or damage to any component can destabilize the tear film and lead to signs and symptoms of dry eye. In the United States, approximately 3.2 million women and 1.6 million men age 50 years and older experience moderate to severe DED;2,3 when Europe is included in this statistic, the number of people with moderate to severe DED soars to 45 million.4 It is likely many more people have DED that is undiagnosed or unaddressed despite symptoms and ocular surface changes. DED can limit and degrade performance of vision-related daily activities such as reading, driving, and computer work, negatively affecting quality of life and productivity.1
The goals in caring for patients with DED are to improve patients’ ocular comfort and quality of life, and, if possible, to return their ocular surface and tear film to its homeostatic state. Numerous therapeutic and palliative products for DED are available. This article reviews some of the options available to patients and eye care practitioners, with emphasis on products approved in the United States.
LUBRICANTS
Ocular lubricants are the first line treatment in patients with mild or moderate DED. Products vary from one to another in their electrolyte composition, osmolarity, viscosity or polymeric system, type of preservative, and the presence or absence of preservative. Patients may need to try several formulations to find one that best relieves their ocular surface symptoms.
Patients with moderate DED may benefit from tears with less toxic preservatives, such as polyquad (polyquaternium-1, used for example in Systane Ultra Lubricant Eye Drops [Alcon]), sodium chlorite (Purite, used for example in Refresh Tears [Allergan]), or sodium perborate (used for example in TheraTears [Advanced Vision Research]; Genteal [Novartis]; and Refresh Tears). These preservatives may be less toxic for the ocular surface while remaining cost effective and convenient.
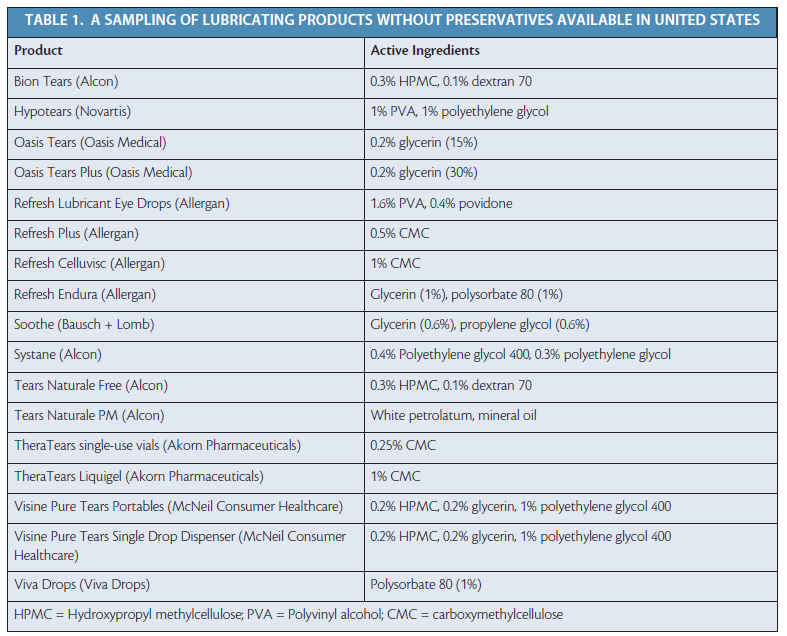
The ideal artificial lubricant should be preservative-free, have a long retention time on the ocular surface, and complement the salts found in normal tears. Table 1 lists some preservative-free lubricating products that are available in the United States (for additional products, see Preservative-Free Preparations Available Outside the United States).
Lacrisert (hydroxypropyl cellulose ophthalmic insert; Bausch + Lomb) is a tear pellet that melts in the inferior cul-de-sac of the eye. Patients who can overcome the learning curve for insertion find Lacrisert to be a convenient and effective lubricant. This log-shaped insert is often used before sleep to provide good lubrication throughout the night and allow patients to start the day with few symptoms.
Ocular ointments and gels are also used in treatment of DED. Ointments are formulated with mineral oil and petrolatum and do not require preservatives. Gels have longer retention times than artificial tears and have less blurring effect than petrolatum ointments.
At a Glance
• Because ocular lubricants vary from one to another in their electrolyte composition, osmolarity, viscosity or polymeric system, type of preservative, and the presence or absence of preservative, patients may need to try several formulations to find one that best relieves their ocular surface symptoms.
• The ideal artificial lubricant should be preservative-free, have a long retention time on the ocular surface, and complement the salts found in normal tears.
• Antiinflammatory agents may provide useful therapeutic approaches to DED.
Sodium–hyaluronate-containing solutions are often the first choice for DED treatment in other parts of the world, but none is currently approved in the United States (see Sodium Hyaluronate Eye Drop Solutions). Sodium hyaluronate is listed as an inactive ingredient in several US over-the-counter formulations intended to lubricate the eyes.
BIOLOGIC TEAR SUBSTITUTES
Autologous serum contains growth factors, vitamins, and fibronectin, and it can help improve ocular surface integrity. Autologous serum drops have been reported to improve ocular symptoms and objective measures in patients with DED associated with Sjögren syndrome and graft-versus-host disease.,5,6 Autologous serum eye drops must be compounded for each patient, although the FDA does not currently have a protocol in place for this process. Use of autologous serum eye drops is contraindicated in patients with active bloodborne infections.
A number of authors have reported the utility of autologous serum in treatment of ocular surface disease; however, a Cochrane review in 2013 showed a paucity of randomized clinical trials demonstrating this effect.7 Well-planned randomized controlled studies are warranted to assess the benefit of autologous serum therapy for dry eye.
ANTIINFLAMMATORY TREATMENTS
Although the root causes of DED are multifactorial and complex, it is increasingly apparent that inflammation plays a key role. Accordingly, antiinflammatory agents may provide useful therapeutic approaches to DED.8
Cyclosporine A. This fungal-derived T-cell inhibitor results in the selective inhibition of interleukin-1 (IL-1), an inflammatory cytokine. In the United States, cyclosporine A is commercialized as Restasis (0.05% cyclosporine; Allergan) and is available in preservative-free unit-dose vials. Restasis is indicated “to increase tear production in patients whose tear production is presumed to be suppressed due to ocular inflammation associated with keratoconjunctivitis sicca.”9 Given its FDA approval and the lack of alternatives, Restasis is typically the next treatment used if artificial tears or lubricants are not sufficient to quell symptoms of DED. Because cyclosporine targets T-cells, it may take months for patients to appreciate symptom improvement. A burning sensation upon application prevents some patients from continuing its use.
Preservative-Free Preparations Available Outside the United States*
Hydroxypropylmethylcellulose
Carbomers
Polyvinyl alcohol
Sodium chloride
Carboxymethylcellulose sodium
Hydroxyethylcellulose
Sodium hyaluronate
* These products are typically intended for use in patients with severe symptoms who need application more than six times per day, in those who use soft contact lenses, and in those who develop irritation from artificial tear products.1
1. Dry eyes. Patient.co.uk website. http://www.patient.co.uk/doctor/dry-eyes-pro. Accessed May 11, 2015.
Corticosteroids. These can be effective antiinflammatory therapies in DED; however, they are typically used only for short-term pulse treatments due to the risk of elevated IOP, infection, and cataract formation. In a 4-week, double-masked, randomized study in 64 patients with keratoconjunctivitis sicca and delayed tear clearance, loteprednol etabonate 0.5% ophthalmic suspension (Lotemax; Bausch + Lomb) four times daily was found to be more effective than its vehicle in improving DED signs and symptoms.10
Corticosteroids with less potential to raise IOP (eg, fluorometholone and loteprendol etabonate) should be considered. Low-dose nonpreserved formulations (methylprednisolone 1%, dexamethasone 0.01%) have been utilized after preparation at compounding pharmacies, but these are considered off-label indications in the United States.
Tetracyclines. With antiinflammatory and antibacterial properties, tetracyclines may be useful for the management of DED. At high concentrations, tetracyclines inhibit staphylococcal exotoxin-induced cytokines and chemokines. These agents may be especially effective in patients with ocular rosacea due to their inhibitory action against matrix metalloproteinase expression.
The optimal dosing schedule for tetracycline use in DED has not been established. Some have proposed use of a low dose of doxycycline (40 mg daily) for treatment of chronic blepharitis on a long-term basis. Most often, this therapeutic approach is used if there is evidence of meibomian gland dysfunction.11
Sodium Hyaluronate Eye Drop Solutions
Artelac Spash
(Bausch + Lomb)
Artelac Rebalance
(Bausch + Lomb)
Blink Intensive
(Abbott Medical Optics)
Clinitas
(Altacor)
Hyabak
(Spectrum Théa)
Hylo-Care
(Scope Ophthalmics)
Hylo-Forte
(Scope Ophthalmics)
Hylo-Tear
(Scope Ophthalmics)
Lumecare
(Lumecare)
Lubristil
(Moorefields Pharmaceuticals)
Ocusan
(Agepha Pharma)
Oxyal
(Bausch + Lomb)
Vismed
(TRB Chemedica)
Essential fatty acids. Essential fatty acids, including omega-6 and omega-3, cannot be synthesized by vertebrates and must be obtained from dietary sources. Omega-6 fatty acids are precursors for arachnidonic acid and certain proinflammatory lipid mediators. In contrast, certain omega-3 fatty acids (eg, eicosapentaenoic acid [EPA], found in fish oil) inhibit the synthesis of inflammatory mediators including IL-1 and tumor necrosis factor-alpha (TNF-α).12
Interest in omega-3 and omega-6 fatty acids in DED treatment has resulted in the use of nutritional supplements containing EPA to target dry eyes, as well as evaluation in several small studies. However, there is no strong empirical data from a well-controlled randomized controlled trial supporting the use of EPA for DED. The Dry Eye Assessment and Management (DREAM) study is a multicenter, double-masked, randomized controlled clinical trial, sponsored by the US National Eye Institute, to evaluate the effectiveness and safety of supplementation with omega-3 fatty acids in treatment of patients with moderate to severe dry eyes.13
OTHER APPROACHES
Several brands of punctal plugs are available, and these are used preferably for permanent closure of the punctum. Their principal use is to increase tear retention time on the ocular surface, although they may also increase the staying time of inflammatory mediators on the surface. Use of plugs does allow testing of their efficacy, as they can typically be easily removed if the design includes a cap resting on the top of the punctum.
Secretogogues, or tear-stimulating agents, are usually reserved for patients with severe DED. Two oral cholinergic agonists, pilocarpine and cevimeline, have been approved by the FDA to treat dry mouth in patients with Sjögren syndrome. However, they are rarely prescribed by ophthalmologists and more typically used by rheumatologists. These medications bind to muscarinic receptors, which stimulate salivary and sweat gland secretion. They can improve tear production, although most studies show greater improvement in dry mouth than in dry eyes. Patients taking pilocarpine 5 mg orally four times daily demonstrated improved ease of reading and symptoms of blurred vision compared with placebo.14 The most common side effect was excessive sweating, which occurred in more than 40% of patients.
CONCLUSION
As researchers are discovering more about the pathophysiology behind DED, there has been a paradigm shift in dry eye management. Although lubricating the ocular surface with artificial tears remains a mainstay of treatment, other strategies have emerged that may better address patients’ signs and symptoms of DED. Initiating treatment early may improve and maintain quality of life and prevent potentially vision-degrading effects of the disease. n
1. 2007 Report of the Dry Eye WorkShop. http://www.tearfilm.org/dewsreport/index.html. Accessed May 28, 2015.
2. Miljanovic B, Dana MR, Sullivan DA, Schaumberg DA. Prevalence and risk factors for dry eye syndrome among older men in the United States. Presented at: Association for Research in Vision and Ophthalmology Annual Meeting; May 6-10, 2007, Fort Lauderdale, FL.
3. Schaumberg DA, Sullivan DA, Buring JE, Dana MR. Prevalence of dry eye syndrome among US women. Am J Ophthalmol. 2003:136;318-326.
4. Herantis website. Cis-UCA eye drops for dry eye. http://herantis.com/pipeline/cis-uca-for-dry-eye/. Accessed May 27, 2015.
5. Tsubota K, Goto E, Fujita H, et al. Treatment of dry eye by autologous serum application in Sjogren’s syndrome. Br J Ophthalmol. 1999;83:390-395.
6. Chiang CC, Lin JM, Chen WL, Tsai YY. Allogeneic serum eyedrops for the treatment of severe dry eye in patients with chronic graft-versus-host disease. Cornea. 2007;26:861-863.
7. Pan Q, Angelina A, Zambrano A, et al. Autologous serum eye drops for dry eye. Cochrane Database Syst Rev. 2013:8:CD009327. doi: 10.1002/14651858.CD009327.pub2.
8. Wei Y, Asbell PA. The core mechanism of dry eye disease is inflammation. Eye Contact Lens. 2014;40(4):248-256.
9. Restasis (cyclosporine ophthalmic emulsion) 0.05% [package insert]. Irvine CA: Allergan; revised June 2013.
10. Pflugfelder SC, Maskin SL, Anderson B, et al. A randomized, double-masked, placebo-controlled, multicenter comparison of loteprednol etabonate ophthalmic suspension, 0.5%, and placebo for treatment of keratoconjunctivitis sicca in patients with delayed tear clearance. Am J Ophthalmol. 2004;138:444-457.
11. Yoo SE, Lee DC, Chang MH. The effect of low-dose doxycycline therapy in chronic meibomian gland dysfunction. Korean J Ophthalmol. 2005;19:258-263.
12. Rosenberg ES, Asbell PA. Essential fatty acids in the treatment of dry eye. Ocul Surf. 2010;8(1):18-28.
13. Dry Eye Assessment and Management Study (DREAM). https://clinicaltrials.gov/ct2/show/NCT02128763. Accessed May 11, 2015.
14. Vivino FB, Al-Hashimi I, Khan Z, et al. Pilocarpine tablets for the treatment of dry mouth and dry eye symptoms in patients with Sjogren syndrome: a randomized, placebo-controlled, fixed-dose, multicenter trial. P92-01 Study Group. Arch Intern Med. 1999;159(2):174-181.
Penny A. Asbell, MD, FACS, MBA
• Professor of Ophthalmology, Director of Cornea and Refractive Services and Director of the Cornea Fellowship Program, Department of Ophthalmology, Icahn School of Medicine at Mount Sinai, New York
• penny.asbell@mssm.edu
• Financial disclosure: Consultant (Alcon, R-tech Ueno, National Institutes of Health, Research to Prevent Blindness, Martin and Toni Sosnoff Foundation)
An Vo, MD
• Fellow in Cornea and Refractive Surgery, Department of Ophthalmology, Icahn School of Medicine at Mount Sinai, New York
• an.vo@mssm.edu
• Financial disclosure: None
