A 29-year-old man presented with decreased vision 6 months after sustaining an injury to the right eye. The patient stated that he had begun treatment for pain and decreased vision immediately after the injury. He reported that pain management was successful but that his vision continued to worsen.
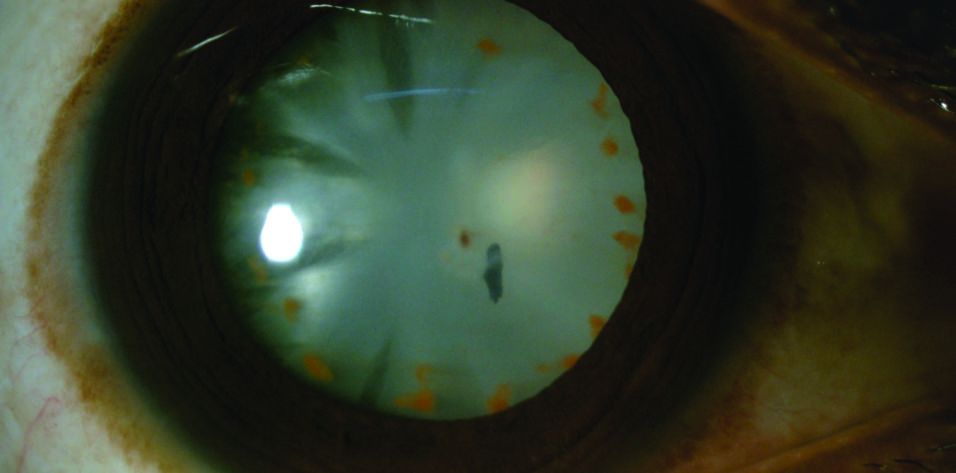
Upon examination, the patient’s BCVA was 3/60 OD and 6/6 OS. His IOP was within normal limits. A slit-lamp examination of the right eye showed a central, small, self-sealed, full-thickness corneal entry wound, an intralenticular iron foreign body, and multiple midperipheral brown deposits on a cataractous lens, which was suggestive of siderosis bulbi (Figure). There was no active inflammation. Gonioscopy showed open angles. Fundus examination findings were normal. No additional foreign bodies were found on a B-scan ultrasound and computed tomography of the orbit.
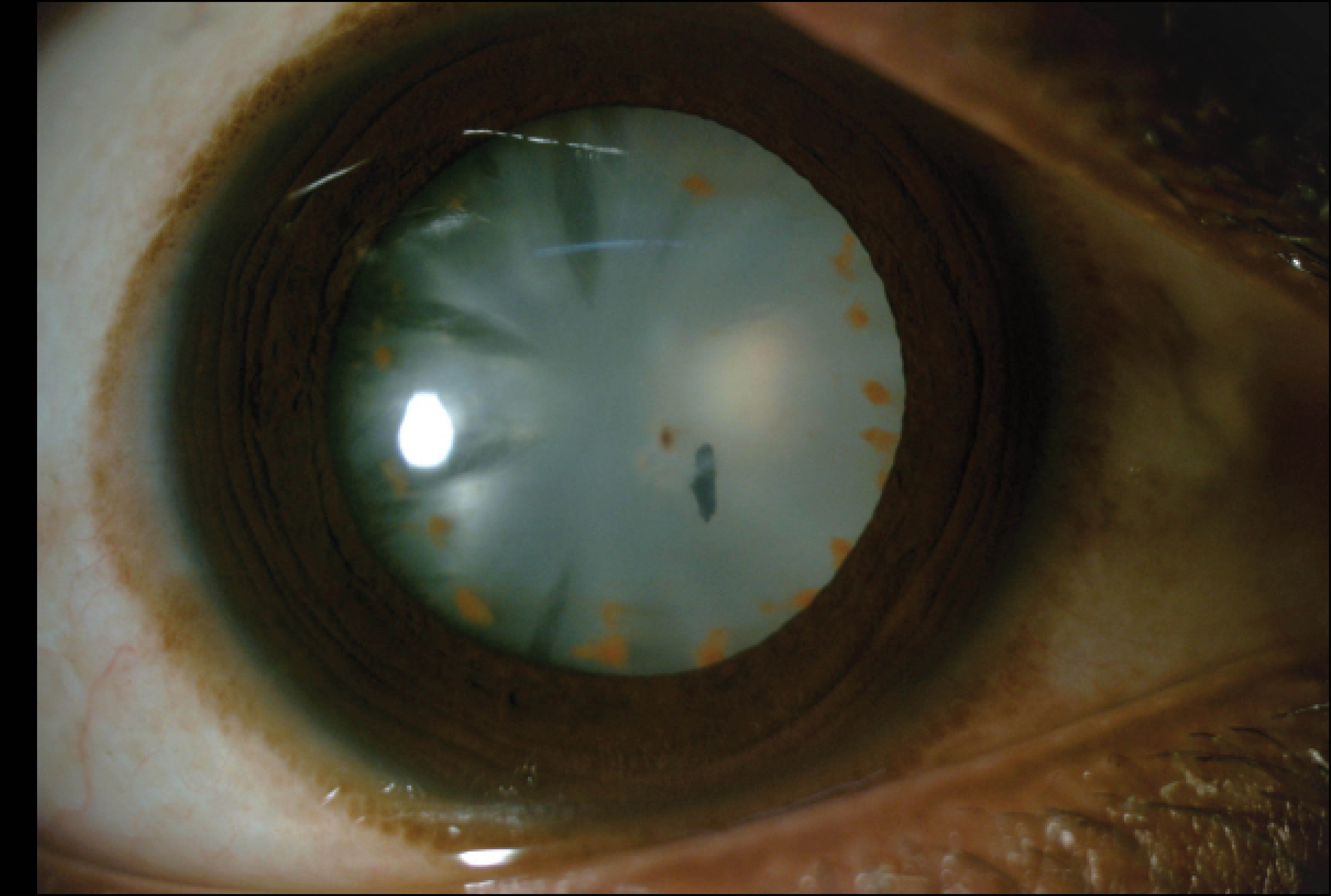
Figure. A slit-lamp image shows an intralenticular iron foreign body and multiple midperipheral brown deposits.
Cataract surgery was performed. During the capsulorhexis, care was taken to include the point of entry within the flap. Phacoaspiration was performed because the cataract was soft. The posterior lens capsule was intact. A hydrophobic acrylic IOL was placed.
The postoperative drug regimen included a 6-week tapering dose of topical prednisolone acetate drops, topical moxifloxacin administered four times daily for 2 weeks, and homatropine drops administered twice daily for 2 weeks. One month after surgery, the patient’s BCVA was 6/9, and there were no signs of inflammation.
Discussion
Siderosis bulbi is caused by the retention and oxidation of an intraocular foreign body that contains iron. Clinical features include iris heterochromia, cataract, glaucoma, and retinal degeneration.1,2 Three phases have been described in the development of siderosis bulbi: (1) an initial latent period after injury, (2) the intraocular spread of iron, and (3) tissue degeneration.3 The structural damage associated with siderosis bulbi is believed to be caused by the deposition of ferritin throughout the cytoplasm in the form of siderosomes. Additionally, hydroxyl radical formation and oxidative stress caused by iron deposition can lead to retinal toxicity.3,4
Immediate removal of an iron foreign body is recommended if it is localized in the anterior chamber. An intralenticular foreign body should be removed at the time of cataract surgery because iron particles may leak out.4
1. Zhu L, Shen P, Lu H, Du C, Shen J, Gu Y. Ocular trauma score in siderosis bulbi with retained intraocular foreign body. Medicine (Baltimore). 2015;94(39):e1533.
2. Loporchio D, Mukkamala L, Gorukanti K, Zarbin M, Langer P, Bhagat N. Intraocular foreign bodies: a review. Surv Ophthalmol. 2016;61(5):582-596.
3. Casini G, Sartini F, Loiudice P, Benini G, Menchini M. Ocular siderosis: a misdiagnosed cause of visual loss due to ferrous intraocular foreign bodies-epidemiology, pathogenesis, clinical signs, imaging and available treatment options. Doc Ophthalmol. 2021;142(2):133-152.
4. Chao HM, Chen YH, Liu JH, et al. Iron-generated hydroxyl radicals kill retinal cells in vivo: effect of ferulic acid. Hum Exp Toxicol. 2008;27:327-339.