CASE PRESENTATION
A 68-year-old man with no history of rheumatoid disease presented for a second opinion regarding his right eye. His visual acuity had not improved after cataract surgery 2 years earlier.
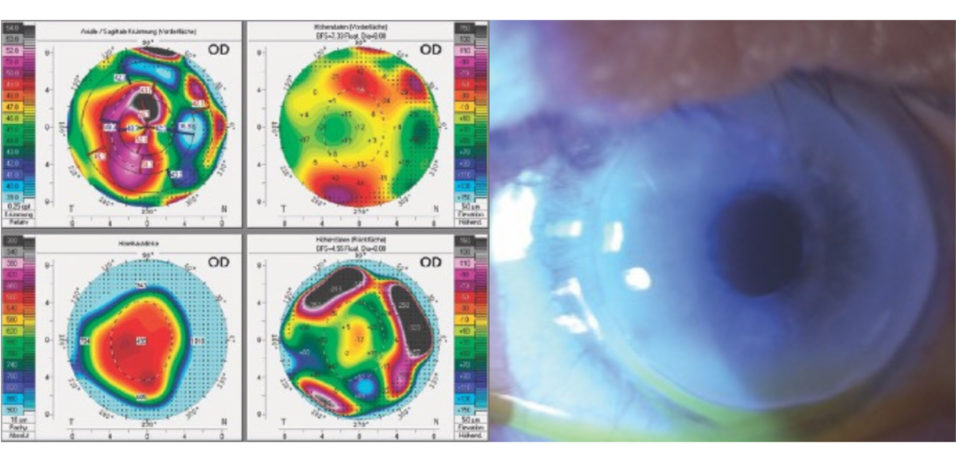
The patient’s manifest refraction was +0.50 -8.50 x 167º = 20/40 OD and -2.00 -2.25 x 177º = 20/63 OS. Lens opacification was evident in the left eye. He had circular Terrien marginal degeneration (TMD) with obvious thinning of the peripheral cornea in both eyes. Imaging with the Pentacam (Oculus Optikgeräte) showed 7.30 D of irregular corneal astigmatism and a maximum keratometry value of 52.60 D OD (Figure 1). A slit-lamp examination found a clear central cornea but peripheral deposition of lipid in each eye (Figure 2).
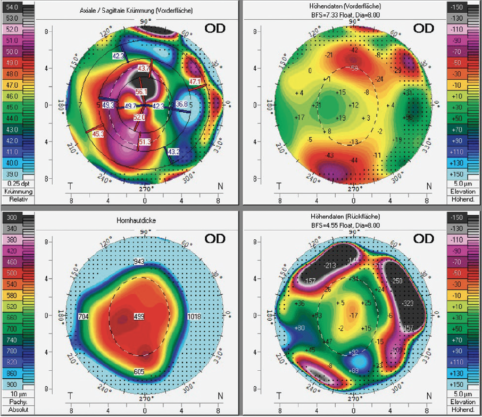
Figure 1. Two years after cataract surgery and the placement of an IOL in the bag, imaging with the Pentacam showed 7.30 D of irregular corneal astigmatism and a maximum keratometry value of 52.60 D OD.
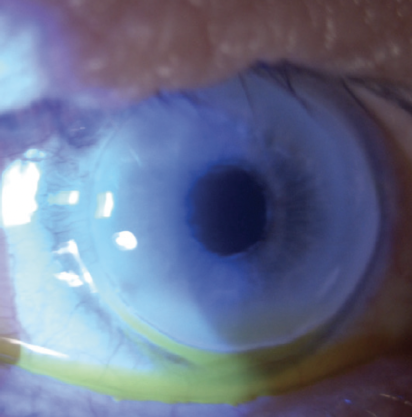
Figure 2. A slit-lamp examination of the right eye found a clear central cornea, peripheral lipid deposition, and marked circular thinning. The appearance of the left eye was similar.
Cataract surgery on the left eye was uncomplicated. Postoperatively, his visual acuity was +0.75 -0.25 x 108º = 20/16 OS. The patient remained dissatisfied with the vision in his right eye. Spectacle correction was of no help, and a contact lens could not be fitted.
How would you proceed?
—Case prepared by Suphi Taneri, MD, FEBOS-CR

ASHRAF ARMIA BALAMOUN, MD, MSC, FRCS (GLASG), FACS
TMD is an infrequent, idiopathic, usually bilateral (56%–86%),1 asymmetric, and often progressive disease that causes thinning, neovascularization, and lipid infiltration at the corneal periphery. The epithelium is, by definition, intact if patients do not have systemic autoimmune diseases. Patients are usually diagnosed with TMD at around 40 years of age, but the disease can manifest between the ages of 10 and 87 years. Male sex is often considered to be a predisposing factor for TMD. Astigmatism, which can be as high as 30.00 D and irregular, is the main cause of reduced vision.2-4
The most sensitive way of detecting TMD progression is to identify a reduction in peripheral corneal thickness, preferably measured from the anterior stromal surface, with anterior segment OCT. Observation is essential because progression is common and varies in speed.
Figure 1 shows a large irregularity in the anterior corneal surface. Spectacle correction was of no help, and a contact lens could not be fitted. My thoughts would therefore turn to crescentic lamellar or full-thickness keratoplasty. A lamellar keratoplasty replaces only the Bowman layer and the anterior portion of the corneal stroma. The Descemet membrane, the endothelium, and the deep stroma are left intact. A crescentic lamellar keratoplasty replaces only the thinned portion of the patient’s cornea with a crescent-shaped donor cornea. Lamellar keratoplasty is often preferred over penetrating keratoplasty because the former is associated with a decreased risk of graft rejection, irregular astigmatism, and corneal opacification.5,6
Crescent-shaped lamellar and full-thickness corneoscleral patch grafts have been reported to arrest the progression of severe against-the-rule astigmatism for up to 20 years.7 Annular lamellar keratoplasty grafts may be required in severe cases of 360º marginal degeneration.

VINICIUS S. DE STEFANO, MD, PHD, AND RONALD R. KRUEGER, MD
TMD is usually found along the superior limbus, so the presence of 360º circular TMD is unusual and signals advanced disease. There is no history of rheumatologic disease. Disorders such as a previous Mooren ulcer, however, can be considered. Information on the patient’s race, ethnicity, and history could assist with clinical and surgical decision-making.
A critical point is to ensure that the corneal findings are relatively stable. Any surgical planning in a patient like this can be quickly invalidated if TMD is actively progressing. Also important is to determine the regional extent of corneal thinning (not apparent in Figure 1) when planning surgical incisions. The corneal axial curvature map shows extreme with-the-rule astigmatism that matches the manifest refraction cylinder axis. The central aspect of the cornea is somewhat regular, so implanting a toric IOL is a reasonable approach if the disease is stable.
A staged surgical approach is warranted in this situation. If a standard monofocal IOL was implanted in the right eye, we would perform an IOL exchange and target maximum astigmatism correction. This by itself cannot eliminate the entire amount of astigmatism present, but the reduction may allow the patient to tolerate the use of spectacles or contact lenses. Additional approaches can be considered if further treatment is necessary. One option would be to implant a secondary IOL using either piggyback techniques with another toric IOL or the placement of a Visian Toric ICL (off-label use; STAAR Surgical). Thereafter, depending on the amount of residual astigmatism, the option of a surface ablation procedure could be entertained if a minimal ablation depth is required, given the already thin pachymetry measurements.
Before each surgical intervention, it would be essential to set realistic expectations and explain to the patient that crisp spectacle-free vision may be impossible to achieve in the right eye.

FARHAD HAFEZI, MD, PHD, FARVO
The patient has distinct irregular astigmatism due to TMD, a rare bilateral condition characterized by progressive peripheral thinning of the stroma. I would obtain anterior segment OCT imaging to assess the extent of the thinning. Based on the information provided, my goals would be twofold.
Goal No. 1: Stabilize the TMD to avoid further thinning. Until recently, no treatment was available to stop sterile melting in eyes with TMD. In 2015, I published the first case report of successful TMD treatment and a partial reversal of the peripheral thinning.8 CXL of the entire cornea, including the limbal area, arrested sterile melting by blocking catalysis of the stromal collagen. (Read the article.)
Goal No. 2: Achieve visual rehabilitation. Once the stromal thinning has stopped, I would consider a scleral contact lens fitting. Even if long-term wear remains impossible, the approach could help to determine and simulate the best possible correction of aberrations and thereby avoid postoperative patient dissatisfaction.
Wavefront-guided transepithelial PRK would follow. Both the anterior and posterior surfaces are affected by TMD, so I would carefully compare ablation profiles based on corneal and total wavefront data because currently available corneal aberrometers provide only anterior corneal surface data. After careful planning, I would perform transepithelial PRK using an algorithm that my colleagues and I developed to compensate for differences in the response to excimer laser ablation demonstrated by corneas that have previously undergone CXL.9
Three months after transepithelial PRK, I would assess whether the use of pilocarpine 1.25% eye drops can improve the patient’s visual acuity and quality of vision. I would exercise caution regarding this form of therapy owing to recent retinal findings in patients who have used pilocarpine regularly.10

WHAT I DID: SUPHI TANERI, MD, FEBOS-CR
The challenges of this unusual case are reflected in the broad spectrum of possible treatment approaches described by the panelists. Extraction of the IOL from the patient’s right eye was deemed too risky because the lens had been in place for 2 years. Because IOLs with the cylindrical power required were not readily available, a customized three-piece, foldable, add-on lens (Torica-sPB, HumanOptics) with 11.00 D of cylinder was implanted in the ciliary sulcus. The optical material is silicone elastomer, and its haptic material is PMMA with a high molecular weight. The total diameter of the IOL is 14 mm, and it has an effective optical zone of 6 mm.
Unfortunately, the IOL that was implanted ceased to be available after 2017. Today, several alternatives are available for implantation in the sulcus in front of another IOL placed in the capsular bag.
One month after surgery, the patient’s BCVA was 20/20 with a manifest refraction of +1.25 -1.75 x 134º OD, and he was very happy.
1. Ruutila M, Fagerholm P, Lagali N, et al. Diagnostic criteria for Terrien marginal degeneration: Nordic Terrien Degeneration Study. Cornea. 2021;40(2):133-141.
2. Chan AT, Ulate R, Goldich Y, Rootman DS, Chan CC. Terrien marginal degeneration: clinical characteristics and outcomes. Am J Ophthalmol. 2015;160(5):867-872.e1.
3. Wang N, Wang CX, Lian XF, Duan SJ, Huang D, Zhou SY. Staging of development in Terrien’s degeneration based on corneal curvatures detected by optical coherence tomography. Graefes Arch Clin Exp Ophthalmol. 2015;253(10):1757-1764.
4. François J. La dégénérescense marginale de la cornée. Arch Ophtalmol (Paris). 1936;53:432-540, 612.
5. John V, Goins KM, Afshari NA. Deep anterior lamellar keratoplasty. EyeNet Magazine. September 2007. Accessed August 10, 2022. http://www.aao.org/publications/eyenet/200709/pearls.cfm
6. Cheng CL, Theng JT, Tan DT. Compressive C-shaped lamellar keratoplasty: a surgical alternative for the management of severe astigmatism from peripheral corneal degeneration. Ophthalmology. 2005;112(3):425-430.
7. Wisner DM, Mihlstin ML, Hwang FS, Bunya VY, Nallasamy N, Kaufman AR. Terrien’s marginal degeneration. EyeWiki. Updated August 8, 2022. Accessed September 9, 2022. https://eyewiki.aao.org/Terrien%27s_Marginal_Degeneration
8. Hafezi F. Reversing peripheral thinning in Terrien marginal degeneration. Cataract & Refractive Surgery Today Europe. April 2015. Accessed August 10, 2022. https://crstodayeurope.com/articles/2015-apr/reversing-peripheral-thinning-in-terrien-marginal-degeneration
9. Richoz O, Arba Mosquera S, Kling S, et al. Determination of the excimer laser ablation rate in previously cross-linked corneas. J Refract Surg. 2014;30(9):628-632.
10. Al-Khersan H, Flynn HW Jr, Townsend JH. Retinal detachments associated with topical pilocarpine use for presbyopia. Am J Ophthalmol. 2022;242:52-55.