
For many surgeons globally, handling a rock-hard nucleus in cataract surgery is a heavy challenge. The cataract has stagnated too long inside the capsular bag, growing in size and density. It has become an oversized mass inside a bag that is distended and may be unhealthy and thin. Further, it may reside behind a shallow anterior chamber and a pupil that remains small despite pharmacologic mydriasis. Therefore, it behooves the surgeon to approach the rock-hard nucleus with the right skill set and ammunition.
AT A GLANCE
• The decision whether to do phacoemulsification on a rock-hard nucleus depends on endothelial cell count, stability of the bag, and density of the cataract.
• Use of an air pump during phacoemulsification reduces the amount of phaco power needed and the phaco time required to address a hard cataract.
• Use of the tire-lever maneuver during MSICS causes the least stress on the zonules while delivering the nucleus from the posterior chamber to the anterior chamber.
• An important step in safely placing the IOL after lens removal is to limit dialing of the lens by instead dropping the IOL into the bag, using a combination of instruments.
This article presents my top 10 tips for handling a rock-hard nucleus (Figure 1): five tips for the surgeon who chooses to perform phacoemulsification, and five for the surgeon who chooses manual small-incision cataract surgery (MSICS). The first tip, in fact, helps the surgeon choose which approach to use.
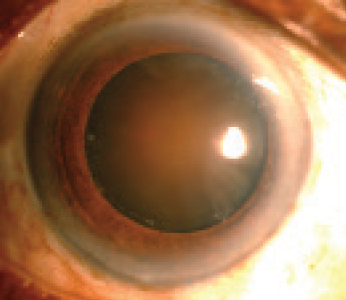
Figure 1. A rock-hard nucleus.
FOR PHACOEMULSIFICATION
Decision-making. The decision whether to do phacoemulsification on a rock-hard nucleus depends on the following factors.
- Endothelial cell count. A low endothelial cell count shifts my attention toward using MSICS instead of phacoemulsification. Using the wider opening to express the nucleus prevents collateral damage to internal structures made by the ultrasound energy, avoiding corneal damage (Figure 2).
- Stability of the bag. A weak posterior capsule or a very unstable capsular bag (Figure 3) with a hard nucleus makes me favor MSICS, as this approach avoids the risk of a dropped nucleus.
- Density of the cataract. There are clearly some cases in which one wonders how a phaco probe could ever chop that piece of rock into two, four, or eight pieces (Figure 4). The energy used to divide these nuclei would be immense and could lead to complications such as corneal damage, corneal burn, iris chafing, zonular rupture, and sometimes even bleeding due to blunt injury from the chopper.
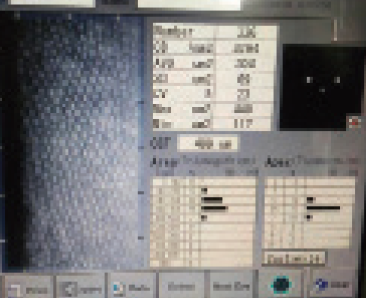
Figure 2. Endothelial cell count helps to determine whether to use phacoemulsification or MSICS.
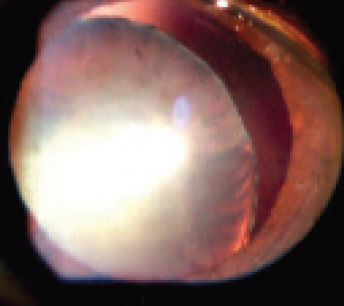
Figure 3. Stability of the bag is a second determining factor in which technique to choose.
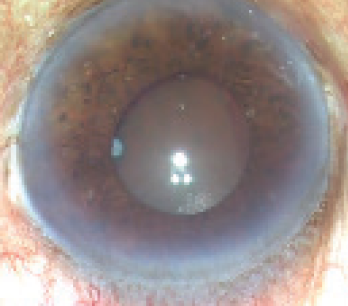
Figure 4. Density of the cataract is the third determining factor.
Visualization. One major additional problem seen in most eyes with a rock-hard nucleus is the presence of a small pupil. Intraoperative disasters can be avoided in eyes with small pupils through the use of iris hooks, iris retractors, or a Malyugin Ring (MicroSurgical Technology). Another aid to visualization is the use of trypan blue vital dye to stain the anterior capsule. With these two adjuvants, it is easier to determine the rhexis size and to visualize how deep to embed the nucleus to facilitate chopping. A good view removes any ambiguity about performing maneuvers inside the anterior chamber (Figure 5).
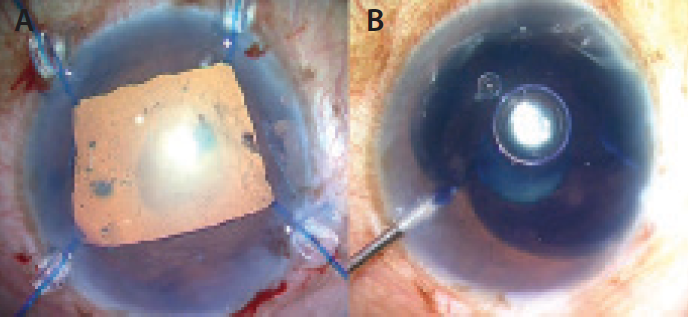
Figure 5. Visualization is aided by the use of a Malyugin Ring (A) and trypan blue dye (B).
OVD choice. The type of OVD used during phacoemulsification can determine the outcome of surgery. With a hard cataract, the OVD must serve as a barrier, preventing ultrasound energy from harming the cornea. The use of a cohesive OVD, such as Viscoat (Alcon), is always preferred over a dispersive OVD in hard cataracts.
Phaco power and the air pump. Phaco time and phaco power are inversely proportional. To understand this relationship, consider the use of an air pump and what it can accomplish. In my operating room, an air pump is connected to the irrigation bottle, forcing air into the balanced saline solution. This in turn pushes saline out of the phaco probe at a faster rate than gravity alone would accomplish. When this highly pressured phaco probe is introduced into the eye, the laws of physics say that what goes in faster must come out faster. Hence, the speed of saline entering the eye is directly proportional to the speed of the chopped pieces exiting the eye. Use of the air pump also reduces the amount of phaco power needed and the phaco time required to address a hard cataract (Figure 6).
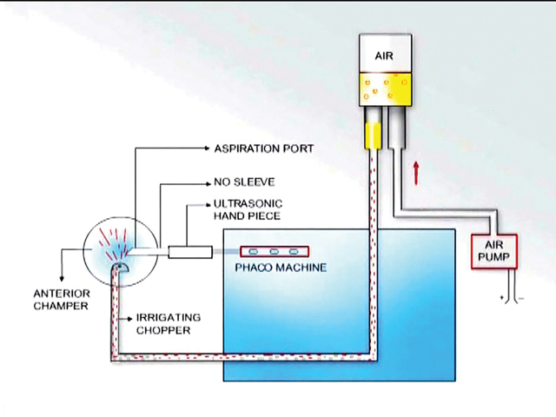
Figure 6. The air pump, hooked to the infusion bottle, allows use of less phaco power.
Break time. Performing phacoemulsification in hard cataracts requires extremely high energy levels. Continuous dispersal of this energy creates heat and, in turn, collateral damage intracamerally. A small trick that helps avoid this is to take a few breaks intraoperatively. Remove the phaco probe while doing so, and take advantage of the opportunity to apply more OVD (Figure 7).
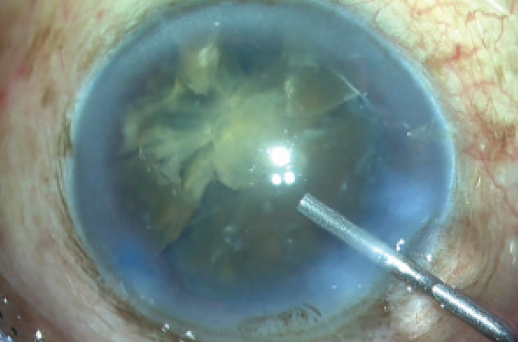
Figure 7. To reduce thermal damage, take occasional breaks and apply more OVD.
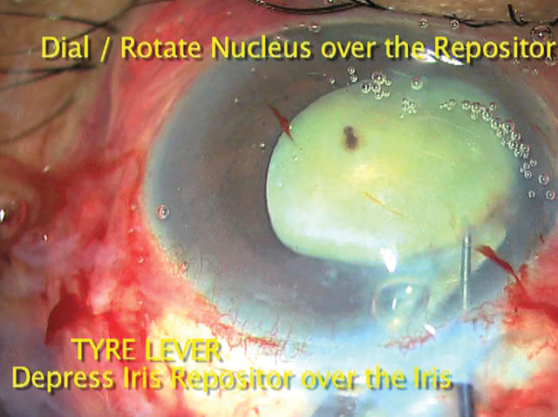
STEP-BY-STEP: THE TIRE-LEVER TECHNIQUE
• The iris repositor, held in the nondominant hand, is placed intracamerally diagonally over the right side of the iris.
• A lens dialer in the dominant hand is placed over the rock-hard cataract.
• The dialer is hooked into the cataract, and the lens is mildly rocked so that the edge of the nucleus equator can be seen.
• At this point, the repositor depresses the iris and the edge of the pupil under the lens equator.
• The dialer is then used to hook and dial the lens on top of the iris and rhexis, in any direction of the surgeon’s choice.
• Once more than 75% of the nucleus has been dialed up, an injection of OVD under the nucleus can be used to bring the rest over the iris.
FOR MSICS

Scleral tunnel construction. The scleral tunnel is specifically required in the MSICS procedure, as it creates a valve-like opening into the eye. The sutureless nature of the wound also reduces induced astigmatism. Therefore, constructing the tunnel well is of utmost importance. Following are two pointers to help you do the right thing.
- At first, point the crescent blade toward the sclerocorneal junction. Start by keeping the keel of the crescent blade flat, or parallel to the ground. This means the tip should enter the sclera 2 mm behind the limbus, pointed toward the sclerocorneal junction.
- Reach beyond the limbus. As soon as the tip of the blade reaches blue sclera, the surgeon should depress the keel down so as to direct the tip up into the corneal lamellae to reach 1 to 1.5 mm beyond the limbus (Figure 8).
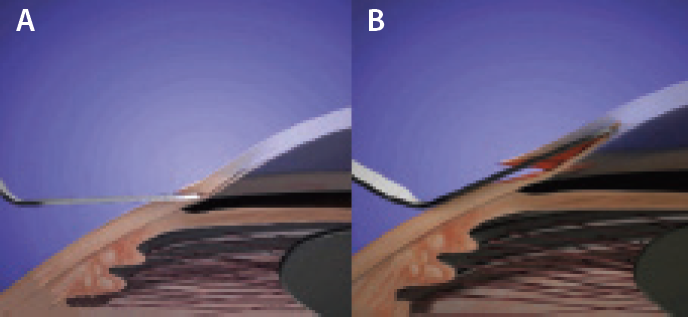
Figure 8. To begin the scleral tunnel, keep the crescent blade parallel to the ground. Enter the sclera 2 mm behind the limbus with the tip pointed toward the sclerocorneal junction (A). When the tip reaches blue sclera, direct it up into the corneal lamellae to reach 1 to 1.5 mm beyond the limbus (B).

Relaxing incisions. Create relaxing incisions on the anterior rhexis margin to help open the bag just enough to bring the large nucleus into the anterior chamber. Ideally, relaxing incisions should be placed at the 10-, 12-, and 2-o’clock positions (Figure 9). These are usually small nicks extending 0.5 mm from the rhexis margin rim. If these are not made, disastrous complications can sometimes ensue.
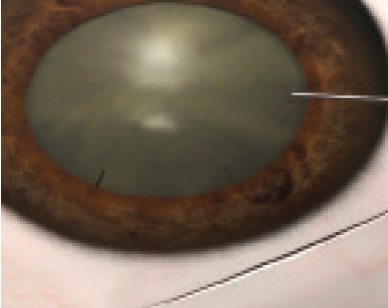
Figure 9. Place relaxing incisions at the 10-, 12-, and 2-o’clock positions to facilitate delivery of the nucleus.

Tire-lever technique. The one trick that has helped me the most in MSICS, and without which I feel no surgeon should perform this surgery, is the tire-lever technique, in which the iris manipulator or repositor is used as a lever or crowbar. Use of this maneuver causes the least stress on the zonules while delivering the nucleus from the posterior chamber to the anterior chamber. For more on the tire-lever technique, see Step by Step .

Viscoexpression. A good understanding of incision size in proportion to nucleus size is important for getting the viscoexpression maneuver right. After the nucleus is brought into the anterior chamber, a dispersive OVD is injected beneath the nucleus. While this is done, the surgeon depresses the posterior lip of the main scleral tunnel. The OVD flows to the inferior quadrant of the anterior chamber and pushes the nucleus out of the eye as the posterior lip of the wound is being depressed (Figure 10). This is because the path of least resistance is through the wound.
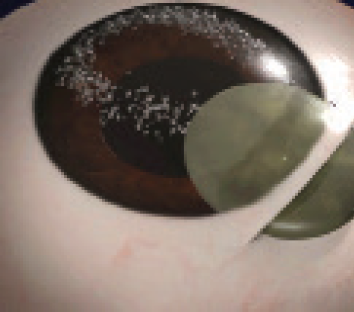
Figure 10. Viscoexpression brings the nucleus out through the scleral tunnel as the posterior lip of the incision is depressed.

Haptic drop. An important step in safely placing the IOL after lens removal is to limit dialing of the lens by instead dropping the IOL into the bag, using a combination of instruments. Dialing involves the following uncertainties.
- Haptic placement. The placement of the haptics cannot be determined if, for example, one haptic is in the capsule and the other out.
- Damage to the bag. If there is a snag during dialing, the bag can be damaged.
To avoid these problems, the IOL can be dropped into the bag. After one haptic of the IOL is placed, the other haptic is taken inside the eye with forceps and handed over to another pair of forceps in the chamber. These forceps then grasp the haptic and flex it so that it goes under the rhexis margin. The haptic can then be dropped into the bag, ensuring the correct placement of the IOL inside the bag (Figure 11).
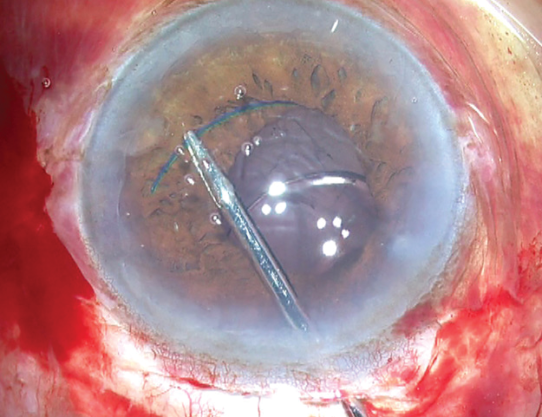
Figure 11. With one haptic already in the bag, the second haptic is grasped with forceps and dropped, rather than dialed, into the bag.
CONCLUSION
Each of these tricks has been a boon to me, and I hope that readers will incorporate the ones they feel comfortable with into their surgical routine for hard nuclei. Use of these tips has reduced my complication rate with hard nuclei by more than 85%, and this has helped to increase my referrals from other physicians by more than 60%, based purely on the safety outcomes I can achieve in this complex surgery.


