Even when we perform LASIK on patients with stable refractive errors, 10 years after surgery their refractive condition may be different than it was initially postoperatively. Chances are, by this time, patients will have reverted to needing spectacles for at least some of their daily activities. In my practice, this is most often the case after myopic LASIK. I rarely perform hyperopic LASIK because younger hyperopic patients tend to adapt to their vision and do not present for refractive surgery; on the other hand, older patients are rarely satisfied with their vision after LASIK. When that is the case, I suggest refractive lens exchange (RLE).
The way I manage recurrent myopia 10-plus years after the original LASIK procedure depends on the amount of ametropia and on the patient’s needs and age. As a rule, I prefer not to treat unilateral low myopia because of the pseudoaccommodation it offers. When the defect is bilateral, the first corrective option is for patients to use spectacles for driving, especially at night. Many patients may be satisfied with this course for a number of years but then request a surgical solution, posing new challenges to surgeons.
In my practice, patients younger than 40 years are offered a LASIK retreatment. We use anterior segment OCT to check flap thickness and estimate the residual thickness after the new ablation. I take into account that these patients have aged and their corneas have stiffened since their original surgeries. Specifically, I will treat patients at this age with an expected residual bed thickness as low as 250 µm—a level not accepted for patients younger than 30. If the cornea is much too thin, I plan to perform PRK with mitomycin C and counsel the patient about the postoperative pain associated with the procedure.
I prefer lifting the flap if a microkeratome was used during the original surgery; in my hands, this method has always been successful, even more than 10 years after the original LASIK procedure. In contrast, my attempts to cut a second, shallower flap with a microkeratome have been unsuccessful, mainly because the second flap cut often slips into the first for part of the path, producing small pieces of corneal lamella that can be impossible to manage properly. Even when I have tried to make a second flap with a femtosecond laser, the resultant bubble layer behaved the same way.
An additional problem with trying to create a shallower flap above the microkeratome flap is that the new cut frequently crosses through the uneven depth of the first cut. Even though epithelial ingrowth is considered to be more frequent after flap relifting than after creation of new cuts, I still prefer the risk of epithelial ingrowth to that of a bad flap. Special maneuvers and careful surgery will reduce the risk of epithelial ingrowth to a minimum.
The steps I use to lift the flap and perform enhancement are outlined in Retreatment Protocol (pg 64).
KEEP IN MIND
The chief complaint of post-LASIK patients with recurrent myopia who are now also presbyopic is usually worsening distance vision. Keep in mind that these patients may not understand what presbyopia is. In these cases, after I explain the condition to them, the first option I discuss is how to improve their vision with unilateral surgery, but this option carries the same technical challenges already mentioned.
I always make patients undergo a unilateral contact lens trial to simulate their postoperative vision. They are then asked to choose which eye they prefer for distance vision; this is the eye that will undergo retreatment. If patients are not satisfied with the results of their unilateral contact lens trial, I plan a bilateral procedure. In these cases, however, poor near vision can result postoperatively. Therefore, I make it clear to patients preoperatively that their improved distance vision will substitute for their near vision—not add to it.
OTHER OPTIONS
Multifocal phakic IOLs. If the patient wants bilateral surgery and spectacle independence, or if the myopic defect is higher than 2.00 D, then multifocal phakic IOLs are an option because they provide good distance and near vision. These lenses have recently become available in my country, helping us to solve the problem of recurrent myopia in the 40- to 50-year-old post-LASIK patient. (At the moment, I would not propose multifocal phakic IOLs to emmetropic post-LASIK patients, however.)
IOL power calculations for phakic IOLs are easier than for post-LASIK cataract surgery, mainly because they are aided by the refractive error (Figure 1). Because the vast majority of these patients retain some accommodation, the base IOL power provides the distance and intermediate vision after surgery, and the near add helps with close reading. Because of these patients’ increasing age, and because these IOLs are designed to stay in place until cataract surgery, I usually propose the full near add (3.00 or 4.00 D) because it should compensate for their loss of accommodation with time (Figure 2).
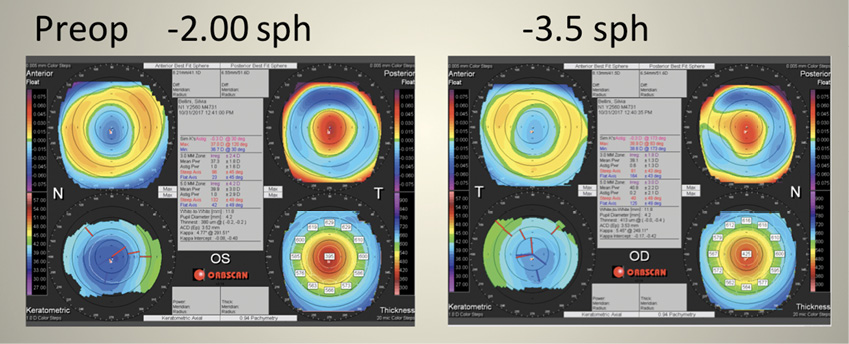
Figure 1. Topography of a 48-year-old patient’s left (A) and right (B) eyes several years after undergoing LASIK.
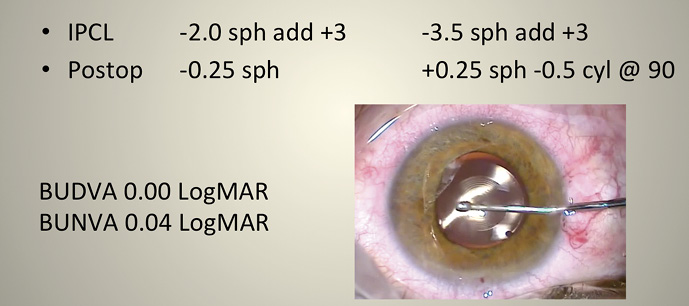
Figure 2. Postoperative results in a patient who underwent post-LASIK retreatment with implantation of multifocal phakic IOLs.
Refractive lens exchange. I usually suggest RLE to patients older than 55 years or to those who want a definitive surgery. For RLE, the preoperative assessment includes corneal topography and tomography and posterior OCT to rule out any retinal problem that might jeopardize the result. When selecting the IOL power, I prefer the Barrett True-K formula. I make every patient aware of the chance of a postoperative refractive error and of the possible need for a laser touchup.
I use a femtosecond laser to assist with the RLE surgery, which I have found improves the precision and repeatability of the procedure. Frequently these patients ask for a multifocal IOL because they do not want to wear spectacles for near vision. The use of multifocal IOLs after LASIK is supported by the literature,1,2 and I have had good experiences with them. None of my patients has reported any more photic phenomena disturbances than those who have not undergone LASIK.
My preferred IOL is the POD F IOL (PhysIOL), but I tend to avoid multifocals in highly myopic eyes because of the frequent presence of macular impairment. If the preoperative corneal topography shows some irregularity, then the IC-8 IOL (AcuFocus) is my choice. I implant this lens unilaterally in the nondominant eye.
Although I have not observed more dry eye problems after lens surgery in these patients than in non-LASIK patients, I always prescribe artificial tears for at least 3 to 4 weeks postoperative.
Retreatment Protocol
The original flap border is identified at the slit lamp and inspected to check whether the hinge is lateral or superior. Next, the epithelium is marked with a 30-gauge needle close to the hinge. At the excimer laser microscope, a thin, blunt instrument penetrates the interface and dissects and lifts the flap, if possible starting from the hinge, without exiting and entering several times. According to many surgeons, this maneuver minimizes the risk of epithelial ingrowth. A water jet may be used at this time, but the irrigating balanced saline solution must be dried immediately so that corneal tissue is not hydrated. The new ablation is planned using the patient’s refraction, with no attempt to improve corneal topography—even in the case of small decentration. The reason for this is that if the patient has been happy for so many years, we have only to correct the refractive error. The postoperative regimen is the same as for regular LASIK.
CONCLUSION
Post-LASIK patients are likely to experience regression 10 years after surgery. Today, we have a plethora of options to address their visual symptoms and provide them with many more years of unaided vision. Unilateral surgery, flap relifting and retreatment, phakic monofocal or multifocal IOL implantation, and RLE with aphakic monofocal or multifocal IOLs are valuable tools.
1. Yoshino M, Minami K, Hirasawa M, Oki S, et al. Clinical results of diffractive multifocal intraocular lens implantation after laser in situ keratomileusis [in Japanese]. Nippon Ganka Gakkai Zasshi. 2015;119(9):613-618.
2. Fisher B, Potvin R. Clinical outcomes with distance-dominant multifocal and monofocal intraocular lenses in post-LASIK cataract surgery planned using an intraoperative aberrometer. Clin Exp Ophthalmol. 2018;46(6):630-636.