Sponsored By

10 Years of SMILE
On May 3, 2006, Walter Sekundo, MD, PhD, of Philipps University of Marburg, Germany, helped to lay the groundwork for an innovative approach to laser vision correction: He performed the first ReLEx FLEx on a seeing human eye using a clinical prototype of the VisuMax femtosecond laser (Carl Zeiss Meditec) to cut an intrastromal lenticule and a flap. The refractive correction was achieved by lifting the flap and extracting the lenticule manually. In the more than 10 years since that first procedure was successfully completed, ReLEx has matured into small incision lenticule extraction, or SMILE, a minimally invasive procedure involving the removal of a refractive lenticule through a keyhole incision in the cornea. Today, SMILE is an extremely predictable procedure, Professor Sekundo said during the 2017 European Refractive Laser Symposium in Paris.
According to Professor Sekundo, patients love the idea of “keyhole” surgery because it eliminates the need to create a corneal flap. He also pointed out that SMILE shortens the duration of surgery to about 24 to 27 seconds and that comparative studies have shown equal or better results with SMILE than with femtosecond LASIK. Another important take-home message for patients is that SMILE alters corneal biomechanics to a lesser extent than flap-based refractive correction procedures,1 thereby causing less dry eye symptoms postoperatively.
SMILE has now been performed more than 750,000 times worldwide, and, according to Professor Sekundo, indications for the procedure will continue to expand. In the future, the treatment of hyperopia by lenticule extraction as well as lenticule implantation techniques for the treatment of keratoconus and hyperopia are likely, he said.
Laser Parameter Optimization
In the 10-year history of SMILE, there have been several key advancements that have helped to improve postoperative outcomes. For instance, small changes in laser parameters can make a big difference in patients’ visual recovery after SMILE, Claudio Orlich, MD, said at the ZEISS symposium. Dr. Orlich observed that, by modifying energy settings with tendency to decrease the energy level, the length of patients’ visual recovery times could be decreased. In order to determine the best laser parameters, Dr. Orlich compared visual recovery in patients treated with SMILE using an energy of 175 nJ, a spot spacing of 4.5 µm for the lenticule, and a spot distance of 2 µm for the sidecut (group 1) to the visual recovery in patients treated with SMILE using an energy of 125 nJ, a spot spacing of 4.3 µm for the lenticule, and a spot distance of 1.8 µm for the sidecut (group 2). The posterior surface of the lenticule was created with a spiral-in pattern and the anterior surface with a spiral-out. One day after surgery, 35.86% of eyes in group 1 and 70.65% of eyes in group 2 achieved a postoperative UDVA of 20/20 or better. Dr. Orlich noted that using lower energy allows faster visual recovery; however, he also noted that the exact settings must be adapted to each VisuMax laser, as the same settings on different lasers do not induce the same effects on the cornea.
Compensating for Cyclotorsion in Astigmatism Treatments
Sri Ganesh, MBBS, MS, DNB, of the Nethradahama Super Specialty Eye Hospital in Bangalore, India, spoke about another hot topic that can help to achieve better postoperative outcomes in eyes with high astigmatism: compensating for cyclotorsion.
During the ZEISS symposium, Dr. Ganesh provided the first outcomes with manual compensation of cyclotorsion* in eyes with clinically significant astigmatism that were treated with SMILE (Figure 1). He found that, although it may not be necessary in low astigmatism, especially if the cyclotorsion is lower than 5º, it is beneficial to manually compensate for cyclotorsion in moderate to high cylinders (>1.50 D). Based on his findings, Dr. Ganesh recommends that manual compensation for cyclotorsion of any degree be performed for all eyes with high astigmatism (>1.50 D) but only for 5º or more in eyes with low astigmatism (≤1.50 D).

Figure 1. Accuracy of corrections in the compensation and no compensation groups.
*Off-label use
Long-Term Results: SMILE Passes the Test of Time
Myopia correction with SMILE is efficacious, predictable, stable, and safe, said Jana Gertnere, MD, of Dr. Solomatin Eye Center in Riga, Latvia, during the ZEISS symposium. This was the conclusion that Dr. Gertnere reached after reviewing the 5-year postoperative results of 42 patients treated with SMILE for myopia and myopic astigmatism between 2011 and 2012. Preoperatively, the mean sphere, cylinder, and spherical equivalent in eyes were -4.35 D, -0.30 D, and -4.50 D, respectively; at 5 years postoperative, they were -0.15 D, -0.15 D, and -0.23 D. Further, 88% of eyes were within ±0.50 D of intended correction and 100% were within ±1.00 D. Figure 2 shows the cumulative visual acuity.
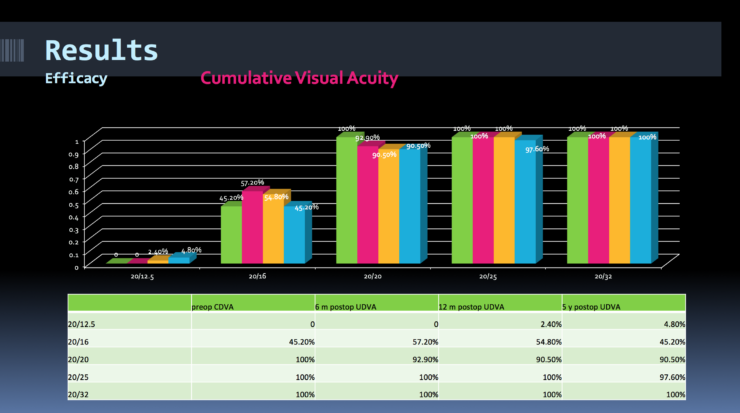
Figure 2. Myopic SMILE: Cumulative visual acuity at 6 and 12 months postoperative.
US Status of SMILE
Not only is SMILE standing its ground in Europe and Asia, but, in the United States, SMILE is gaining momentum. John F. Doane, MD, of the Discover Vision Centers in Kansas City Missouri, shared the current status of SMILE in the United States, where it received FDA approval in September 2016 for the treatment of spherical myopia from -1.00 to -8.00 D. Since then, SMILE has already been performed in more than 46 clinics. With about 50 US surgeons trained in the technique, more than 550 eyes have already been treated. One reason might be the excellent outcomes of the FDA study. Dr. Doane, who participated in the prospective, multicenter, open-label, single-arm FDA study in which 360 patients received a unilateral SMILE treatment, reported on patient outcomes at 6 and 12 months. At 6 months, 87.5% of eyes achieved 20/20 UCVA or better; by 12 months, 88.1% had (Figure 3). Additionally, by 6 and 12 months, 70% and 74% of patients, respectively, had achieved a UCVA that was better than or equal to their preoperative BCVA. This compared to only 67% of eyes that had been treated by Dr. Doane with LASIK. Further, 98.5% of eyes had a mean refractive spherical equivalent within ±1.00 D and 93% within ±0.50 D.
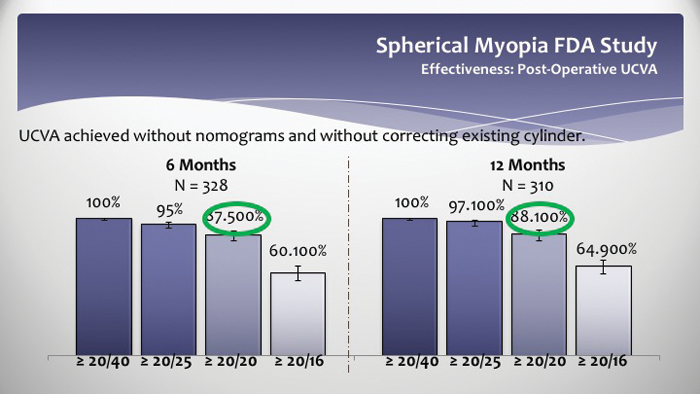
Figure 3. US FDA clinical study: UCVA achieved at 6 and 12 months postoperative.
1. Data on file with Carl Zeiss Meditec.
MASTERING PRESBYOPIA
Another hot topic at the 2017 ZEISS European Refractive Laser Symposium in Paris was how to treat presbyopic patients, with surgeons offering their tips on mastering presbyopia correction and sharing best practices. Talking about his strategies for the treatment of presbyopic patients at the symposium, Sri Ganesh, MBBS, MS, DNB, of the Nethradahama Super Specialty Eye Hospital in Bangalore, India, said that he considers four clinical decision pathways: (1) corneal inlays; (2) monovision laser vision correction procedures including SMILE; (3) PRESBYOND Laser Blended Vision; and (4) refractive lens exchange with a multifocal or presbyopic phakic IOL.
Matteo Piovella, MD, of the Centro di Microchirurgia Ambulatoriale in Milan, Italy, who had the AT LISA trifocal IOL (Carl Zeiss Meditec) implanted bilaterally in his own eyes, shared his tips for why and when to implant trifocal IOLs. According to Dr. Piovella, surgeons not only have to select the right patient type but the right kind of eye. He cautioned against implanting a trifocal IOL in eyes with high myopia or high hyperopia and advocated for the use of accurate biometry in IOL power calculation. Dr. Piovella concluded that trifocal IOLs now have replaced first-generation multifocal bifocal IOL technologies and are less sensitive to small amounts of postoperative refractive errors.
Another approach is PRESBYOND Laser Blended Vision. According to Glenn I. Carp, MBBCh, FC Ophth(SA), of the London Vision Clinic, PRESBYOND is intended for the treatment of emmetropic presbyopic patients and can be used to treat a wide range of refractive errors (+5.00 to -9.00 D).* Based on the induction of natural aberrations, the PRESBYOND Laser Blended Vision also corrects cylindrical errors. It can be easily enhanced in the future if required, and the majority of patients (>95%) tolerate the treatment, which is performed bilaterally and takes about 10 minutes. Visual recovery is fast, Dr. Carp said.
* The validated treatment range lies in the spherical equivalent between -8.00 and +2.00 D and for the cylinder at a maximum value of 2.00 D.



