


In Europe, age-related macular degeneration (AMD) and diabetic eye disease are two of the main causes of blindness and severe visual loss. AMD alone is estimated to affect more than 34 million people in the European Union.1 Pharmacologic treatment of retinal disorders is currently based on drug injections performed either subretinally or intravitreally. In recent years, intravitreal (IVT) injections have drawn more interest than subretinal injections because of the difficulty of subretinal injection technique and the associated risks such as retinal detachment. IVT injection is considered to be a more accessible technique, but these injections are not without risk.
In 2009, several organizations representing ophthalmologists in Belgium came together to form the Workgroup of Belgian Extramural Eye Surgery. This workgroup was tasked with developing criteria for dedicated rooms for performing IVT injections. Once the criteria were developed, ophthalmologists in Belgium were asked to follow them.
This article outlines the product of the workgroup’s efforts: a list of practical conditions under which IVT injection is feasible in an extramural ambulatory setting (ie, outside the hospital) in Belgium, while limiting the risks of this mode of administration, such as infection or damage to intraocular structures. In this article, we present considerations for structural organization of the extramural setting, the organization of staff, guidelines for performing IVT injections, and the responsibilities of staff members during the process of IVT injections.
STRUCTURAL ORGANIZATION
Following are the considerations for the structural organization of the IVT room, as devised by the Workgroup of Belgian Extramural Eye Surgery.
Outside the IVT room. It is important to take several considerations into account concerning infrastructure outside the IVT room (Figure). First, a waiting room for patients and their relatives with wheelchair-accessible toilets and bathrooms is needed. Second, it is convenient to have a storage room for spare materials that is separate from the waiting and procedure rooms and close to the IVT room.
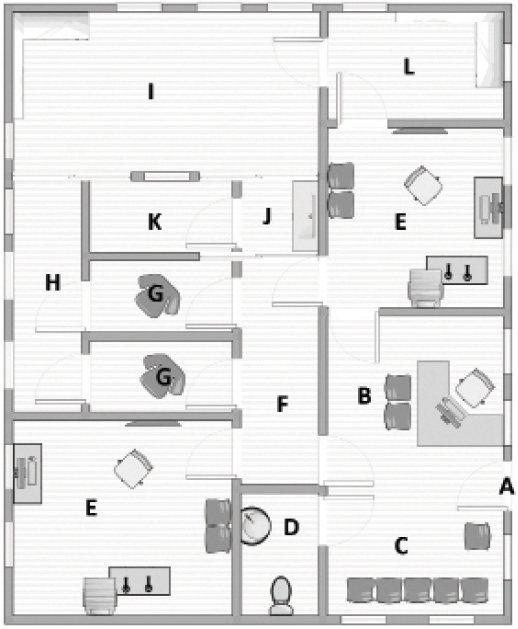
Figure. Schematic design illustrates how to incorporate many of the features of the extramural IVT center described in this article: (A) entrance; (B) administrative staff desk; (C) waiting room; (D) bathroom; (E) doctor’s office; (F) corridor to changing room (for patients) and IVT room (medical staff only); (G) changing room (for patients); (H) corridor to IVT room; (I) IVT room; (J) sink; (K) changing room (for ophthalmologists and assistants); (L) storage space.
Because the IVT room is situated in an extramural setting, it is compulsory to have a type B autoclave in a separate room, but again close to the IVT room. Alternatively, the ophthalmologist can use disposable instruments.
Finally, it is advisable to have a recovery space where patients can rest after the procedure if necessary.
Inside the IVT room. IVT procedures can be performed in any room that complies with the following requirements in infrastructure, sterility, and resuscitation facilities guidelines.
Regarding infrastructure, to secure standard hand hygiene measures, the medical staff must have access to a washbasin. It can be situated either in the IVT room itself or in a separate dedicated room as in OR designs. The washbasin should contain antiseptic soap and disinfecting products for scrubbing and sterilizing the hands.
To supply the medical staff with sufficient materials during the day, a storage closet within the IVT room should be restocked daily with material from the aforementioned storage space outside the IVT room. These materials would include sterile drapes, sterile compresses, and trays, among other items.
A refrigerator with DIN 58345 certification should be available for storage of drugs that cannot be kept at room temperature. The temperature regulation of this unit must be checked daily.
To ensure the accessibility of the IVT room to all patients, it is recommended to avoid stairs in the medical practice; if stairs are present, solutions should be provided for patients who are unable to access them. For safety reasons, there must be a fire detection unit in every room, and firefighting equipment must be present. A modern heating system is recommended to provide an optimal environment. Furthermore, an air purification system complying with ISO Class 8 generates the best air circulation.
Regarding sterility, easily cleanable surfaces are required in the IVT room. Documentation of the cleaning must be done chronologically by date and time. Autoclaves should have a weekly checkup and a technical support visit at least every 2 years. It is important to keep a record of sterilization of the instruments, and this record should be kept for at least 1 year and be indexed both by patient and by date. Additionally, sterility detection methods, such as autoclave tape, should be used to indicate the sterility of instrument boxes.
Regarding resuscitation facilities, the extramural center should follow guidelines outlined in the Royal College of Ophthalmologists’ Ophthalmic Service Guidance: Intravitreal Injection Therapy,2 combined with some additional designated medications and facilities. The following materials are vital and therefore required in an extramural center: oxygen; oxygen mask with reservoir and connection tubing; intubation material; automated external defibrillator with monitoring for electrocardiography, oxygen saturation, and noninvasive blood pressure; syringes and needles; IV catheters; infusion cannulas with maintenance solutions; oropharyngeal airway (eg, Mayo tube); and biting block and aspiration material (a pump, a Yankauer suction tip, and an aspiration tube).
Obligatory resuscitation medications include adrenaline, amiodarone, atropine, ephedrine, diazepam, methylprednisolone succinate, promethazine, rocuronium, and succinylcholine.
ADMINISTRATION
Administrative considerations, as outlined by the Workgroup of Belgian Extramural Eye Surgery, are as follows.
Emergency numbers. In the IVT room, a telephone must be present with clearly visible emergency numbers. On the letter of discharge, the extramural center’s phone number must be visible in order to ensure that patients can find the number to call when complications are experienced. Additionally, the discharge letter should contain information on any prescribed drug (eg, eye drops), including instructions on using the medication and the prescribed dose regimen.
Database. A database must record at least a minimum number of data: the type of intervention, the method of intervention, any pre-, peri-, and postoperative medications, the name and address of the patient, and the name of the patient’s general practitioner.
Quality assurance. There must be documentation of several procedures, including training for emergency situations and training for the evacuation plan, and a visit from technical support for the environmental heating system must be scheduled every 2 years for gas and yearly for liquid fuel checkups. According to Belgian law, there must be a revision of the electrical installation every 5 years, and this must be recorded. There should also be a record of every medical accident that takes place.
National authorities. Every country in the European Union has different regulations. In order to be compliant with national regulations, it is important to continually verify and implement the regulatory requirements that apply to your medical practice. Because we operate in Belgium, we will briefly discuss the regulatory situation in Belgium, but this will vary in other countries. The Belgian authorities require the registration and documentation of the orders for and turnover of drugs and other medical devices (eg, purchase, current stock, usage, expiration date monitoring). When an order of medication is received, verification of the expiration date is done by randomly sampling 10 products from the order. The temperature of the refrigerator must be documented continuously, for example through a temperature probe in the device.
When audit visits of the extramural center are performed, invoices of purchases and patient files must be shown to the auditors. In Belgium, the dedicated IVT room must inspected by an organization accredited by the Belgian Accreditation Structure in order to be recognized as a extramural center. The list of recognized extramural centers is updated every 4 months by the workforce; accreditation is vital to acquire civil liability in case problems arise.
IVT INJECTION GUIDELINES
To ensure the quality of an IVT injection, the experience of the ophthalmologist with IVT is of great importance. Generally, it is recommended to inject only one eye of a patient per day and avoid injecting both eyes simultaneously. In exceptional cases, both eyes can be injected simultaneously with the use of new disposables and injectables with different batch numbers and attributes, syringes, and needles for each eye.
Preparation of the injectable fluid. It is essential that any substance injected into the eye be sterile. The occurrence of contamination during the procedure is hazardous. Preparation and injection of the injectable substance must therefore be done in sterile conditions. The container should be intact, and this must always be checked prior to use. If the seal is broken, the sterility of the injectable cannot be guaranteed, and it must be discarded immediately.
Preparation of the operator. IVT injections should always be performed using standard operation procedures regarding sterility of the operator, handling of the IVT materials, and injection protocols. These protocols are essential in obtaining and maintaining consistent quality.
Hand hygiene and protection. The operator’s hands should undergo surgical disinfection, and sterile gloves should be worn. Sterile gloves must be removed after each injection. It is recommended that masks be worn, as the operator’s face is proximal to the operating field, although this is not mandatory.3
Post-IVT monitoring. The operator and staff must be familiar with recognizing and managing complications and adverse events.
ORGANIZATION OF STAFF RESPONSIBILITIES
Guidelines for staff responsibilities include staff in the IVT room and informed consent.
Staff in IVT room. The operational staff that is required in the IVT room consists of an ophthalmologist and a medical assistant. The medical assistant works under the responsibility of the ophthalmologist and must have professional liability insurance that can be verified.
Informed consent. A signed informed consent from the patient is obligatory before any off-label use of a medication, and it is the duty of the ophthalmologist to inform every patient correctly. Explanation can be done orally, but is much better retained by the patient when it is combined with visual information, such as informative flyers, pictures, and videos.
CONCLUSION
Following the increased popularity in the use of IVT injections for the management of retinal disorders including AMD and diabetic eye disease, there has been a surge in the number of extramural ambulatory surgery settings that are performing this procedure. By following these guidelines for performing IVT and the considerations for the structural organization of the extramural setting, ophthalmologists can be ensured that they are providing safe, convenient services for their patients.
1. Li JQ, Welchowski T, Schmid M, et al. Retinal diseases in Europe: prevalence, incidence and healthcare needs. http://www.euretina.org/downloads/EURETINA_Retinal_Diseases.pdf. Accessed August 9, 2018.
2. Ophthalmic Service: Guidance Intravitreal injection therapy. Royal College of Ophthalmologists 2018. https://www.rcophth.ac.uk/wp-content/uploads/2018/02/Intravitreal-Injection-Therapy.pdf. Accessed August 9, 2018.
3. Wen JC, McConnel CA, Mochon A, Garner OB. Bacterial dispersal associated with speech in the setting of intravitreous injections. Arch Ophthalmol. 2011 Dec 1;129(12):1551-1554.


