CASE PRESENTATION
A 67-year-old Ethiopian woman presents with a complaint of a 1-month decrease in vision in her right eye. She suspects the problem is the result of a recent fall.
The patient underwent cataract surgery several years ago. Examination shows inferior corneal edema and a dislocated three-piece IOL, with optic capture inferiorly and the inferior haptic in the anterior chamber (Figure). Her visual acuity is 20/400, and there is no improvement with refraction. OCT imaging of the retina is normal.
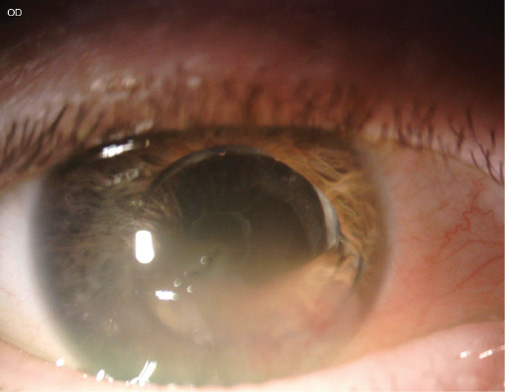
Figure. Corneal edema inferiorly and optic capture of a three-piece IOL, the inferior haptic of which is in the anterior chamber.
How would you approach this case? Would you attempt to reposition the IOL with or without iris suturing? Would you exchange the IOL? Would you consider combining intervention with Descemet membrane endothelial keratoplasty (DMEK) or Descemet-stripping endothelial keratoplasty?
—Case prepared by Audrey R. Talley Rostov, MD

JORGE L. ALIÓ, MD, PhD, FEBOPHTH
In all likelihood, complications occurred during this patient’s cataract procedure, and her current presentation has no connection with the trauma that she described. The original surgeon probably mistakenly placed the lens haptic in the anterior chamber, as the presence of iridocapsular synechiae suggests. The presence of the haptic in the anterior chamber has led to chronic corneal decompensation, and the condition probably precipitated recently.
The patient requires a corneal transplantation procedure, preferably DMEK. First, however, I would try to resolve the problem of the IOL because this surgery often causes inflammation. In the OR, with the patient under local retrobulbar anesthesia, I would de-epithelize the cornea and administer topical glycerol 20% to clear it for about 5 minutes. I would dissect the iridocapsular synechiae to study how extensive they are in order to replace the inferior IOL haptic in the sulcus. This maneuver can be traumatic and can lead to inadequate haptic support in the sulcus, in which case a 9-0 polypropylene suture, located inferiorly behind the iris, would be needed to provide support. I would constrict the pupil and execute tractional centripetal maneuvers at the inferior iris as needed with Alió MICS forceps (MicroSurgical Technology) to fully detach the iris from the synechiae and create a round pupil. At the conclusion of the case, I would instill 0.1 mL of triamcinolone in the anterior chamber and 0.9 mL in the orbital floor to prevent anterior segment inflammation.
Once the eye is quiet, the pupil is round, and the outcome was successful, I would proceed with DMEK. I have found that, in some cases, once the anatomy has been restored, corneal transparency returns, and DMEK is no longer necessary. More often, however, DMEK is finally required.

BERNARDO DE PADUA SOARES BEZERRA, MD
This is most likely a case of posttraumatic subluxation of a three-piece IOL. The timing of surgery is important here. I would look for anterior segment inflammation and uveitis-glaucoma-hyphema syndrome. Media opacity from focal corneal edema must also be considered. Intervention should happen as soon as intraocular inflammation is under control, given the ever-growing risk of corneal decompensation and reduced visibility during surgery.
Less would be more in this case. In an initial procedure, I would only reposition or replace the IOL. I would prefer a superior approach to avoid constructing the wound in the area of corneal edema. I would fill the anterior and posterior chambers with OVD using a soft-shell technique to prevent further endothelial cell loss. I would viscodissect the IOL haptics from the iris and reposition the three-piece IOL in the sulcus by rotating and directing the haptics clockwise to the supraciliary space. The Figure shows sufficient capsular bag support for sulcus placement. For IOL replacement, power calculations should consider standard keratometry values with a slightly myopic target and sulcus implantation.
The two other problems can be managed secondarily if they become clinically significant, with endothelial keratoplasty after appropriate medical management of the corneal decompensation. I would prefer DMEK, although it could be technically challenging in a posttraumatic eye. If, after the first procedure the patient had significant glare or light sensitivity that was determined to be due to a focal iris defect or large corectopic pupil, I would perform a pupilloplasty or iris implant.

RUSSELL SWAN, MD
Given that neither the optic nor the inferior haptic is in the capsular bag complex, this three-piece IOL was likely previously placed in the sulcus. An open posterior capsule and associated corneal decompensation increase the complexity of the case.
I would perform a retrobulbar block and start by marking the sclerotomy sites for the Yamane technique. I would place a 25-gauge trocar via an inferior temporal incision through the pars plana and then perform a limited anterior vitrectomy with anterior infusion to ensure the absence of vitreous. Through a temporal 2.75-mm incision and paracentesis, I would secure the IOL with MST microforceps and cut the IOL with MST IOL scissors (both instruments from MicroSurgical Technology) three-quarters of the way across the optic. I would externalize the IOL through the main incision. Next, I would use the Yamane technique for scleral fixation of a three-piece CT Lucia IOL of the appropriate power based on the previous IOL and residual refractive error prior to corneal decompensation.
Given the corneal decompensation, I would discuss with the patient preoperatively the option of a staged procedure, but my recommendation would be to combine IOL exchange with ultrathin Descemet-stripping automated endothelial keratoplasty (DSAEK) because of the vitrectomy. At the conclusion of the IOL procedure, I would perform standard DSAEK using the Endoserter (CorneaGen) for controlled delivery of the graft into the anterior chamber and place an air bubble at the conclusion of the case. This approach has the greatest chance of long-term success with the fewest interventions for this woman.

WHAT I DID: AUDREY R. TALLEY ROSTOV, MD
I proceeded with the case using a peribulbar block. I placed one paracentesis incision temporally and additional ones at the 12 and 6 clock positions. I performed an injection of Healon GV (Johnson & Johnson Vision) and explored the position of the IOL. I separated the synechiae with viscodissection and a cyclodialysis spatula as well as with microforceps and horizontal scissors.
No vitreous was present in the anterior chamber. I repositioned the three-piece IOL and rotated it 90º into the sulcus. I irrigated out the OVD. I was prepared to perform an IOL exchange or to suture the haptic to the iris with a polypropylene suture on a CIF-4 needle, but the IOL appeared to be stable after rotation. I instilled carbachol intraocular solution and hydrated the paracentesis incisions.
Since the surgical intervention, I have been observing the patient. Thus far she has not required further IOL manipulation or DMEK surgery, but I am prepared to perform these procedures in the future if required.



