CASE PRESENTATION
A 62-year-old man reports experiencing a decrease in vision. The patient is employed as a high-end wallpaper hanger and says that he can no longer see well enough to align the patterns. He currently does not wear spectacles.
The patient underwent LASIK approximately 20 years ago. He says that he might have received a LASIK enhancement in the right eye at some point. He wore glasses before having refractive surgery but is unsure if he was farsighted or nearsighted.
On presentation, his UCVA is 20/40 OD and 20/70 OS. His BCVA is 20/30 with a manifest refraction of -1.00 + 0.50 x 010º OD and 20/25 with a manifest refraction of -1.00 + 0.25 x 090º OS. IOP readings with a Tono-Pen (Reichert) are 7 mm Hg OD and 9 mm Hg OS.
An examination finds a patch of epithelial ingrowth superonasally under the flap in the right eye. The ingrowth measures approximately 2 mm at its widest point. Each eye has a 2+ nuclear sclerotic cataract. A fundus examination and macular OCT imaging of both eyes are normal. Figures 1 through 3 show other preoperative measurements.
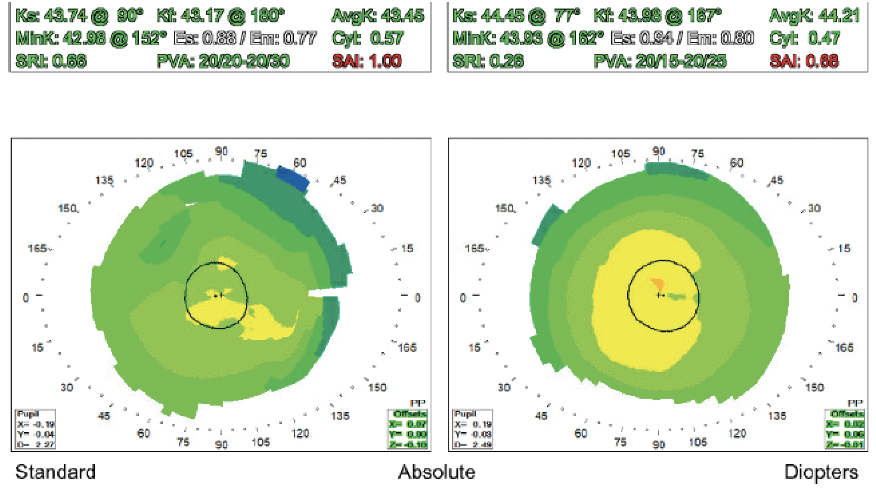
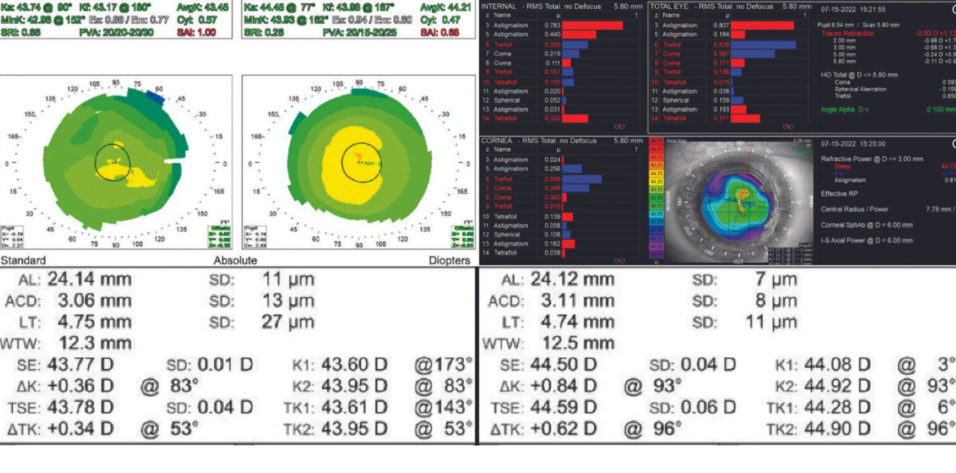
Figure 1. Placido disc topography of both eyes.
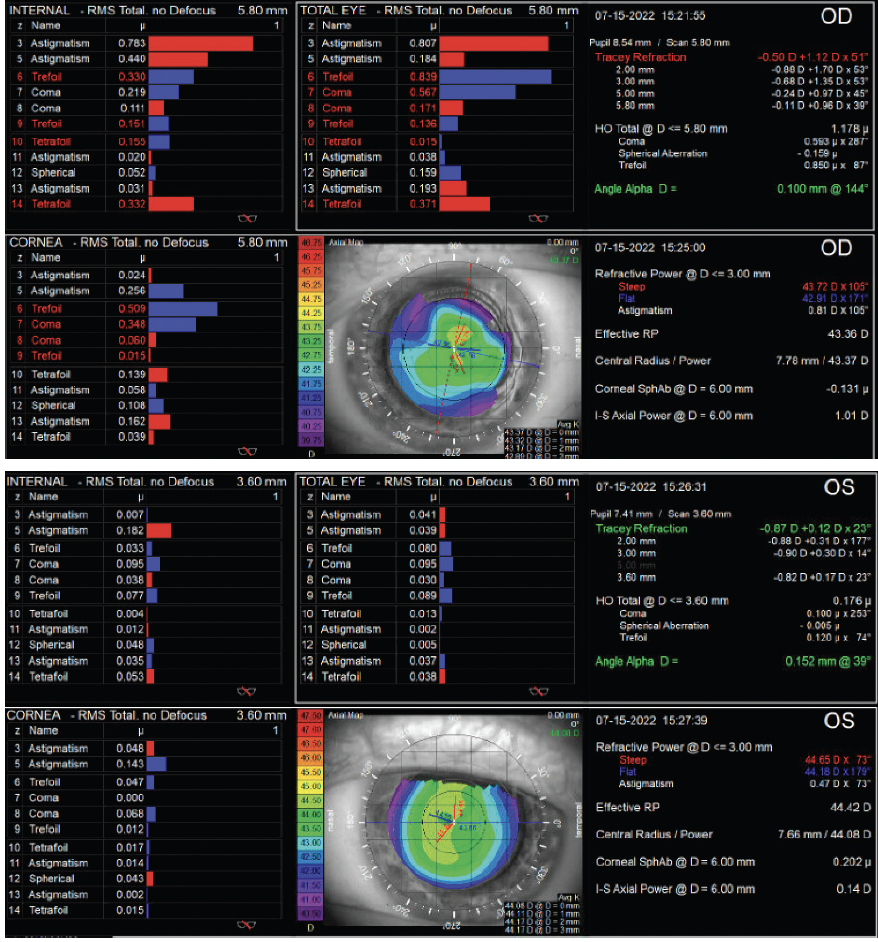
Figure 2. Change analysis for both eyes.
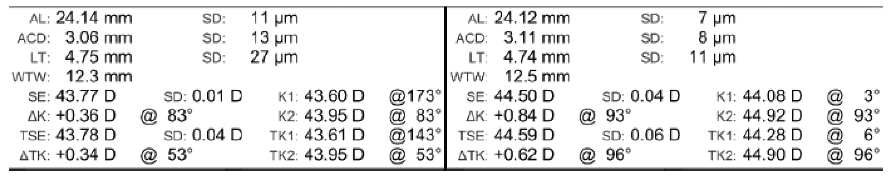
Figure 3. IOLMaster 700 data for both eyes.
The patient desires spectacle independence at all distances but especially when hanging wallpaper because he plans to continue working for the foreseeable future. How would you proceed?
—Case prepared by Julie Schallhorn, MD, MS

CLAUDIA PEREZ-STRAZIOTA, MD
The epithelial ingrowth present in the right eye is neither inducing significant topographic irregularity nor causing symptoms and is therefore best left alone. I would not lift the LASIK flap.
Judging from the central steepening pattern on topography, the patient underwent hyperopic LASIK. The procedure puts him at increased risk of inaccurate IOL power calculations, which should be discussed with him. His candidacy for a refractive enhancement after cataract surgery is uncertain. Preoperative Scheimpflug imaging could be useful depending on his motivation for spectacle independence.
The patient’s primary concerns appear to center on quality of vision rather than night vision symptoms or refractive error. The bilateral implantation of a monofocal IOL would provide him with the best quality of vision, whereas the placement of extended depth of focus (EDOF) IOLs—prioritizing distance and intermediate visual acuity—would deliver the best functional range of uncorrected vision.
Patients can have regular or irregular astigmatism after LASIK, and many have altered spherical aberration profiles. This patient has minimal regular cylinder and does not require astigmatism correction with a toric IOL. His spherical aberration profile is nearly neutral in both eyes (+0.10 D OD and -0.04 D OS), so most optic options are reasonable here.
His occupation requires good intermediate acuity and depth perception. I would hesitate to recommend monovision. Instead, I would recommend the bilateral implantation of an EDOF IOL and a refractive target between plano and -0.50 D in each eye to maximize functional intermediate visual acuity.

BRIAN M. SHAFER, MD
The clinical vignette presented here is becoming increasingly common. Patients who underwent excimer laser ablation in the late 1990s while they were in their 40s are now cataract age. They all remember the 20 years of spectacle independence they enjoyed before their “LASIK wore off.” Aside from the rare case of ectasia, these individuals are experiencing dysfunctional lens syndrome.
The patient has tolerated residual ametropia and decreased BCVA well, indicating that he is committed to spectacle independence even if it means sacrificing perfect vision. Based on the iTrace (Tracey Technologies) data, his corneal higher-order aberrations (HOAs)—including coma and trefoil—are elevated. Based on these values, I would avoid diffractive multifocal and EDOF IOLs. The HOAs are relatively normal, and Placido disk imaging is regular in the left eye. I nevertheless generally avoid multifocal technology in patients with a history of refractive surgery because it is difficult to nail the refractive target. The result could likely be fine-tuned but is less predictable overall.
A Light Adjustable Lens (LAL; RxSight) would be my preference for both eyes. To hang wallpaper, the patient likely needs excellent intermediate and functional near vision. I would start with a plano target in both eyes and probably move toward a final target of -1.25 D in the nondominant eye. The induced spherical aberration may provide excellent intermediate and near vision in the nondominant eye and crisp distance vision in the dominant eye. This approach has been successful for similar patients in my clinic, most of whom achieved the spectacle independence they desired.

O. BENNETT WALTON IV, MD, MBA
The epithelial ingrowth has encroached by only 2 mm, and further progression would be unexpected. The removal of epithelial ingrowth has not been associated with a reduction in HOAs.1
Especially if the patient’s left eye is dominant at distance with best correction, I would plan to implant an IC-8 Apthera IOL (AcuFocus) in the right eye and target a -0.75 D refraction. The off-label use of this IOL could reduce the visual effect of his HOAs while delivering an extended range of vision. I should note that I have not implanted one of these lenses yet.
I would implant an LAL in the left eye to allow customization of the patient’s vision while the right eye heals. The LAL is a monofocal, but my patients tend to end up with a greater range of vision than expected. This could allow the patient to tailor his binocular vision profile to optimize the tradeoff between distance and near. If forced to pick a nonadjustable lens, my usual preference is an EDOF IOL for patients with a history of refractive surgery.
The patient in this case, however, needs to be able to perceive extremely fine, close detail for precise wallpaper pattern matching. I would therefore feel comfortable offering a trifocal IOL for the left eye after an extended discussion with the patient.
Whenever post-LASIK topography is neither obviously very flat nor centrally steep, I check the Gullstrand ratio on the Pentacam (Oculus Optikgeräte), displayed as a fractional ratio of posterior to anterior radius of curvature. A result that is less than 83% indicates a prior myopic ablation, more than 83% indicates a prior hyperopic ablation, and anything around 83% was mostly astigmatism. The number can guide my choice of IOL calculation.

WHAT I DID: JULIE SCHALLHORN, MD, MS
As the panelists note, the patient is not a good candidate for a diffractive multifocal IOL in the right eye because of the irregular topography and because his history of (likely) hyperopic LASIK renders achieving the refractive target less predictable. He was counseled extensively that he was unlikely to achieve total spectacle independence. He therefore agreed on a distance/intermediate target and wearing readers for detailed near work.
The patient declined an offer for the LAL owing to concerns about cost. An IC-8 Apthera would have been an excellent choice for the right eye but unfortunately was not available at the time. He received an enVista IOL (Bausch + Lomb) with zero spherical aberration in each eye. The left eye was targeted for distance, and -1.00 D was targeted in the right eye.
One month after surgery, the patient’s refraction was -1.25 +0.25 x 090º OD and -0.25 D OS. His UCVA was 20/50 and J3 at near OD and 20/20 OS. He stated that he could function well on a daily basis.
1. Chen LY, Kung JS, Manche EE. Management of complex epithelial ingrowth after laser in situ keratomileusis using fibrin tissue glue. Eye Contact Lens. 2018;44(suppl 2):S210-S214.