

Acute adenoviral keratoconjunctivitis (AAK) is the most common external ocular viral infection worldwide. It may present epidemically or sporadically and is highly contagious. After an incubation period of 2 to 14 days, typical symptoms, including conjunctival hyperemia and chemosis, follicular reaction, and intense tearing, usually begin in one eye (Figure 1).
AT A GLANCE
- Acute adenoviral keratoconjunctivitis (AAK) is the most common external ocular viral infection worldwide; the aim of treatment in AAK is to alleviate symptoms, but there is no single effective treatment.
- In the authors’ study to assess the use of topical 2% cyclosporine A, patients received either topical 2% cyclosporine four times daily for 21 days in the acute phase of AAK, starting right upon diagnosis, or topical corticosteroids four times daily for 1 week or only symptomatic treatment, including cold compresses and unpreserved artificial tears.
- The authors found that the use of topical 2% cyclosporine A in the early phase of AAK decreases the severity and duration of symptoms, has an inhibitory effect on the development of corneal subepithelial infiltrates, and carries the advantage of being a steroid-sparing agent.
Unfortunately, corneal involvement and impaired vision can be complications of AAK. In more than half of patients, corneal subepithelial opacities develop approximately 10 days after the onset of symptoms (Figure 2).
Visual impairment can persist for months because of subepithelial corneal infiltrates and irregular astigmatism. Patients with this complication usually make repeated office visits and are devastated by their suddenly impaired visual acuity, which can include light scattering and photophobia. The corneal infiltrates usually regress in a few weeks, but they can persist for years in rare cases. Some patients experience marked symptoms of ocular dryness, which can persist for an extended time after an episode of adenoviral conjunctival infection.1
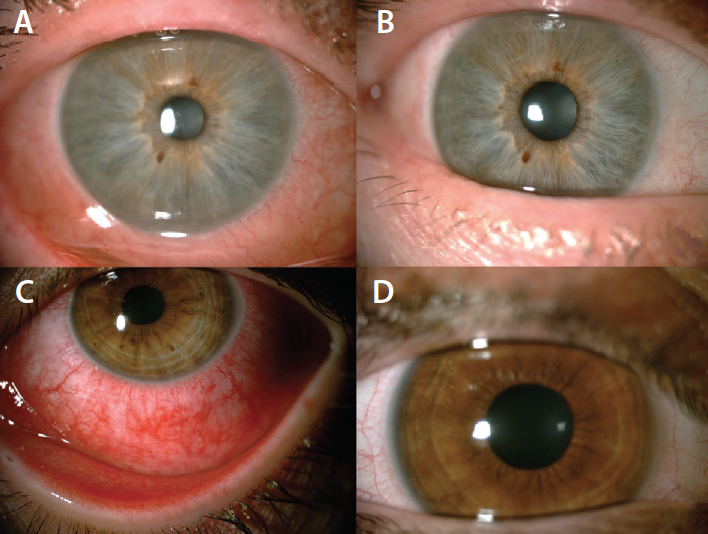
Figure 1. Anterior segment images of a patient who received topical unpreserved artificial tears at the beginning (A) and after the first week (B) of treatment. Anterior segment images of a patient receiving topical 2% cyclosporine A at the beginning (C) and after the first week (D) of treatment. Improvement in signs including conjunctival hyperemia, inflammation, and watery discharge appear to be more prominent after 1 week of treatment with topical cyclosporine A.
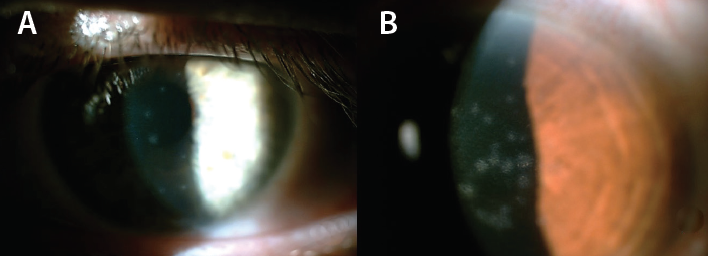
Figure 2. Anterior segment images of two patients who developed corneal subepithelial infiltrates (A, B).
WHAT TO DO?
The aim of treatment in AAK is to alleviate symptoms, but there is no single effective treatment. It is of utmost importance to try to prevent the spread of the infection, so clinicians must inform patients about precautions that can be taken to limit disease transmission. Unfortunately, virustatic agents such as trifluridine, vidarabine, ganciclovir, and cidofovir have been found to be only mildly effective or entirely ineffective against adenoviral diseases.2,3 Povidone-iodine, which is a non–causally directed treatment option for AAK, has a virucidal effect on adenoviruses. Its use can reduce the viral load and decrease the severity of symptoms.4 However, there are no controlled trials supporting the use of this nonspecific treatment option.
The AAO Preferred Practice Pattern for conjunctivitis proposes symptomatic treatment in the form of artificial tears, topical antihistamines, or cold compresses to mitigate symptoms.5 The document states that there is inadequate evidence to support the use of available antiviral agents for treating adenoviral conjunctivitis. The use of topical steroids is recommended only to reduce scarring in severe cases of adenoviral keratoconjunctivitis with marked chemosis, lid swelling, epithelial sloughing, or membranous conjunctivitis.
Most clinicians withhold topical steroids unless there are signs of severe conjunctival inflammation or corneal involvement. However, short-term topical steroids are still used with the aim of decreasing patient discomfort. In a randomized, placebo-controlled study of topical steroids in the treatment of presumed viral conjunctivitis, patients who received topical dexamethasone were significantly more likely to report that they felt the drops helped, compared with patients who received vehicle control drops.6
Restasis (cyclosporine ophthalmic emulsion 0.05%; Allergan) was approved by the US FDA in 2002 for the treatment of dry eye disease (DED). The company filed for regulatory approval of the drops in the European Union in 2016, with a potential decision expected in late 2017 or in 2018.7 Before the commercial availability of topical cyclosporine 0.05%, researchers used fortified topical 2% cyclosporine with promising results for prevention of corneal transplant rejection and severe allergic keratoconjunctivitis.8,9
Since the FDA approval, cyclosporine 0.05% has been widely used as an effective tool to combat ocular surface inflammation in keratoconjunctivitis sicca and other inflammatory ocular surface diseases. It was among the first medications to treat the cause of DED by increasing tear production, rather than providing only symptomatic relief. Topical cyclosporine is considered to be safer than topical corticosteroids because it has negligible effects on IOP and cataractogenesis and minimal to no association with delayed wound healing. Other than in treatment of DED patients, cyclosporine 0.05% has been found to be helpful for patients intolerant of contact lenses and in treatment of severe allergic conjunctivitis, including vernal and atopic types, posterior blepharitis, and superior limbic keratoconjunctivitis.10
TOPICAL CYCLOSPORINE FOR AAK
Cyclosporine A is a selective T-cell immunosuppressive agent, with high clinical efficacy and a good safety profile, that may be effective for ocular diseases in which T-lymphocyte–mediated immunity plays a causative role. It has been used in varying concentrations between 0.5% and 2% to minimize the recurrence of corneal subepithelial infiltrates in adenoviral keratoconjunctivitis.11-13 In a study evaluating the effect of 1% cyclosporine eye drops in the acute phase of infection (four times daily for 21 days), accelerated subjective improvement of local symptoms was observed with topical cyclosporine, in comparison with topical cidofovir or a combination of cidofovir and cyclosporine.2
Because there is still need for a safe and effective treatment to reduce patient discomfort and prevent development of vision-altering complications such as corneal subepithelial infiltrates in AAK, we conducted a study to assess the use of topical 2% cyclosporine A. Patients received topical 2% cyclosporine four times daily for 21 days in the acute phase of AAK, starting right upon diagnosis. Other patients in the study received topical corticosteroids four times daily for 1 week or only symptomatic treatment, including cold compresses and unpreserved artificial tears. In this setting, we had the opportunity to compare the effects of these treatment options in terms of patient comfort and frequency of corneal complications.14
QUESTIONS AND ANSWERS
Our study addressed three questions about topical 2% cyclosporine A in AAK.
Question No. 1: Does it alleviate symptoms in the acute phase of adenoviral keratoconjunctivitis?
SYMPTOMS
All patients in the study received topical unpreserved artificial tears; a fraction of patients also received simultaneous topical corticosteroids, and another fraction also received topical 2% cyclosporine A. We observed that symptoms were less severe and had a shorter duration with concomitant use of cyclosporine A compared with artificial tears alone. Patients who received topical corticosteroids experienced subjective symptoms and objective signs, including conjunctival injection, stinging, burning, and grittiness, for a slightly shorter time than those receiving topical cyclosporine A and those receiving only artificial tears. The rapid alleviation of symptoms caused some patients who received topical corticosteroids to stop the medication prematurely. Using topical cyclosporine A four times daily for 21 days from the onset of the symptoms was associated with high patient comfort with the advantage of a good safety profile.
Question No. 2: Does it prevent the development of corneal subepithelial infiltrates?
DEVELOPMENT OF INFILTRATES
In our experience, the incidence of subepithelial infiltrates was highest in patients who received only artificial tears. The percentage of patients with corneal subepithelial infiltrates was significantly lower with topical cyclosporine treatment compared with topical lubricants alone. As a result, it appears that topical cyclosporine has an inhibitory effect on the development of subepithelial infiltrates when used in the acute phase of the infection.
Question No. 3: Does it increase the duration of viral shedding and cause increased risk of disease spreading?
First, the use of topical 2% cyclosporine A in the early phase of adenoviral keratoconjunctivitis decreases the severity and duration of symptoms (Figure 1). Second, it also appears to have an inhibitory effect on the development of corneal subepithelial infiltrates. Third, it has the advantage of being a steroid-sparing agent.
DURATION OF VIRAL SHEDDING
There are two potential side effects of topical corticosteroids when used in the treatment of viral conjunctivitis: (1) the promotion of viral replication and (2) the exacerbation of an unrecognized herpes simplex virus infection. Ideally, the diagnosis of adenoviral keratoconjunctivitis should be confirmed with commercially available rapid antigen tests or polymerase chain reaction when possible. In this way, other possible causes of viral conjunctivitis can be ruled out. It is known that topical steroids increase and prolong viral shedding in adenoviral conjunctivitis, even when used for only a few days. The same effect has been shown with topical cyclosporine.15 Therefore, patients presenting with AAK should be informed about the highly contagious nature of the disease and the necessary precautions that should be taken to minimize the spread of the disease in an attempt to prevent epidemic outbreaks.



