


The original inspiration for endothelial keratoplasty was the realization that, no matter how expertly performed, PKP—with its full-thickness incisions—almost necessarily invited a slew of difficult postoperative challenges, including allograft reaction and graft rejection, irregular and fluctuating astigmatism, and a high steroid burden, among many others. Solving these problems required a fundamentally different approach to the management of endothelial disorders, necessitating the development of new surgical techniques. It may be that solving these same problems for patients with keratoconus will likewise require new surgical approaches. Bowman layer (BL) transplantation, a new treatment option for patients with advanced keratoconus, may prove to be useful in this regard.
AT A GLANCE
- Bowman layer (BL) transplantation represents a new treatment option for patients with advanced keratoconus; the operation entails the implantation of an isolated BL into the midstroma of a keratoconic cornea.
- The procedure may offer a unique opportunity to stabilize the cornea, preserve contact lens tolerance, and avoid more invasive alternatives.
- In a cohort of 22 eyes with more than 4 years of follow-up, recipient corneas flattened by an average 8.00 D; 90% experienced a cessation of ectatic progression; and BCVA improved by 2 Snellen lines in approximately 40% of patients, by 1 line in about 33% of patients, was unchanged in about 20% of patients, and fell by 1 line in one patient.
BL transplantation entails the implantation of an isolated BL into the midstroma of a keratoconic cornea. The implanted tissue functions to flatten the recipient cornea into a more normal shape and to stiffen it against further ectasia. The primary advantage of the procedure is that it seems to arrest the progression of keratoconus, thereby preserving, and in some cases restoring, contact lens tolerance. As a result, many patients—even with very advanced keratoconus—may achieve satisfactory and stable vision and thereby postpone or avoid more invasive surgical alternatives such as PKP or deep anterior lamellar keratoplasty (DALK).1-4
POTENTIAL ADVANTAGES
In contrast, with both PKP and DALK, BL transplantation is a sutureless procedure. The graft is also acellular, so the risk of allograft reaction may be significantly reduced; this may also permit a substantially lower postoperative steroid burden, thereby lessening the occurrence of steroid-related complications including cataract formation and glaucoma. Because the surgical manipulations of BL transplantation are confined to a pocket within the recipient cornea, the operation may also be regarded as an extraocular procedure, entailing fewer risks than are associated with intraocular surgery, including infectious endophthalmitis and choroidal hemorrhage or effusion.
BL transplantation may also be regarded as especially low-risk because the transplanted tissue may be removed later if necessary and because subsequent DALK and PKP are possible if the results of BL transplantation are unfavorable. Therefore, BL transplantation may represent one of the safest surgical treatments for patients with advanced, progressive keratoconus, and it may offer a unique opportunity to stabilize the cornea, preserve contact lens tolerance, and avoid more invasive alternatives.1-4
GRAFT PREPARATION
Typically, donor tissue for BL transplantation is prepared in an eye bank in the days before surgery. A corneoscleral rim is excised from a whole globe and then mounted endothelial-side–down on an artificial anterior chamber. After debriding the epithelium with a surgical spear, a 30-gauge needle is used to gently score the BL around 360°, just inside the limbus. McPherson forceps are then used to delicately peel the BL from the underlying anterior stroma using small circular motions.
After separation, the isolated BL spontaneously curls into a roll, not unlike the graft for Descemet membrane endothelial keratoplasty (DMEK), but, in this case, the epithelial side is always on the outside of the scroll (Figure 1). The donor tissue is submerged in 70% ethanol for 30 seconds to remove any lingering epithelial cells, then kept in storage solution until the time of transplantation.5,6
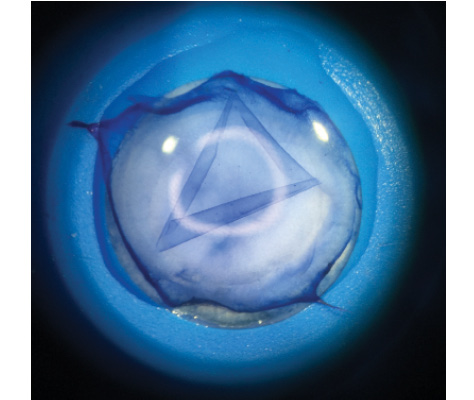
Figure 1. Isolated BL graft immediately after manual stripping.
One advantage of BL transplantation is that the tissue preparation can be performed on anterior corneal remnants after previous DMEK-graft preparation using the standardized no-touch DMEK preparation technique.6,7 As a result, from one donor cornea, two grafts may be produced: one for BL and one for DMEK. This may double the available supply of transplantable tissue compared with conventional PKP.6
One potential limitation, however, is that the donor BL graft is at present somewhat tedious and time-consuming to prepare by hand. As a result, we have investigated the possibility of automated dissection using the femtosecond laser with modified cutting parameters. Preliminary results have been encouraging; a pilot study of the technical feasibility of femtosecond BL graft creation showed that laser-cut tissues demonstrated smoother, more regular edges compared with those prepared by manual stripping, but that they were significantly thicker overall because they contained some additional amounts of anterior stroma (Figure 2).8 The optical impact of these differences in graft morphology is at present unknown.
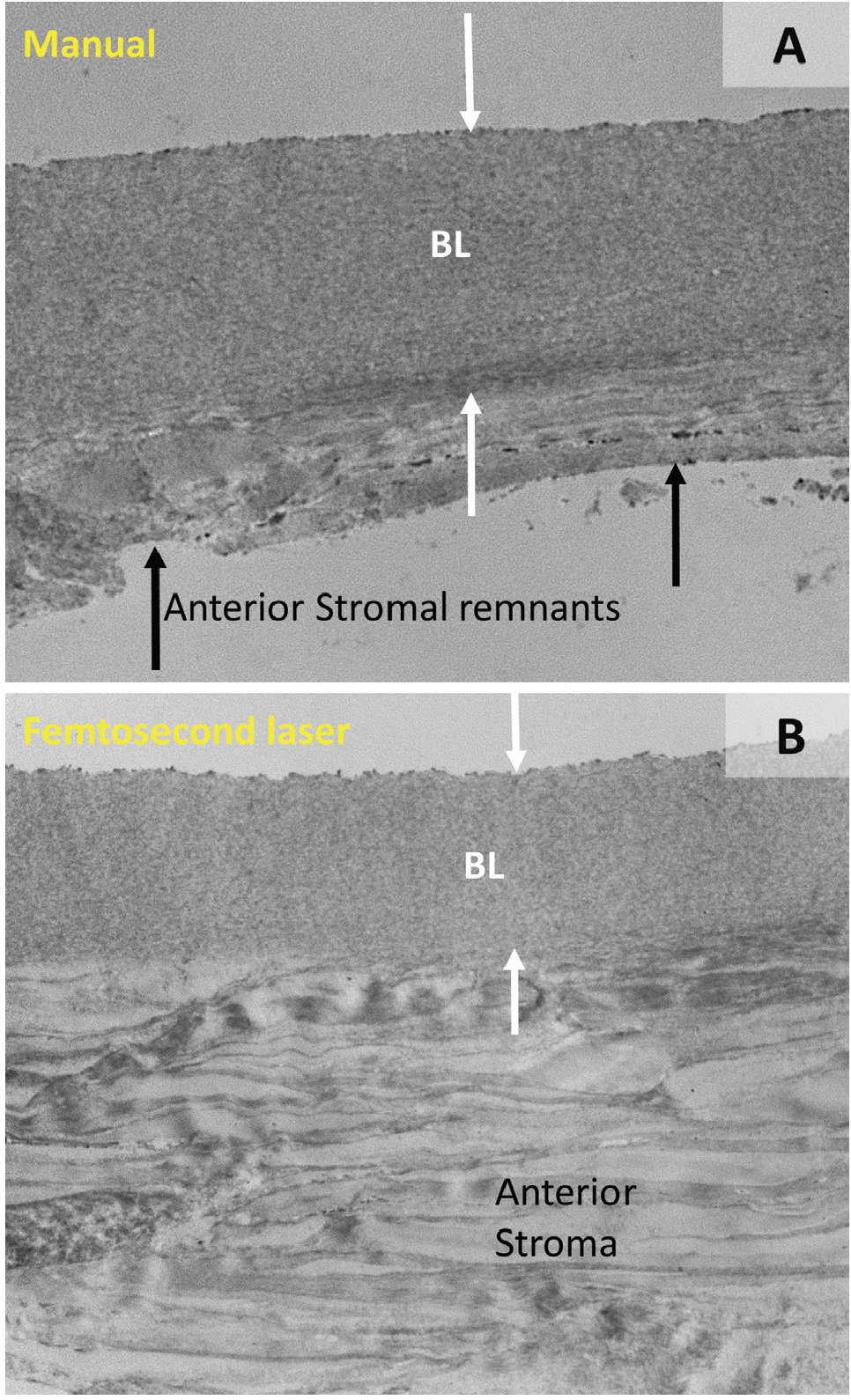
Figure 2. Transmission electron microscopy images of a BL graft prepared by manual (A) and femtosecond laser (B) dissection. Note that the femtosecond laser graft is thicker, owing to the presence of anterior stromal lamellae underneath the BL (white arrows), whereas the graft produced by manual dissection shows only BL tissue with some irregular stromal remnants (black arrows).
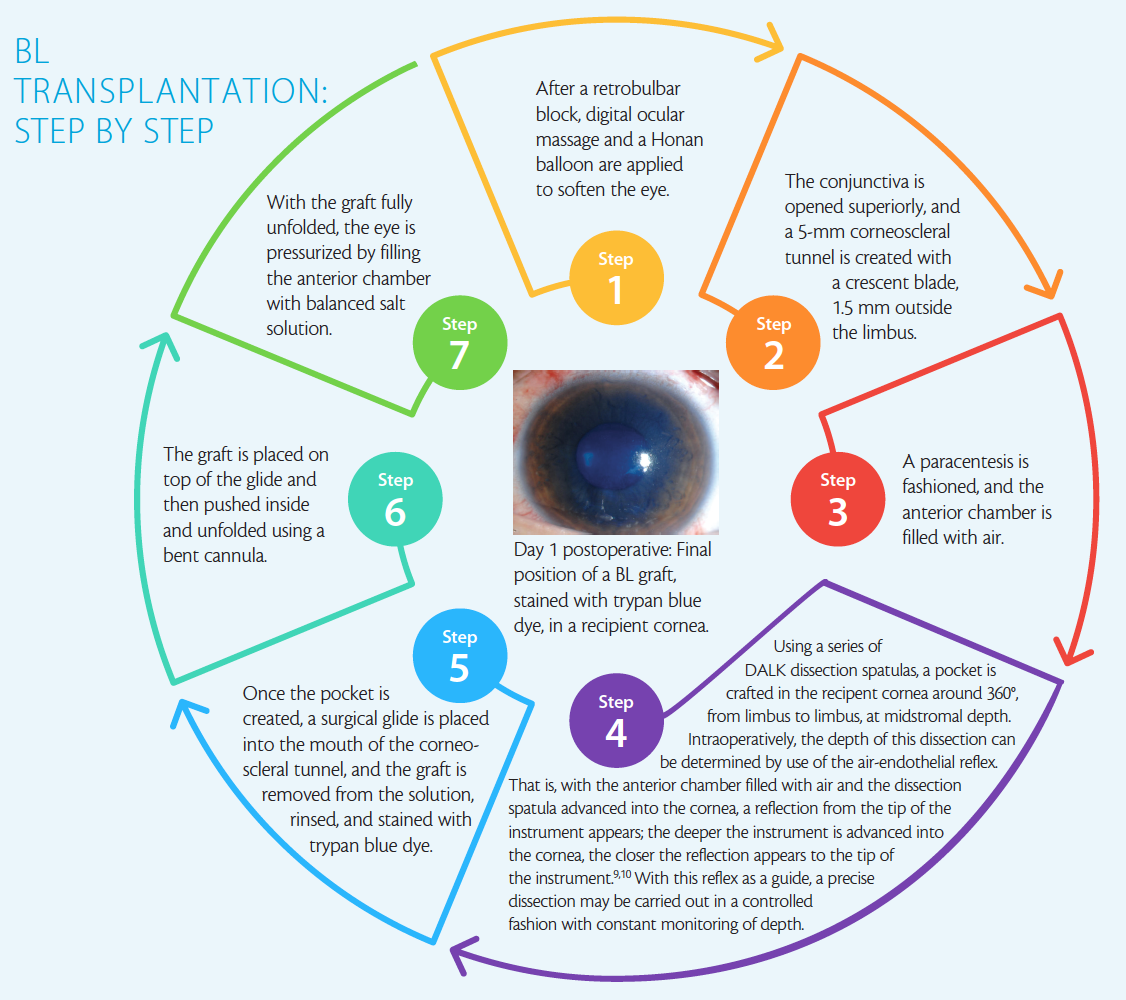
A description of the surgical procedure, which can be performed under local anesthesia, can be found in BL Transplantation: Step by Step on the following page.
RESULTS
The first cohort of patients to receive BL transplantation comprised a group of 22 eyes, now with more than 5 years of follow-up data. Inclusion criteria in the original study were a documented history of keratoconic progression and maximum keratometry (Kmax) values of 70.00 D or greater. All included patients were additionally ineligible for alternative therapies including CXL and intrastromal corneal ring segment implantation.
After surgery, recipient corneas flattened by an average 8.00 D (Kmax values), and 90% experienced a cessation of ectatic progression.2,3 All patients were able to comfortably wear scleral contact lenses after surgery, and best contact lens–corrected visual acuity was stable from pre- to postoperative, whereas best spectacle-corrected vision improved by 2 Snellen lines in approximately 40% of patients, by 1 line in about 33% of patients, was unchanged in 20%, and fell by
1 line in one patient. These gains in spectacle-corrected vision may be attributable to a reduction in higher-order corneal aberrations after surgery, which may be the product of the flattening and regularizing effect of the transplanted tissue on the corneal topography.3,4
In two of the original 22 operated eyes, surgery was aborted secondary to intraoperative perforation of Descemet membrane during attempted stromal dissection. Because no surface incisions were made, the procedures could be safely terminated and the eyes allowed to heal. One of these eyes slowly cleared, and the other was scheduled for PKP. No postoperative complications were observed. Most patients were continued on low-dose topical steroids for 1 year after surgery, and then the therapy was stopped.3
TYPICAL CASE REPORT
Since our original studies, BL transplantation has begun to spread internationally. Here we describe a recent case from the United States.
A 24-year-old black man presented with moderate (Amsler-Krumeich stage 2) bilateral keratoconus. Rigid gas permeable (RGP) contact lenses were prescribed, but the patient could not tolerate the lenses. Symmetrical superior and inferior Intacs (Addition Technology) were then implanted. Three months postoperatively, in the right eye, the patient’s BCVA had improved from 20/40 (0.5) to 20/25 (0.8), and mean keratometry (Kmean) decreased by almost 2.50 D, from 44.30 to 41.90 D.
Two years later, his right eye, measured by Scheimpflug-based corneal tomography (Pentacam HR; Oculus), appeared stable (Kmean, 40.90 D; Kmax, 53.60 D). The left eye, however, seemed to be progressing: Kmean had increased from 43.60 to 45.20 D, and Kmax from 47.00 to 53.00 D. Therefore, we recommended that the patient undergo CXL in his left eye. Because the procedure was not approved by the US FDA at the time, and treatment would therefore require that he travel internationally, the patient declined and opted instead for a course of watchful waiting.
Eighteen months later, he returned for examination. Both corneas had progressed: the right eye only slightly (Kmean, 42.20 D; Kmax, 52.10 D), but the left more substantially (Kmean, 45.50 D; Kmax, 57.10 D). After 6 more months, further progression was evident: mild in the right eye (Kmean, 43.10 D; Kmax, 52.90 D) and severe in the left (Kmean, 47.30 D; Kmax, 62.90 D). Although the patient’s BCVA remained relatively good in both eyes (20/40 OD, 20/30 OS), the relentless progression of disease, particularly in the patient’s left eye, prompted us to proceed with BL transplantation in that eye.
Both manual graft preparation and surgery proceeded according to our earlier description. No intra- or postoperative complications were observed. The day after surgery, best spectacle-corrected vision had decreased from 20/30 to 20/40, where it remained stable at 1-week and 1- and 3-month follow-up visits. At the 3-month visit, refraction with an RGP contact lens was also performed, which restored visual acuity to 20/30. Likewise, by 3 months postoperatively Kmean had decreased by 1.40 D (from 47.30 to 45.90 D) and Kmax by nearly 5.00 D (from 62.90 to 58.30 D).
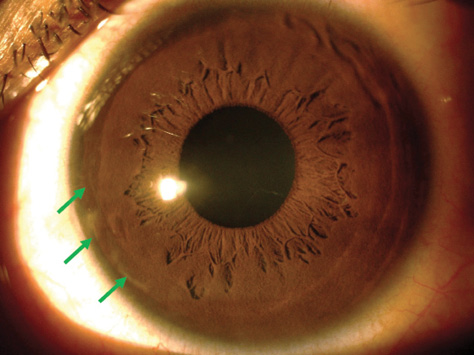
Figure 3. Three months postoperatively, the BL transplant is only faintly visible at the graft edges (green arrows).
Compared with preoperative values, the central and thinnest point corneal thicknesses were hardly affected, changing from 465 to 482 μm centrally and from 459 to 464 μm at the thinnest point. Over this same period, the average total corneal densitometry measurements (a unitless metric indicating the amount of light backscattered by the cornea) increased slightly, from 15.3 to 18.2. On Scheimpflug imaging and slit-lamp biomicroscopy, the edges of the graft have remained only barely visible as a thin line with no accompanying inflammation (Figure 3). Meanwhile, the cornea of the right eye has continued to show progression (Kmean, 43.10 D; Kmax, 55.50 D).
1. Parker JS, van Dijk K, Melles GR. Treatment options for advanced keratoconus: a review. Surv Ophthalmol. 2015;60:459-480.
2. van Dijk K, Parker J, Tong CM, et al. Midstromal isolated Bowman layer graft for reduction of advanced keratoconus: a technique to postpone penetrating or deep anterior lamellar keratoplasty. JAMA Ophthalmol. 2014;132(4):495-501.
3. van Dijk K, Liarakos V, Parker J, et al. Bowman layer transplantation to reduce and stabilize progressive, advanced keratoconus. Ophthalmology. 2015;122:909-917.
4. Luceri S, Parker J, Dapena I, et al. Corneal densitometry and higher order aberrations after Bowman layer transplantation: 1-year results. Cornea. 2016;35:959-966.
5. Lie J, Droutsas K, Ham L, et al. Isolated Bowman layer transplantation to manage persistent subepithelial haze after excimer laser surface ablation. J Cataract Refract Surg. 2010;36(6):1036-1041.
6. Groeneveld van-Beek, Parker J, Lie J, et al. Donor tissue preparation for Bowman layer transplantation. Cornea. 2016;35:1499-1502
7. Groeneveld-van Beek EA, Lie JT, van der Wees J, et al. Standardized “no-touch” donor tissue preparation for DALK and DMEK: harvesting undamaged anterior and posterior transplants from the same donor cornea. Acta Ophthalmol. 2013;91:145-150.
8. Parker J, Huls F, Cooper E, et al. Technical feasibility of isolated Bowman layer graft preparation by femtosecond laser: a pilot study [published online ahead of print May 26, 2017]. Eur J Ophthalmol.
9. Melles GR, Rietveld FJ, Beekhuis WH, Binder PS. A technique to visualize corneal incision and lamellar dissection depth during surgery. Cornea. 1999;18(1):80-86.
10. Melles GRJ, Lander F, Rietveld FJR, et al. A new surgical technique for deep, anterior lamellar keratoplasty. Br J Ophthalmol. 1999;83:327-333.


