Cataract is a common complication of uveitis,1,2 arising as a result of the intraocular inflammation itself or chronic corticosteroid use or both.3 The success rate in eyes with these conditions concomitantly has improved immensely.4 Nonetheless, visual recovery may still be limited by the underlying uveitic condition.5
The visual outcome in eyes with juvenile idiopathic arthritis-associated uveitis, for example, may be poor due to the development of band keratopathy, extensive posterior synechiae (PS), cyclitic membrane, cystoid macular edema (CME), glaucoma, or postoperative hypotony.6-8 Therefore, the management of cataracts in these uveitic eyes requires meticulous attention to the control of inflammation perioperatively, in consultation with a uveitis specialist as appropriate, so as to minimize the risks of complications such as postoperative exacerbation of the inflammation and CME.9
As much as possible, the eye should be quiescent for at least 3 months prior to cataract surgery; in Behçet disease, 6 months would be preferred.9,10 In the event that surgery must be carried out urgently, as in the case of an intumescent cataract, and the inflammation cannot be adequately suppressed, intravenous methylprednisolone (1 g) may be given 1 day prior to surgery.
Even when quiescence has been achieved, additional corticosteroid prophylaxis should be given perioperatively in eyes that have previously had CME or certain at-risk uveitis syndromes such as Vogt-Koyanagi-Harada disease, Behçet disease, and birdshot retinochoroidopathy. This may be in the form of topical prednisolone acetate 1% every 2 hours for 2 days prior to surgery with oral and topical NSAIDs; oral (1 mg/kg per day for 3 days preoperatively) or periocular (40 mg/1 mL) prednisolone; or intraoperative intravitreal triamcinolone acetonide (4 mg/0.1 mL).11-14 An intravitreal dexamethasone implant (Ozurdex; Allergan) may be placed at the time of surgery in eyes that require long-term immunosuppressive therapy in patients who are steroid nonresponders.
Preoperative prophylaxis may also be required in eyes with infectious uveitis, such as ocular toxoplasmosis or herpes simplex uveitis, as surgery may trigger a reactivation of the infection.15
CONCOMITANT CONDITIONS
The evaluation of concomitant complications is crucial to optimizing outcomes in these eyes, as a combined or staged surgical approach may be required if there is coexisting corneal, glaucomatous, or retinal pathology.16,17 Comanagment with a glaucoma specialist or vitreoretinal surgeon may be helpful. In particular, for eyes with intermediate uveitis, simultaneous vitrectomy and cataract surgery enables better control of the intraocular inflammation and resolution of any persistent CME.18
A comprehensive assessment of preexisting complications also improves the surgeon's ability to suitably counsel and manage these patients' expectations, especially today when improvements in technology have elevated their demands. Most patients will expect an IOL to be implanted, and uveitis is no longer an absolute contraindication to IOL implantation if the inflammation has been adequately controlled.22-26 However, if there is severe vitreoretinal pathology or uncontrolled inflammation, it may be prudent to defer IOL implantation until the retinal issues or inflammation have been adequately managed.
AT A GLANCE
- Management of cataract in uveitic eyes requires meticulous attention to the control of inflammation perioperatively, in consultation with a uveitis specialist as appropriate.
- Uveitic eyes should be quiescent for at least 3 months prior to cataract surgery.
- In the presence of severe vitreoretinal pathology or uncontrolled inflammation, it may be prudent to defer IOL implantation until the retinal issues or inflammation have been adequately managed.
- An exaggerated inflammatory response postoperatively should be anticipated in eyes with chronic uveitis and should be treated promptly with periocular or intravitreal corticosteroids and/or escalation of systemic immunosuppressants as indicated.
Acrylic square-edged IOLs are preferred for their better biocompatibility and reduced likelihood of posterior capsular opacification (PCO).27 In these eyes, in which pupils are often small and the capsulorrhexis opening may also be small, one-piece hydrophobic acrylic IOLs are easier to insert compared with one-piece plate-haptic hydrophilic IOLs. Silicone IOLs are associated with higher incidences of postoperative CME, development of PS, pupillary membrane formation, and PCO, and they may also impede visualization during subsequent vitrectomy.
The risk of suboptimal quality of vision with multifocal and/or toric IOLs must also be explained and emphasized to patients who have posterior segment complications or in whom progressive fibrosis of the capsular bag is expected.
SURGICAL TECHNIQUE
Choice of cataract removal technique. Phacoemulsification is the preferred method of nucleus removal in these eyes due to superior anterior chamber stability and a lower incidence of iris trauma, postoperative inflammation, and induced astigmatism compared with manual extracapsular techniques.28,29 However, a manual technique may be safer if there are significant comorbidities such as corneal opacities, small pupils, or zonular weakness.
Topical anesthesia is not adequate in patients with uveitis, as these surgeries are likely to be prolonged and iris manipulation is expected. Rather a regional block or intracameral injection of preservative-free lidocaine 1% should be used. General anesthesia is required in children or adults who cannot tolerate prolonged surgery under sedation. Careful attention must be paid to patients with ankylosing spondylitis, as they may have fixed flexion deformities of the cervical spine. In such cases, a Trendelenburg position facilitates ergonomic surgical access to the eye.30
Incisions. A clear corneal tunnel approach gives better access, especially for eyes with shallow anterior chambers, and reduces the risk of surgically induced necrotizing scleritis in eyes with scleritis. On the other hand, a scleral incision should be used in eyes that have had previous peripheral corneal melts. Unless the nucleus is hard and/or large, a 2.2-mm incision with a length 70% of its width gives an optimal balance between efficiency of nucleus removal and anterior chamber stability.31
Small pupil management. A two-stage approach may be required to achieve an adequately sized pupil if PS, pupillary membranes, or dilator dysfunction is present.
If the pupil fails to dilate with intracameral adrenaline (0.5 mL of 1:1,000 adrenaline in 500 mL) and/or preservative-free intracameral lidocaine 1%, and/or if PS or membranes are present, an adaptive ophthalmic viscosurgical device (OVD) such as Healon5 (Abbot Medical Optics) can be used to free any peripheral anterior synechiae prior to using it to release any PS and to stretch the pupil. If the PS are too adherent or there is extensive membrane formation, a 27-gauge cystotome may be used instead. Iris manipulators such as Kuglen hooks and intraocular forceps such as the Ahmed micrograsper (MicroSurgical Technology) can also aid in pupil stretching and membrane peeling and removal (Figure 1). If these measures fail, there are other options for dilating the pupil (see Alternative Means for Pupil Dilation).
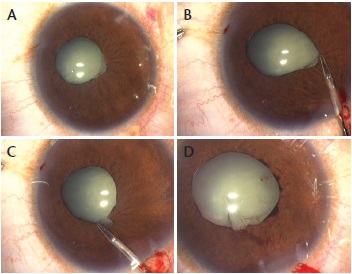
Figure 1. Eye with pupillary membranes (A); the membrane is grasped with a pair of intraocular forceps (B) and gently freed from the pupil margin (C), resulting in a significantly larger pupil (D).
Continuous curvilinear capsulorrhexis. A capsular stain such as trypan blue dye may be used to enhance visualization of the capsule during capsulorrhexis creation if there is corneal pathology or an intumescent lens. If significant zonular weakness is present, iris hooks or a two-handed technique can help to stabilize the lens and provide countertraction.32
The capsulorrhexis should not be smaller than 4 mm in diameter to avoid capsular phimosis, nor should it be larger than 5.5 mm to reduce the likelihood of iris chafing during removal of the nuclear fragments and of PCO.
Nucleus management. The capsule and zonules in these eyes may be more fragile than usual, and a capsular tension device (CTD) may be inserted prior to nucleus removal to minimize floppiness of the posterior capsule. The choice of the device is determined by the extent of zonular weakness, and more than one type may be required.
To facilitate insertion, an OVD can be used to gently open a small segment of the subcapsular space following creation of the capsulorrhexis. The presence of the intact nucleus serves to keep the bag open, and this allows smooth insertion of the CTD. Placement in the subcapsular space minimizes the impact on cortex removal. Self-retaining iris hooks may also be placed to give more support to the capsular bag if there is diffuse zonular weakness or more than 6 clock hours of zonular loss.
Alternative Means for Pupil Dilation
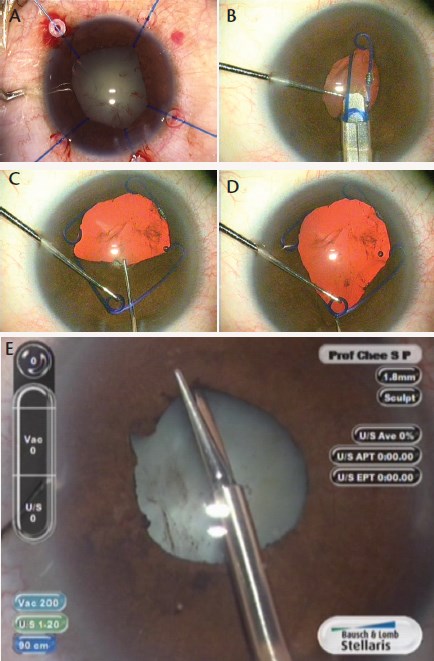
• Use of a Beehler pupil dilator, with two or three prongs
• Placement of self-retaining iris hooks (A)
• Implantation of a Malyugin Ring (B through D)
• Creation of multiple small sphincterotomies with intraocular scissors, taking care to avoid large cuts as the free edges may be further traumatized during the phacoemulsification process (E)
Hydrodissection should be performed gently or omitted altogether in hard cataracts, as aggressive hyrodissection may worsen any existing zonular weakness. In soft cataracts, hydrodelamination may be performed instead to protect the zonules and capsule. In hard cataracts, use of an in situ vertical quick-chop technique can reduce the risk of trauma to the iris and zonules. If the nucleus cannot be adequately freed due to zonular deficiencies, a lamellar chop combined with a cross chop is an alternative technique.33
The femtosecond laser is a valuable tool in these eyes, as it is able to create an adequately sized and centered capsulorrhexis even in the presence of zonular weakness. A small pupil may be enlarged prior to performing the capsulorrhexis. The Malyugin Ring (MicroSurgical Technology) is particularly helpful in achieving a large-enough pupil. Moreover, by fragmenting and softening the nucleus, the femtosecond laser reduces the phacoemulsification energy required as well as the amount of stress on the zonules.
Cortical cleanup and IOL implantation. Any residual cortical material increases the risk of exaggerated postoperative inflammation and PCO. If the pupil is small, rotation of the eye helps to locate any cortical remnants. A mini-aspiration cannula can be used via a sideport incision to facilitate removal of the subincisional cortical material. This is especially helpful when a femtosecond laser was used to create the capsulorrhexis, as in this situation the leverage afforded by the usual frill of cortex is lost. If cortical material is trapped by a CTD, it should be freed by stripping it in a tangential direction rather than toward the center, as this may displace the CTD or further damage the zonules.
Removal of OVD from behind the IOL encourages its adhesion to the posterior capsule, thereby reducing the risk of PCO.24 However, eyes with juvenile idiopathic arthritis-associated uveitis have a higher propensity to PCO, and a Nd:YAG laser capsulotomy may be difficult in a young child. Therefore, after the IOL has been placed in the capsular bag, a primary posterior capsulorrhexis combined with limited anterior vitrectomy may be performed in these eyes. The increased risks of postoperative endophthalmitis, CME, and retinal detachment that accompany these steps should be discussed preoperatively with the patient's parents.8
POSTOPERATIVE MANAGEMENT
An exaggerated inflammatory response should be anticipated in eyes with chronic uveitis and treated promptly with periocular or intravitreal corticosteroids and/or escalation of systemic immunosuppressants as indicated. Transient elevations of IOP, another expected event in the early postoperative period, can usually be controlled with antiglaucomatous medications.
Postoperative hypotony is of greater concern. If it persists despite maximizing the immunosuppressive regime, cyclitic membranes causing ciliary body detachment must be excluded or managed accordingly if present.28
CONCLUSION
Advances in microsurgical techniques, including the use of LACS, coupled with the greater armamentarium of therapy now available to control inflammation perioperatively, have enhanced the outcomes of cataract surgery in eyes with uveitis. n
1. Malinowski SM, Pulido JS, Folk JC. Long term visual outcome and complications associated with pars planitis. Ophthalmology. 1993;100:818-824.
2. Velilla S, Dios E, Herraras JM, Calonge M. Fuchs' heterochromic iridocyclitis; a review of 26 cases. Ocular Immunol Inflamm. 2001;9:169-175.
3. Hooper PL, Rao NA, Smith RE. Cataract extraction in uveitis patients. Surv Ophthalmol. 1990;35:120-144.
4. Foster CS, Rashid S. Management of coincident cataract and uveitis. Curr Opin Ophthalmol. 2003;14:1-6.
5. Javadi MA, Jafarinasab MR, Araghi AA, et al. Outcomes of phacoemulsification and in-the-bag intraocular lens implantation in Fuchs' heterochromic iridocyclitis. J Cataract Refract Surg. 2005;31:997-1001.
6. Kanski JJ, Shun-Shin GA. Systemic uveitis syndrome in childhood: an analysis of 340 cases. Ophthalmology.1984;91:1247-1252.
7. Foster CS, Barett F. Cataract development and cataract surgery in patients with juvenile rheumatoid arthritis-associated iridocyclitis. Ophthalmology. 1993;100:809-817.
8. Flynn HW Jr, Davis JL, Culbertson WW. Pars plana lensectomy and vitrectomy for complicated cataracts in juvenile rheumatoid arthritis. Ophthalmology. 1988;95:1114-1119.
9. Belair ML, Kim SJ, Thorne JE, et al. Incidence of cystoid macular edema after cataract surgery in patients with and without uveitis using optical coherence tomography. Am J Ophthalmol. 2009;148:128-135.
10. Matsuo T, Takahashi M, Inoue Y, et al. Ocular attacks after phacoemulsification and intraocular lens implantation in patients with Behçet disease. Ophthalmologica. 2001;215:179-182.
11. Herbort CP, Jauch A, Othenin-Girard P, et al. Diclofenac drops to treat inflammation after cataract surgery. Acta Ophthalmol Scand. 2000;78:421-424.
12. Jonas JB. Intravitreal triamcinolone acetonide: a change in a paradigm. Ophthalmic Res. 2006;38:218-245.
13. Okhravi N, Morris A, Kok HS, et al. Intraoperative use of intravitreal triamcinolone in uveitic eyes having cataract surgery: pilot study. J Cataract Refract Surg. 2007;33:1278-1283.
14. Dada T, Dhawan M, Garg S, et al. Safety and efficacy of intravitreal injection of triamcinolone acetonide injection after phacoemulsification in cases of uveitic cataract. J Cataract Refract Surg. 2007;33:1613-1618.
15. Bosch-Drissen LH, Plaiser MB, Stilma JS, et al. Reactivation of ocular toxoplasmosis after cataract extraction. Ophthalmology. 2002;109:41-45.
16. Tran VT, LeHoang P, Herbort CP. Value of high-frequency ultrasound biomicroscopy in uveitis. Eye (Lond). 2001;15:23-30.
17. Gupta P, Gupta A, Gupta V, Singh R. Successful outcome of pars plana vitreous surgery in chronic hypotony due to uveitis. Retina. 2009;29:638-643.
18. Tranos P, Scott R, Zambarakji H, et al. The effect of pars plana vitrectomy on cystoid macular oedema associated with chronic uveitis: a randomised, controlled pilot study. Br J Ophthalmol. 2006;90:1107-1110.
19. Markomichelakis NN, Halkiadakis I, Pantelia E, et al. Patterns of macular edema in patients with uveitis: qualitative and quantitative assessment using optical coherence tomography. Ophthalmology. 2004;111:946-945.
20. De Laey JJ. Fluorescein angiography in posterior uveitis. Int Ophthalmol Clin. 1995;35:33-58.
21. Palestine AG, Alter GJ, Chan CC, Nussenblatt RB. Laser interferometry and visual prognosis in uveitis. Ophthalmology. 1985;92:1567-1569.
22. Probst LE, Holland EJ. Intraocular lens implantation in patients with juvenile rheumatoid arthritis. Am J Opthalmol. 1996;122:161-170.
23. Quinones K, Cervantes Castaneda RA, Hynes AY, et al. Outcome of cataract surgery in children with chronic uveitis. J Cataract Refract Surg. 2009;35:725-731.
24. Sijssens KM, Los LI, Rothova A, et al. Long-term ocular complications in aphakic versus pseudophakic eyes of children with juvenile idiopathic arthritis-associated uveitis. Br J Ophthalmol. 2010;94:1145-1149.
25. Ganesh SK, Sudharshan S. Phacoemulsification with intraocular lens implantation in juvenile idiopathic arthritis. Ophthalmic Surg Lasers Imaging. 2010;41:104-108.
26. Grajewski RS, Zurek-Imhoff B, Roesel M, et al. Favourable outcome after cataract surgery with IOL implantation in uveitis associated with juvenile idiopathic arthritis. Acta Ophthalmol. 2012;90:657-662.
27. Alió JL, Chipont E, BenEzra D, Fakhry MA; International Ocular Inflammation Society, Study Group of Uveitic Cataract Surgery. Comparative performance of intraocular lenses in eyes with cataract and uveitis. J Cataract Refract Surg. 2002;28:2096-2108.
28. Pande MV, Spalton DJ, Kerr-Muir MG, Marshall J. Postoperative inflammatory response to phacoemulsification and extracapsular cataract surgery: aqueous flare and cells. J Cataract Refract Surg. 1996;22 Suppl 1:770-774.
29. Chee SP, Ti SE, Sivakumar M, Tan DT. Postoperative inflammation: extracapsular cataract extraction versus phacoemulsification. J Cataract Refract Surg. 1999;25(9):1280-1285.
30. Chee SP. Positioning patients with severe kyphosis during cataract surgery. J Cataract Refract Surg. 2010;36:366-367.
31. Chee SP, Ti SE, Lim L, Chan AS, Jap A. Anterior segment optical coherence tomography evaluation of the integrity of clear corneal incisions: a comparison between 2.2-mm and 2.65-mm main incisions. Am J Ophthalmol. 2010;149(5):768-776.
32. Chee SP, Jap A. Management of traumatic severely subluxated cataracts. Am J Ophthalmol. 2011;151(5):866-871.
33. Chee SP. Management of the hard posterior polar cataract. J Cataract Refract Surg. 2007;33:1509-1514.
Soon-Phaik Chee, MD
- Senior Consultant Ophthalmic Surgeon, Head of Cataract Service and Ocular Inflammation Service, Singapore National Eye Centre, Singapore
- Professor, Department of Ophthalmology, Yong Loo Lin School of Medicine, National University of Singapore, Singapore
- Professor, Duke-National University of Singapore Graduate Medical School, Singapore
- Head of Ocular Inflammation Research Group, Singapore Eye Research Institute, Singapore
- chee.soon.phaik@snec.com.sg
- Financial disclosure: None
Aliza Jap, MD
- Senior Consultant Ophthalmic Surgeon, Singapore National Eye Centre, Singapore
- Senior Consultant Ophthalmic Surgeon, Division of Ophthalmology, Changi General Hospital, Singapore
- Financial disclosure: None
