Cystoid macular edema (CME) is a common complication that can affect visual acuity after cataract surgery. Studies using OCT or fluorescein angiography have found it to occur in up to 23% of nondiabetic patients after uncomplicated cataract surgery. Fortunately, CME does not manifest itself to a clinically significant degree in most of those patients, and it is usually reversible. Clinically significant CME occurs in 0% to 6% of nondiabetic patients and is reported in up to 56% of diabetic patients.1-7
The pathophysiology of CME after cataract surgery is not well understood, although it is believed to be the result of an inflammatory process initiated by surgical manipulation. Subsequently, vasodilatation, increased vascular permeability, and blood-retina barrier disruption occur through downstream mediators including vascular endothelial growth factor (VEGF), various cytokines, and prostaglandins (Figure 1). As a result, fluid leaks into the retina and CME develops.8-10
PREVENTING CME
Various therapies have been investigated to prevent the development of CME after cataract surgery. Most important among these are antiinflammatory drugs, such as NSAIDs and corticosteroids, and drugs that directly target mediators in the above-mentioned pathophysiologic pathway, such as the anti-VEGF drugs bevacizumab and ranibizumab.
Postoperative treatment with topical NSAIDs, corticosteroids, or a combination, is typically used in patients undergoing cataract surgery. A recent systematic review by Kessel et al11 compared the efficacy of topical NSAIDs versus corticosteroids in nondiabetic patients at 1 month postoperatively. They found that CME occurred in 25.3% of patients receiving only a topical corticosteroid and in 3.8% of patients receiving only a topical NSAID (risk ratio [RR] 5.35). High-potency corticosteroids (eg, betamethasone and dexamethasone) were more effective than less potent ones (eg, fluorometholone), but they were nonetheless not as effective as NSAIDs (RR, 4.77 and 5.84 for high- and low-potency, respectively). It should be noted that this meta-analysis included six randomized controlled trials (RCTs)— four from the same Japanese research group and two from other Asian groups.
No previous studies have compared the efficacy of topical NSAIDs to topical corticosteroids in diabetic patients. One study, however, found that CME occurred 30 days postoperatively in only 2.4% of diabetic patients treated with a combination treatment of topical NSAIDs and corticosteroids and in 8.7% treated with only a corticosteroid.12 It is currently unknown whether topical corticosteroids have an additive effect to topical NSAIDs in preventing CME after cataract surgery in nondiabetic or diabetic patients. Only one study compared an NSAID with combination treatment, but no patient in either group developed CME.13
At a Glance
• Eighteen European study centers are participating in PREMED, an international multicenter study that aims to provide conclusive evidence for prevention of CME after cataract surgery in both diabetic and nondiabetic patients.
• The primary endpoint is the change in central subfield mean macular thickness on OCT at 6 weeks postoperative.
Due to the limited amount of evidence, it is unknown which NSAID is most effective in preventing CME after cataract surgery. Only small studies have compared the use of different NSAIDs after cataract surgery, and in most of these studies topical corticosteroids were also used in all patients.
It has been recommended to start prophylactic application of NSAIDs up to 3 days preoperatively, based on a study by Yavas et al.14 Their study found that CME occurred in 15% of patients who started using an NSAID postoperatively, but no CME occurred in a group of patients who started the NSAID 3 days preoperatively.
Because diabetic patients have a significantly increased risk of developing CME after cataract surgery, additional prophylactic treatments have been studied in this population. Typically they involve intravitreal injection of a corticosteroid or anti-VEGF drug at the end of cataract surgery. Ahmadabadi et al compared a combination of topical and intravitreal corticosteroids to topical corticosteroids alone and found that the combination treatment was more effective in preventing CME at 1 month postoperatively, albeit the difference was not statistically significant (incidence: 0% vs 19%, P=.059).15 Similarly, Udaondo et al compared a combination of intravitreal anti-VEGF agent and topical corticosteroid to topical corticosteroid alone and found a nonsignificant effect in favor of the combination treatment, with incidences of CME of 3.7% and 25.92% with and without anti-VEGF therapy, respectively.16 The nonsignificant findings in these studies are likely related to the small number of patients included.
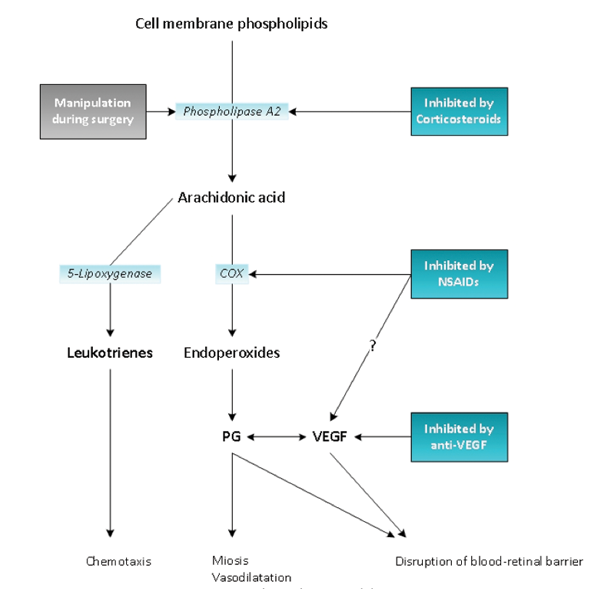
Figure 1. Pathogenesis of cystoid macular edema after cataract surgery. COX: cyclooxygenase; PG: prostaglandin; VEGF: vascular endothelial growth factor
In diabetic patients with diabetic macular edema preoperatively, it has been shown that intravitreal bevacizumab at the end of cataract surgery led to a significant reduction in retinal thickness and macular volume increase on OCT.17-21
ESCRS PREMED STUDY
The literature provides no firm evidence, in the form of well-designed RCTs, for the most appropriate prophylactic treatment against CME after cataract surgery. For this reason, the European Society of Cataract and Refractive Surgeons (ESCRS) PREvention of Macular EDema after cataract surgery (PREMED) study was initiated.
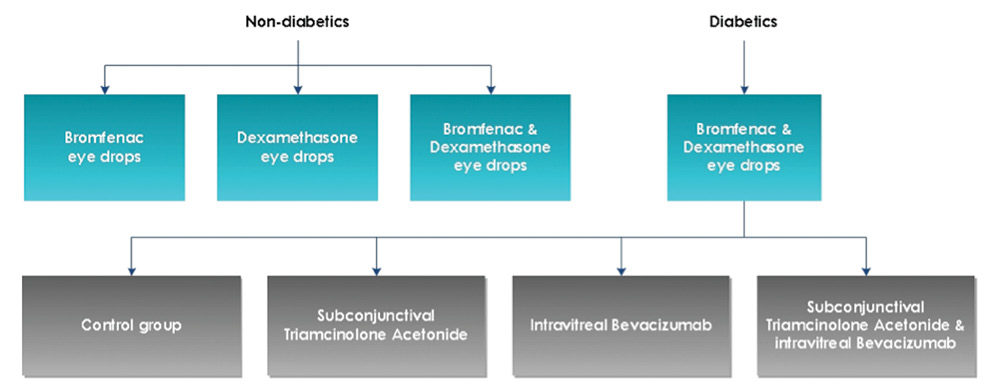
Figure 2. Design of the ESCRS PREMED study.
The ESCRS PREMED study is an initiative of the University Eye Clinic Maastricht UMC, in the Netherlands, under the supervision of Rudy M.M.A. Nuijts, MD, PhD. Eighteen European study centers are participating in this international multicenter study that aims to provide conclusive evidence for prevention of CME after cataract surgery in both diabetic and nondiabetic patients.
Nondiabetic patients will be treated postoperatively with topical bromfenac twice daily, dexamethasone four times daily, or a combination of both eye drops (Figure 2). Topical treatment starts 2 days preoperatively, as suggested by Yavas et al.14 Diabetic patients—who have a higher risk of developing CME after cataract surgery—will receive both bromfenac and dexamethasone eye drops. Further, these patients will be randomized to a control group receiving no additional treatments, or to a subconjunctival injection of triamcinolone acetonide, an intravitreal injection of bevacizumab, or both.
The primary endpoint is the change in central subfield mean macular thickness on OCT at 6 weeks postoperative. Recruitment for this study started in July 2013, and final results are expected in 2016.
1. Eriksson U, Alm A, Bjärnhall G, et al. Macular edema and visual outcome following cataract surgery in patients with diabetic retinopathy and controls. Graefes Arch Clin Exp Ophthalmol. 2011;249(3):349-359.
2. Katsimpris JM, Petropoulos IK, Zoukas G, et al. Central foveal thickness before and after cataract surgery in normal and in diabetic patients without retinopathy. Klin Monbl Augenheilkd. 2012;229(4):331-337.
3. Mentes J, Erakgun T, Afrashi F, Kerci G. Incidence of cystoid macular edema after uncomplicated phacoemulsification. Ophthalmologica. 2003;217(6):408-412.
4. Rossetti L, Chaudhuri J, Dickersin K. Medical prophylaxis and treatment of cystoid macular edema after cataract surgery. The results of a meta-analysis. Ophthalmology. 1998;105(3):397-405.
5. Krepler K, Biowski R, Schrey S, et al. Cataract surgery in patients with diabetic retinopathy: visual outcome, progression of diabetic retinopathy, and incidence of diabetic macular oedema. Graefes Arch Clin Exp Ophthalmol. 2002;240(9):735-738.
6. Dowler JG, Sehmi KS, Hykin PG, Hamilton AM. The natural history of macular edema after cataract surgery in diabetes. Ophthalmology. 1999;106(4):663-668.
7. Diabetic Retinopathy Clinical Research Network Authors/Writing Committee. Macular edema after cataract surgery in eyes without preoperative central-involved diabetic macular edema. JAMA Ophthalmol. 2013;131(7):870-879.
8. Lobo C. Pseudophakic cystoid macular edema. Ophthalmologica. 2012;227(2):61-67.
9. Colin J. The role of NSAIDs in the management of postoperative ophthalmic inflammation. Drugs. 2007;67(9):1291-1308.
10. Yonekawa Y, Kim IK. Pseudophakic cystoid macular edema. Curr Opin Ophthalmol. 2012;23(1):26-32.
11. Kessel L, Tendal B, Jørgensen KJ, et al. Post-cataract prevention of inflammation and macular edema by steroid and nonsteroidal anti-inflammatory eye drops: a systematic review. Ophthalmology. 2014;121(10):1915-1924.
12. Singh R, Alpern L, Jaffe GJ, et al. Evaluation of nepafenac in prevention of macular edema following cataract surgery in patients with diabetic retinopathy. Clin Ophthalmol. 2012;6:1259-1269.
13. Miyanaga M, Miyai T, Nejima R, et al. Effect of bromfenac ophthalmic solution on ocular inflammation following cataract surgery. Acta Ophthalmol. 2009;87(3):300-305.
14. Yavas GF, Ozturk F, Kusbeci T. Preoperative topical indomethacin to prevent pseudophakic cystoid macular edema. J Cataract Refract Surg. 2007;33(5):804-807.
15. Ahmadabadi HF, Mohammadi M, Beheshtnejad H, Mirshahi A. Effect of intravitreal triamcinolone acetonide injection on central macular thickness in diabetic patients having phacoemulsification. J Cataract Refract Surg. 2010;36(6):917-922.
16. Udaondo P, Garcia-Pous M, Garcia-Delpech S, et al. Prophylaxis of macular edema with intravitreal ranibizumab in patients with diabetic retinopathy after cataract surgery: a pilot study. J Ophthalmol. 2011;2011:159436.
17. Takamura Y, Kubo E, Akagi Y. Analysis of the effect of intravitreal bevacizumab injection on diabetic macular edema after cataract surgery. Ophthalmology. 2009;116(6):1151-1157.
18. Chen CH, Liu YC, Wu PC. The combination of intravitreal bevacizumab and phacoemulsification surgery in patients with cataract and coexisting diabetic macular edema. J Ocul Pharmacol Ther. 2009;25(1):83-89.
19. Akinci A, Batman C, Ozkilic E, Altinsoy A. Phacoemulsification with intravitreal bevacizumab injection in diabetic patients with macular edema and cataract. Retina. 2009;29(10):1432-1435.
20. Lanzagorta-Aresti A, Palacios-Pozo E, Menezo Rozalen JL, Navea-Tejerina A. Prevention of vision loss after cataract surgery in diabetic macular edema with intravitreal bevacizumab: a pilot study. Retina. 2009;29(4):530-535.
21. Cheema RA, Al-Mubarak MM, Amin YM, Cheema MA. Role of combined cataract surgery and intravitreal bevacizumab injection in preventing progression of diabetic retinopathy: prospective randomized study. J Cataract Refract Surg. 2009;35(1):18-25.
Rudy M.M.A. Nuijts, MD, PhD
• University Eye Clinic Maastricht, Maastricht University Medical Center, Maastricht, Netherlands
• Atrium Medical Center Parkstad, Heerlen, Netherlands
• Member, CRST Europe Editorial Board
• rudy.nuijts@mumc.nl
• Financial disclosure: None
Jan S.A.G. Schouten, MD, PhD
• University Eye Clinic, Maastricht University Medical Center, Maastricht, Netherlands
• Atrium Medical Center Parkstad, Heerlen, Netherlands
• Financial disclosure: None
Rob W.P. Simons, BSc
• University Eye Clinic Maastricht, Maastricht University Medical Center, Maastricht, Netherlands
• rob.simons@mumc.nl
• Financial disclosure: None
Frank J.H.M. van den Biggelaar, PhD
• University Eye Clinic Maastricht, Maastricht University Medical Center, Maastricht, Netherlands
• Financial disclosure: None
Laura H.P. Wielders, MD, PhD student
• University Eye Clinic Maastricht, Maastricht University Medical Center, Maastricht, Netherlands
• laura.wielders@mumc.nl
• Financial disclosure: None
Bjorn Winkens, PhD
• Department of Methodology and Statistics, Maastricht University, Maastricht, Netherlands
• Financial disclosure: None
