CASE PRESENTATION
A 24-year-old woman with advanced keratoconus underwent CXL in her right eye 4 years ago and repeat CXL 2 years ago. The patient wore scleral contact lenses successfully until 1 year ago when she presented with a peripheral stromal infiltrate that developed into multifocal stromal infiltrates under the contact lens in her right eye. Herpes simplex virus (HSV) was suspected, and therapy with valacyclovir 1,000 mg (Valtrex, GSK) twice daily was initiated. The infiltrates eventually formed a ring configuration. Steroid therapy was initiated, and the patient was referred for testing to rule out an Acanthamoeba infection.
The examination (Figure 1) and confocal microscopy (Figure 2) were suspicious for Acanthamoeba, and a culture was obtained. The patient was started on an alternating regimen of polyhexamethylene biguanide 0.02% and chlorhexidine 0.02% every 2 hours while awake. When the steroids were discontinued, the patient’s condition promptly worsened. A corneal biopsy was performed. Gabapentin, ibuprofen, and a narcotic were required to provide adequate control of the severe pain she was experiencing. Culture results and the biopsy were positive for Acanthamoeba. Treatment with polyhexamethylene biguanide 0.02% and chlorhexidine 0.02% was continued, and therapy with propamidine isetionate 0.1% (Brolene, Thornton & Ross) was initiated.
The patient recalled showering in Mexico while her contact lenses were in place 2 weeks before the onset of inflammation. The infection improved with treatment, but she began to develop prominent neovascularization and scarring (Figures 3 and 4).
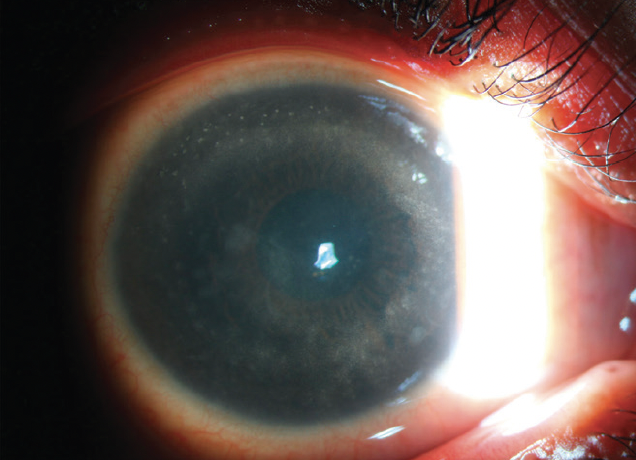
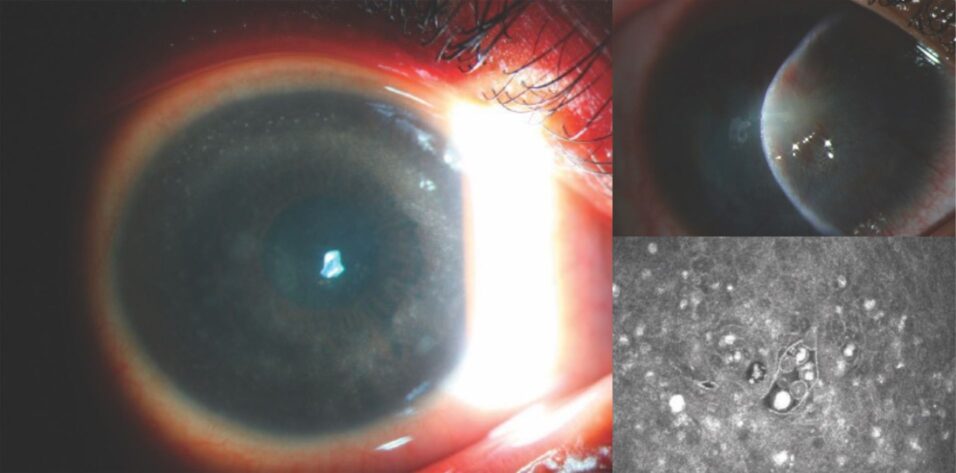
Figure 1. Scleral scatter illumination shows an infiltrate forming a ring in the stroma.
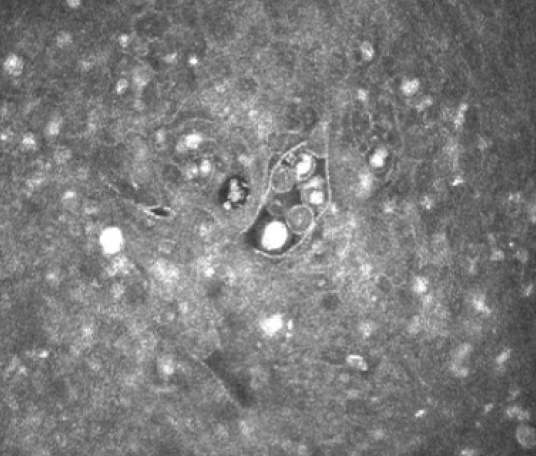
Figure 2. Confocal microscopy supports a diagnosis of Acanthamoeba cysts in the corneal stroma.
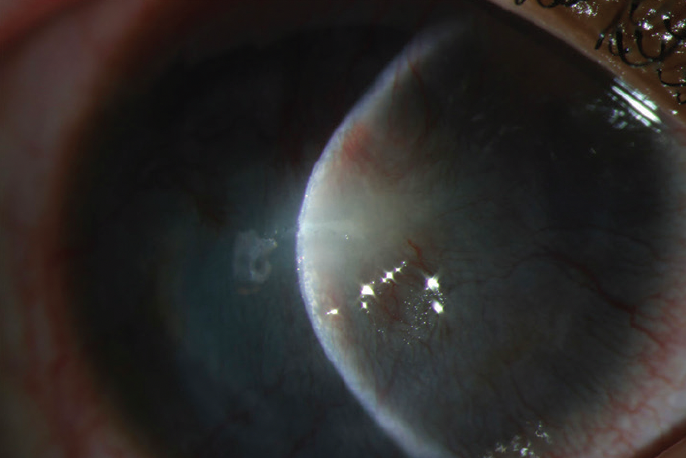
Figure 3. Focal slit-beam illumination shows a shrinking infiltrate in a steep cornea, extensive scarring, and neovascularization.
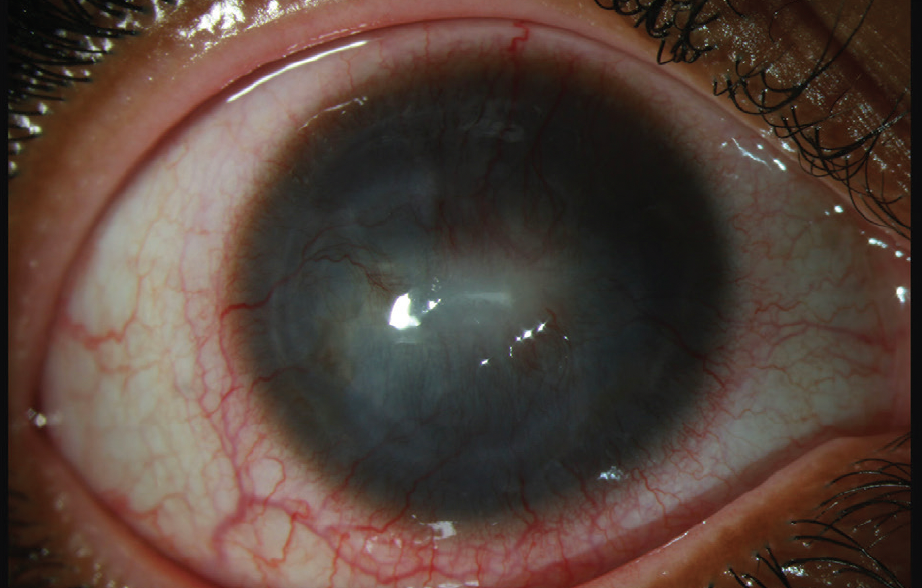
Figure 4. Broad-beam diffuse illumination demonstrates an impressive improvement in the overall redness of the bulbar conjunctiva, now with limbal flush. Central scarring and peripheral stromal neovascularization are prominent.
All figures courtesy of Christiaan Lopez-Miro, ophthalmic photographer, Duke Eye Center
How would you proceed? When do you prescribe steroid treatment to reduce inflammation in the setting of Acanthamoeba? When is a therapeutic keratoplasty indicated for the condition? Is there a role for deep anterior lamellar keratoplasty (DALK) in this situation? Do you think the patient’s history of CXL had an impact on her clinical course?
—Case prepared by Alan N. Carlson, MD

JOHN K.G. DART, MA, DM, FRCOPHTH
Topical steroid treatment for Acanthamoeba keratitis (AK) is controversial. After an appropriate period of treatment with effective antiamoebic therapy (AAT) consisting of biguanide(s) with or without propamidine 0.1%, the addition of a topical steroid often helps control pain and inflammation. (Both biguanide disinfectants and propamidine are used off label for the treatment of AK.) In the current situation, AAT administered every 2 hours for a total of eight times daily was probably a suboptimal initiating dose. The subsequent period of therapy and its frequency are unstated.
If the patient completes more than 3 weeks of an intensive course of AAT—16 times daily for 5 days, eight times daily for 7 days, and then six times daily for 7 days—followed by four daily doses as maintenance therapy and exhibits signs and symptoms of deterioration, then topical steroid therapy would be started, and the eye would be reassessed frequently. A potential management mistake was the sudden—rather than tapered—withdrawal of the steroid prescribed for misdiagnosed HSV keratitis upon the initiation of a suboptimal dose of AAT. Corneal vascularization often leads to the resolution of AK.
In my experience, the outcome of therapeutic keratoplasty for AK is poor. I would perform the procedure only for corneal perforation. I would avoid DALK here.
The patient’s history of CXL may be germane to her situation. The procedure can have a prolonged effect on corneal keratocyte levels, which are involved in orchestrating the innate immune response. The principal reason for the poor outcome in this case, however, is the 12-month diagnostic delay. AK is as common as primary HSV keratitis among contact lens wearers in the United Kingdom, where I practice. Unfortunately, AK was not considered at the outset. The oversight was then compounded by a failure to reassess the diagnosis after the patient’s poor response to HSV treatment.


ALEJANDRO NAVAS, MD, PHD, AND RUTH ESKENAZI BETECH, MD
Because AAT elicited some response, we would consider topical steroid therapy. Prednisolone acetate 1% dosed two or three times daily would be initiated, and the AAT would be tapered. If a better response is observed without signs of reinfection, then the frequency of steroid administration would be increased slowly and the AAT taper would continue.
Topical steroids must be used judiciously because of the potential risks of worsening the AK and producing suboptimal outcomes, especially if the agents are administered before AAT is initiated.1,2 Interestingly, topical steroids have been shown to accelerate trophozoite excystment, which may increase trophozoites’ susceptibility to AAT.3 In our practice, we usually start steroid treatment to limit severe corneal inflammation when a dramatic clinical improvement is observed after 2 to 4 weeks of AAT.
Therapeutic keratoplasty should be reserved for eyes with impending or actual corneal perforation and to eradicate the organism when keratitis is unresponsive to medical treatment.4
DALK can be considered in patients with advanced AK. There is a risk of recurrent disease, but the speed of invasion is usually slower in AK than other infections. There is therefore a better chance of total removal of the infected corneal tissue. The patient is at increased risk of endothelial rejection because of her history of corneal inflammation and the presence of deep corneal neovascularization. For this reason, we would attempt DALK.4,5
There is no solid evidence, but it is possible that CXL had a stabilizing effect on the collagen of the cornea, limiting the tissue damage.

ELMER TU, MD
Because some control of the infection has been achieved, antiacanthamoebal medications would be reduced slowly. The patient would be observed for signs of worsening.
Adding corticosteroids after 2 to 3 weeks of effective AAT would not worsen the prognosis, but I would add them only for a demonstrable benefit of preserving corneal clarity, preventing central corneal neovascular invasion, or reducing pain. In the current situation, the changes in the cornea appear to be irreversible, limiting the benefit of corticosteroid therapy for a patient committed to undergoing high-risk keratoplasty for vision restoration in the future.
For late-stage AK, I recommend therapeutic keratoplasty only if the disease is progressing or if recovery has stalled definitively despite maximum-tolerated medical therapy. The utility of the intervention for early, superficial Acanthamoeba remains debatable when DALK may be an option.
I do not perform DALK to treat full-thickness disease or if there is no clear exclusion of the posterior stroma. Numerous studies, however, have shown shallow infections to be amenable to DALK.6,7
The cornea is likely depleted of immune elements soon after CXL, but the effect may not remain after 2 years. Persistent corneal hypesthesia after CXL as a mechanism of susceptibility, however, may continue. Any break in contact lens hygiene, including showering while wearing the lenses, heightens the risk of infection. The greatest proportional risk is contact lens wear itself. Gas permeable contact lenses worn for orthokeratology are associated with a significantly elevated risk, and a concerning increase in the instances of AK among wearers of gas permeable hybrid and scleral lenses was reported recently.8,9

WHAT I DID: ALAN N. CARLSON, MD
In the absence of a history of oral fever blisters, HSV serology—if negative—is an excellent test to rule out HSV, as in this case. The patient was treated for presumed HSV, which is the misdiagnosis that most often delays a correct diagnosis of Acanthamoeba. Confocal microscopy identified the organism cysts and facilitated a prompt change to the correct treatment.
Acanthamoeba can cause severe pain, particularly when corneal perineuritis develops (Figure 5). A modified post-PRK regimen was helpful here. It included pouring two bottles of chilled saline onto the cornea followed by the instillation of one drop each of atropine 1%, cyclopentolate 1%, phenylephrine 10%, and an NSAID. An oral narcotic was administered to enhance pain control. Treatment with a topical corticosteroid is relatively contraindicated during the early phase of active Acanthamoeba infection. It is typically reserved for the later stages of infection to reduce corneal neovascularization and scarring.
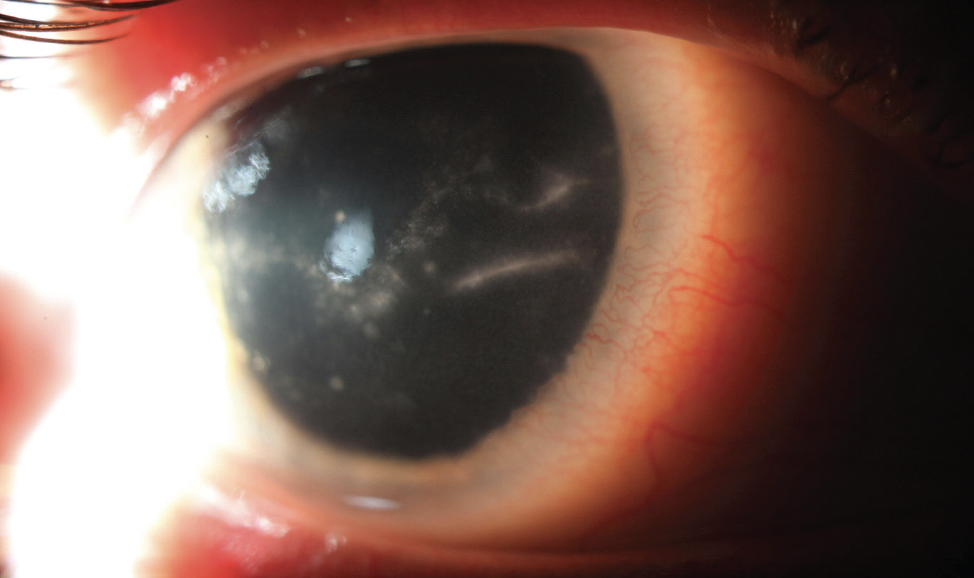
Figure 5. The eye of another patient treated for Acanthamoeba keratitis has prominent corneal nerves surrounded by inflammatory white cells.
Acanthamoeba tends to respect Descemet membrane as a barrier, whereas organisms such as Fusarium can easily traverse it. A hypopyon in the setting of Acanthamoeba infection is therefore usually sterile. DALK is an excellent option in any eye with quiescent disease because corneal neovascularization is common, increasing the risk of rejection with a full-thickness penetrating keratoplasty.
The patient was treated successfully once the correct diagnosis was made with confocal microscopy and confirmed with a corneal culture and biopsy. Treatment was aggressive, specifically targeting Acanthamoeba and pain control. It involved patiently adding topical steroids to reduce inflammation and corneal neovascularization. Ultimately, surgical intervention was necessary to reestablish useful vision.
1. Carnt N, Robaei D, Watson SL, Minassian DC, Dart JK. The impact of topical corticosteroids used in conjunction with antiamoebic therapy on the outcome of Acanthamoeba keratitis. Ophthalmology. 2016;123(5):984-990.
2. Robaei D, Carnt N, Minassian DC, Dart JK. The impact of topical corticosteroid use before diagnosis on the outcome of Acanthamoeba keratitis. Ophthalmology. 2014;121(7):1383-1388.
3. Maycock NJ, Jayaswal R. Update on Acanthamoeba keratitis: diagnosis, treatment, and outcomes. Cornea. 2016;35(5):713-720.
4. Robaei D, Carnt N, Minassian DC, Dart JK. Therapeutic and optical keratoplasty in the management of Acanthamoeba keratitis: risk factors, outcomes, and summary of the literature. Ophthalmology. 2015;122(1):17-24.
5. Taenaka N, Fukuda M, Hibino T, et al. Surgical therapies for Acanthamoeba keratitis by phototherapeutic keratectomy and deep lamellar keratoplasty. Cornea. 2007;26(7):876-879.
6. Bagga B, Garg P, Joseph J, Mohamed A, Kalra P. Outcome of therapeutic deep anterior lamellar keratoplasty in advanced Acanthamoeba keratitis. Indian J Ophthalmol. 2020;68(3):442-446.
7. Sarnicola E, Sarnicola C, Sabatino F, Tosi GM, Perri P, Sarnicola V. Early deep anterior lamellar keratoplasty (DALK) for Acanthamoeba keratitis poorly responsive to medical treatment. Cornea. 2016;35(1):1-5.
8. Scanzera AC, Yoon HH, Shorter E, Tu E. Risk factors and contact lens modality in patients with Acanthamoeba keratitis. IOVS. 2022;63:539-A0237.
9. Scanzera AC, Tu EY, Joslin CE. Acanthamoeba keratitis in minors with orthokeratology (OK) lens use: a case series. Eye Contact Lens. 2021;47(2):71-73.