
I often encounter patients whose pupillary dilation is limited or suboptimal (Figure 1). A poorly dilating pupil does not provide a sharp red reflex or adequate visibility for either the creation of a sufficiently large capsulorhexis or phacoemulsification. This article, the accompanying sidebar (Key Points), and Figures 1 to 6 provide advice on how to proceed in these cases.
Key Points
- When intraoperative floppy iris syndrome is encountered, reduce fluidics settings to the minimum to prevent iris prolapse.
- Dilate the pupil if you suspect its size will compromise your ability to perform all of the steps of the cataract procedure.
- Attempt to release posterior synechiae and pupillary membranes and administer intracameral mydriatics before switching to a mechanical stretching strategy.
- If using iris hooks, avoid stretching the pupil beyond 5.0 mm because damage to the iris sphincter and iris root ischemia may occur.
- Choose the surgical technique and dilatation strategy that work best in your hands.
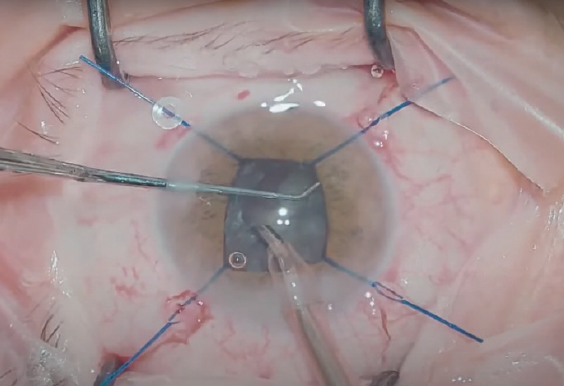
Figure 1. At the beginning of surgery, pupillary dilation was inadequate in this eye.
RISK FACTORS FOR INTRAOPERATIVE MIOSIS
A major part of successfully executing cataract surgery is being cognizant of seven risk factors for intraoperative miosis:
- No. 1. Pseudoexfoliation syndrome, iris sphincter atrophy, tissue hypoxia,1 and a compromised zonular apparatus;
- No. 2. Uveitis, a chronic condition characterized by posterior synechiae and iris atrophy;
- No. 3. Diabetes, which can lead to a weak response to mydriatic agents2 and intraoperative constriction of the pupil;
- No. 4. Prolonged surgical time;
- No. 5. Therapy with systemic alpha-adrenergic blockers such as tamsulosin (it is important to note that not only men are treated with alpha-adrenergic blockers; this drug class is also often used to treat women for high blood pressure and urinary retention3);
- No. 6. Topical therapy with pilocarpine and carbachol, which can lead to the formation of posterior synechiae and sphincter hypertrophy; and
- No. 7. The use of intravenous anesthetics such as fentanyl.
PHARMACOLOGIC STRATEGIES
Pharmacologic and mechanical approaches may be employed to manage a small pupil intraoperatively. The former includes the off-label administration of either epinephrine 0.5 mL/0.1% in 500 mL balanced salt solution or phenylephrine 1.5% and lidocaine 1.0% intracamerally in the irrigating solution.4 An alternative is to perform an off-label intracameral direct injection of either preservative-free epinephrine 1:4,000 diluted in balanced salt solution or a lidocaine/epinephrine preparation.5
In Bulgaria, where I practice, epinephrine is added to the irrigating solution in all cases. This approach maintains an adequately sized pupil during surgery. When intraoperative floppy iris syndrome (IFIS) develops unexpectedly during cataract surgery (the size of the pupil is sufficient at the outset of surgery but later constricts), oftentimes it is because the patient failed to recall prior short-term therapy with a systemic alpha-adrenergic blocker.
REQUIREMENTS
Pupillary opening. The continuous curvilinear capsulorhexis (CCC) is a key factor in successful cataract surgery. This step demands an adequately sized pupillary opening of approximately 4.5 to 5.5 mm. Some surgeons can effectively guide the CCC even under the iris with forceps or a needle and achieve an adequately sized CCC. Alternatively, automated devices such as a femtosecond laser, CapsuLaser (Excel-Lens), or Zepto (Centricity Vision) may be used to perform the capsulotomy. Adequate pupillary dilation is required even with automated devices.6
The appropriate pupillary size depends mainly on the surgeon’s experience and the technique used to extract the nucleus. In general, divide and conquer requires a pupillary size of at least 4.5 mm, whereas vertical and horizontal chop techniques (Figure 5) require a pupillary size of 3.5 to 4.0 mm for safe nuclear removal.
An adequately sized pupil is also important during irrigation and aspiration, and the size required depends on the I/A technique used. Coaxial irrigation and aspiration require approximately a 4.5-mm pupil, whereas bimanual irrigation and aspiration require a pupil larger than 3.5 mm. This is because the ability to reach under the iris and anterior capsular margin is greater.
Fluidics. This also plays an important role, especially in cases of IFIS. Low fluidics settings are preferable because iris tissue tends to follow fluid waves. Proper wound architecture and a good match between the sleeve and incision are therefore advisable, as is minimizing maneuvers of the phaco tip and feeding pieces into the phaco tip with the chopper. My preferred settings are as follows: a bottle height of 80 cm, a flow rate of 14 mL/min, a vacuum level of 280 mm Hg, and a dynamic rise of 0. Further, I use torsional ultrasound and Intelligent Phaco software (Alcon) depending on the density of the nucleus, and I rely on mechanical chopping more than on the holding properties of the phaco tip.
IOL selection. Another key consideration is lens choice. I use an IOL that has a slowly unfolding hydrophobic C-loop design because it is easy to insert through a small pupil and to position in the bag.
MECHANICAL STRATEGIES
If the pupil size is not sufficient despite the pharmacologic strategies and maneuvers discussed earlier, the next options are mechanical stretching and retaining maneuvers.
Devices. My preferred approach is to place iris hooks because they can act as a stretching device, iris tissue retainer, and capsular support when needed (in case of pseudoexfoliative capsular-zonular instability). I typically place four iris hooks, one in each quadrant, and stretch the pupillary diameter to no more than 5.0 mm to avoid an irregular postoperative shape and to prevent iris root ischemia from overstretching (Figure 2).7,8 The paracenteses for the hooks should be created slightly more limbally and parallel to the iris so that the iris does not lift as the pupil is stretched to the desired size. Some surgeons place five iris hooks in a diamond fashion, with the fifth hook positioned under the main corneal incision to prevent the iris from prolapsing during surgery.

Figure 2. Iris retractors were placed to provide a sufficient and safe pupillary size.
Other pupillary expansion devices include a Malyugin Ring (MicroSurgical Technology), made of polypropylene; Canabrava Ring (AJL Ophthalmic), made of PMMA; and an I-Ring Pupil Expander (Beaver-Visitec International), made of polyurethane. All of these devices achieve round, smooth expansion of the pupil to between 6.0 and 7.0 mm, which allows cataract surgery to be performed safely.
Manual maneuvers. Manual stretching maneuvers can be tried but generally with limited success. A manual approach is also more likely than a device to traumatize the iris, which increases the risk of postoperative cystoid macular edema.9 The injection of a high-density OVD may facilitate certain steps such as the capsulorhexis, but the success and duration of this strategy, again, are limited.
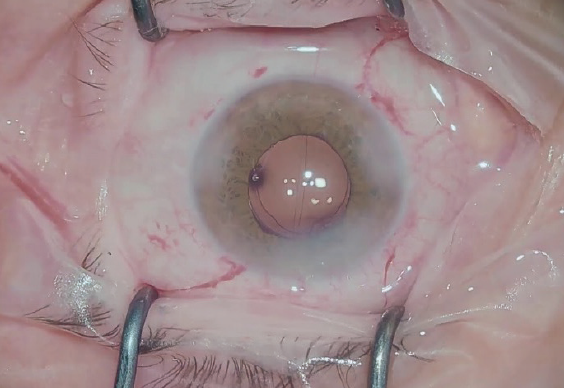
Figure 3. At the end of uneventful surgery, the pupil was round, regular, and well centered.
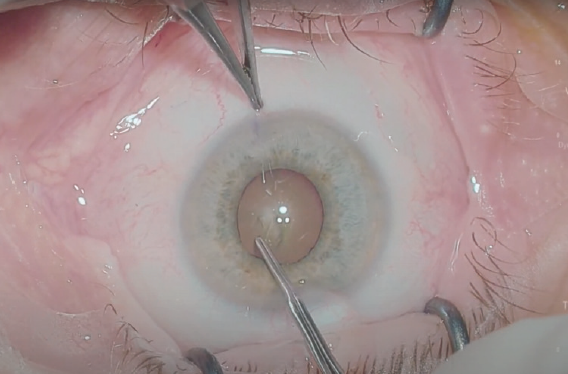
Figure 4. At the beginning of surgery, this pupil, even though it was not fully dilated, was large enough to allow creation of the capsulorhexis.
CONCLUSION
Cataract surgery in the presence of a small pupil (Figure 4) is extremely challenging. Maintaining a balance between surgical trauma and safe and effective cataract extraction is the key to success (Figures 3 and 6). The choice of management strategy depends on the surgeon’s experience, the presence of ocular comorbidities, and the availability of devices.
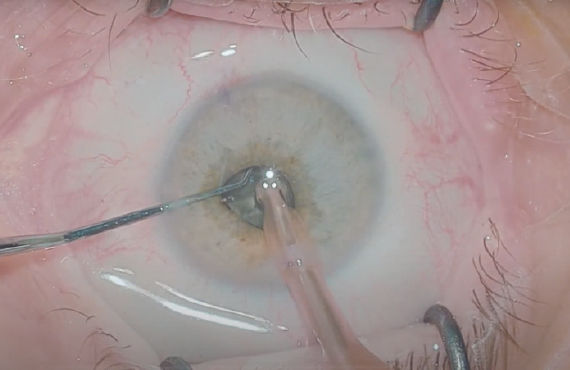
Figure 5. Horizontal chop maneuvers in an eye with a small pupil. In this case, unexpected IFIS occurred during surgery.
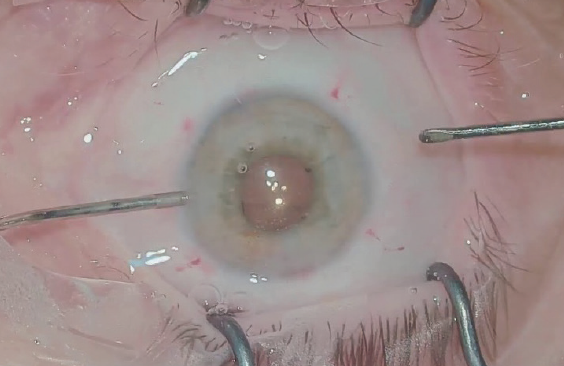
Figure 6. At the end of this uneventful surgery, an IOL was implanted and centered on the round and regular pupil.
1. Asano N, Schlötzer-Schrehardt U, Naumann GO. A histopathologic study of iris changes in pseudoexfoliation syndrome. Ophthalmology. 1995;102(9):1279-1290.
2. Zaczek A, Zetterström C. Cataract surgery and pupil size in patients with diabetes mellitus. Acta Ophthalmol Scand. 1997;75(4):429-432.
3. Chang DF, Campbell JR. Intraoperative floppy iris syndrome associated with tamsulosin. J Cataract Refract Surg. 2005;31(4):664-673.
4. Lundberg B, Behndig A. Intracameral mydriatics in phacoemulsification cataract surgery—a 6-year follow-up. Acta Ophthalmol. 2013;91(3):243-246.
5. Labetoulle M, Findl O, Malecaze F, et al. Intracameral Mydrane Study 2 Group. Evaluation of the efficacy and safety of a standardised intracameral combination of mydriatics and anaesthetics for cataract surgery. Br J Ophthalmol. 2016;100(7):976-985.
6. Nagy ZZ, Takacs AI, Filkorn T, et al. Complications of femtosecond laser–assisted cataract surgery. J Cataract Refract Surg. 2014;40(1):20-28.
7. Masket S. Avoiding complications associated with iris retractor use in small pupil cataract extraction. J Cataract Refract Surg. 1996;22(2):168-171.
8. Yuguchi T, Oshika T, Sawaguchi S, Kaiya T. Pupillary functions after cataract surgery using flexible iris retractor in patients with small pupil. Jpn J Ophthalmol. 1999;43(1):20-24.
9. Flach AJ. The incidence, pathogenesis and treatment of cystoid macular edema following cataract surgery. Trans Am Ophthalmol Soc. 1998;96:557-634.


