The Evolution of Ophthalmic Drug Delivery Systems
Rohan Bir Singh, MD
Leiden, Netherlands
Victoria Ly, MD, MPH
Texas, United States


Ophthalmology patients have long endured the treatment burden of multidrop regimens and frequent clinic visits as a result of a significant unmet need for effective, sustained drug delivery to the eye. During the past decade, however, there has been great interest in the development of systems that efficaciously deliver drugs with comfort and ease. Our response in this article and the accompanying Figure provide an overview of current methods and innovative drug delivery systems that are emerging.
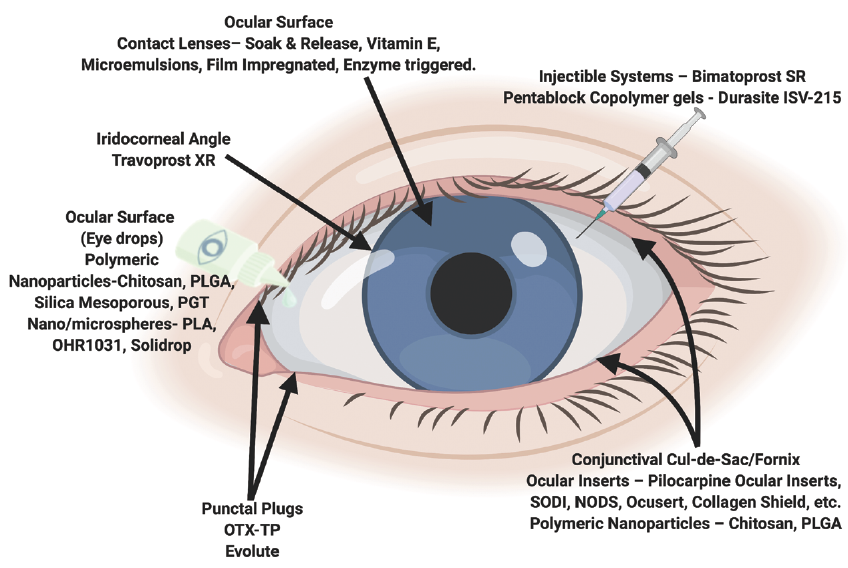
Figure. Depiction of current methods of drug delivery and emerging drug delivery systems.
Prepared by Dr. Singh using Biorender.com.
EYE DROPS
Topical eye drops are generally the preferred route of drug administration because they are noninvasive and can be administered by the patient. Unfortunately, the ocular bioavailability of medication is severely limited by tear dilution and turnover, rapid blinking reflex, and poor corneal penetration.
Polymeric nanoparticles such as poly(lactic-co-glycolic acid), silica mesoporous, and propoxylated glyceryl triacylate overcome these barriers and enhance drug delivery to the anterior chamber by increasing transcorneal permeability and protecting drugs from degradation. Lengthening residence time on the corneal surface is another approach, and it is accomplished by nanospheres and microspheres because of their unique structures and by chitosan because of its mucoadhesive properties. Alternatively, pentablock copolymer gels enable drugs to accumulate in the lower fornix because of changes in the viscosity of the gel that are triggered by body heat on contact.
These approaches decrease dosing frequency and impart sustained drug release. Most of these delivery systems, except for OHR1031 (Ohr Pharmaceutical) and Durasite ISV-215 (Insite Vision), however, have an undesirable initial burst effect.
DRUG-ELUTING CONTACT LENSES
Therapeutic contact lenses are widely used in refractive surgery and the management of corneal injuries and disease. The development of drug-eluting contact lenses has garnered significant interest from ophthalmic researchers. Advantages of this technology are that the lenses are noninvasive, easy to insert by the user, and capable of extended wear. They also provide much higher drug bioavailability compared to eye drops.
Drugs can be loaded onto the lenses using soak and release, vitamin E, microemulsions, and film impregnation. Enzymes can also trigger drug release from the lenses. Despite their potential for sustained drug delivery, the application of drug-eluting contact lenses is limited by reduced oxygen permeability, protein adherence, altered mechanical properties, and lens-related infections associated with continuous wear.
INTRACAMERAL INJECTABLES
In March 2020, the FDA approved the use of the first intracameral bimatoprost implant (Durysta, Allergan). It is administered as an outpatient clinic procedure. The implant was developed to provide sustained-release therapy for glaucoma over the course of 4 to 6 months. Its design allows therapeutic concentrations within the anterior chamber to be reached with less drug required, which reduces the risk of corneal and systemic side effects. Durysta facilitates targeted drug delivery, particularly for prostaglandin analogues that act primarily at the iris–ciliary body in the intracameral space.
Other implants using travoprost are in phase 2 clinical trials. These adverse effects have not been reported with implants. The intracameral injection of drugs carries risks, however, including toxic anterior segment syndrome and toxic endothelial cell destruction syndrome.
PUNCTAL PLUGS
Similar to drug-eluting contact lenses, punctal plugs have been redeveloped for sustained drug release. OTX-TP (Ocular Therapeutix) and Evolute (Mati Therapeutics) deliver travoprost and latanoprost, respectively, for 3 months via drug-eluting punctal plugs. The primary endpoint in the phase 3 study of OTX-TP was not met. A significant reduction in IOP, however, was seen at eight of the nine time points.
Punctal plugs can be advantageous for drug delivery because they can be easily inserted in outpatient clinics and worn comfortably by patients. Patients and ophthalmologists, however, may be deterred from using these devices because of foreign body sensation or patients’ inability to confirm that the punctal plugs are still in place.
CONCLUSION
Recent developments in drug delivery are promising. As these systems progress through clinical trials, the release of more extensive clinical data is highly anticipated.
Challenges in Glaucoma Therapy
Sahil Thakur, MBBS, MS
Singapore

The standard routes of eye drops, ointments, and intravitreal injections are the backbone of ocular drug delivery in India. Global developments such as nanoparticles, drug-eluting contact lenses, and ocular implants are slowly being introduced. Their benefits, however, remain out of reach of the developing world.1 My contribution to this article focuses on glaucoma management and the challenges of drug delivery and ensuring adherence in glaucoma patients.
BARRIERS
The cost of generic prostaglandin therapy is around 300 rupees ($4) per month, and the average monthly income in India is approximately 32,000 rupees ($437). Durysta, on the other hand, retails for approximately 146,500 rupees ($2,000). This exercise in finance makes it easy to understand why more cost-effective solutions are required for countries without robust medical insurance programs.
Technological innovation in the delivery of safe and targeted drugs is vital. I feel it is more important, however, to ensure patient adherence and persistence with prescribed medical therapy. In my experience, most patients simply stop administering eye drops after a few months of use because of the insidious nature of glaucoma and the lack of perceived immediate benefits. A multipronged approach is required to address these barriers.
No. 1: Handle expectations properly. Full disclosure of risks and benefits should be made when initiating treatment.
No. 2: Targeted communication during consultation. Specifics about the drops, including what they do and why they are important, should be provided, side effects should be managed early, and patients’ overall financial situations should be considered when prescribing medications.2
No. 3: Schedule reminders via telehealth modalities. Patients should be encouraged to use smartphone applications for drug reminders and eye drop alarms and to keep glaucoma diaries to facilitate monitoring.3,4
No. 4: Measures to ensure compliance . Yearly audits of patients’ eye drop usage should be conducted. It would be helpful if metrics and innovations were developed to measure compliance and accurate dosage, such as might be possible with eye drop bottle sensors.5
CONSIDERATIONS
It is important to understand patient expectations about medical therapy to effectively design and market drug delivery devices and technologies. The end user’s interests are often compromised in the pursuit of technical perfection. It is thus imperative to consider factors such as affordability, availability, accessibility, adequacy, and acceptability for new models of drug delivery. Until such consensus models are developed, eye drops will remain the vehicle of choice for delivering drugs to the eye.
1. Singh RB, Ichhpujani P, Thakur S, Jindal S. Promising therapeutic drug delivery systems for glaucoma: a comprehensive review. Ther Adv Ophthalmol. 2020;12:2515841420905740.
2. Ichhpujani P, Kalra G, Singla E, Kumar S. Breaking bad news about glaucoma: a SPIKES strategy primer. Int Ophthalmol. July 28, 2021. doi:10.1007/s10792-021-01956-1
3. McDonald L, Glen FC, Taylor DJ, Crabb DP. Self-monitoring symptoms in glaucoma: a feasibility study of a web-based diary tool. J Ophthalmol. 2017;2017:8452840.
4. Ichhpujani P, Thakur S. Apps and Social Networking Pages for Glaucoma. Smart Resources in Ophthalmology: Applications and Social Networking. Springer Singapore; 2018:71-96.
5. Nishimura K, Tabuchi H, Nakakura S, et al. Evaluation of automatic monitoring of instillation adherence using eye dropper bottle sensor and deep learning in patients with glaucoma. Transl Vis Sci Technol. 2019;8(3):55.