Accuracy of Astigmatic Outcome
By Michael Goggin, FRCSI(Ophth), FRANZCO, MS

Almost all my decisions regarding choice of toric IOL are based on achieving the greatest accuracy of astigmatic outcome. Lens material, haptic design, and other factors are also important, of course, but they do not have a direct bearing on refractive outcome. My go-to choice for toric IOLs is the range of monofocal and trifocal toric lenses from Carl Zeiss Meditec (although I choose lenses from other manufacturers in some specific instances). In this article I discuss some of the factors that influence my decision-making.
INCISION SIZE AND SIA
Calculation of corneal surgically induced astigmatism (SIA) is inaccurate because of the variability of measurement of small differences between preoperative and postoperative corneal astigmatism, in terms of both axis and power, the source data for calculation of SIA.1 Applying a mean SIA for a population in individual cases, consequently, has the potential for error and is best avoided, in my opinion.
Use of this parameter in toric IOL calculation can be avoided by use of astigmatically neutral incisions, which can be achieved by reducing the length of clear corneal incisions in the temporal cornea to less than 2 mm.2 This means that the toric IOL I choose must pass through such incisions. This largely, though not exclusively, reduces my choices to hydrophilic acrylic lenses introduced in an incision-assisted manner.
ACCURATE TORIC CALCULATOR
In the past 1 to 2 years, most IOL manufacturers have modified their online toric IOL calculators to improve outcome prediction. This required the inclusion particularly of the sphere power of the IOL, which alters the cylinder power requirement for any given corneal astigmatic error.3,4 Inclusion of more accurate effective lens position prediction, usually by incorporation of preoperative anterior chamber depth measurement, has also become more common. The Zeiss online calculator has had these features since its introduction many years ago, and they were included in the calculator’s upgrade in 2018.
LOW CORNEAL ASTIGMATISM
Astigmatism of 0.75 D or greater reduces unaided visual acuity. Postoperative refractive astigmatism should be minimized if optimal refractive outcome and spectacle independence are our targets.
A target of spectacle independence should not be just for the refractive cataract patient seeking an excellent outcome from a trifocal IOL. It is also appropriate for those with a lack of access to optometric services, for example in developing nations. Likewise, it can benefit those with a comorbidity limiting mobility and communication, such as severe Parkinson disease or dementia, for whom choosing and putting on spectacles may be challenging.
Incidentally, it is a shame none of the major IOL manufacturers has addressed the lack of toric IOLs available in the developing world. Astigmatic control is possible with small-incision extracapsular cataract surgery. With the precise astigmatic control available with phacoemulsification, elimination of visually significant astigmatism is both possible and desirable.
Surgeons should not be afraid of overcorrection of astigmatism (or axis flip) with toric IOLs if the predicted and achieved astigmatism is lower than that achievable with either a spherical IOL or an under-correcting toric IOL.5 This, of course, applies to all toric IOL astigmatism correction, but it seems to be of particular concern to some surgeons when considering the appropriate corneal astigmatism threshold for toric IOL use.
POSTERIOR CORNEAL ASTIGMATISM
The inclusion of posterior corneal astigmatism in toric IOL calculations has become essential to increase the refractive accuracy of toric IOL outcomes.6 Accurate measurement of this entity in individual eyes has been elusive. Its effect, however, can be inferred from population average outcomes, and such data can be included in these calculations to increase the accuracy of outcomes.7
I have introduced a method to include this information in toric IOL calculation that produces a new set of keratometric data from those measured on the anterior cornea.8 It is available at goggintoric.com. The new data can be used in any accurate toric calculator, as long as no routine adjustment for posterior corneal astigmatism is already incorporated in that calculator. I do not adjust for posterior corneal astigmatism if the IOL as calculated on the basis of anterior corneal astigmatism has 2.50 D of cylinder power or greater, based on my observation of outcomes in such cases.9
CYLINDER POWER STEPS AND RANGE
The Zeiss range of monofocal and multifocal toric IOLs is available in cylinder power steps of 0.50 D. This self-evidently enhances the routine accuracy of outcomes over manufacturers that provide 0.75-D steps. Furthermore, the company provides routine access to a wide range of lens cylinder powers (1.00 D to 12.00 D), although it requires individual manufacturing orders for extremely high powers.
EXTREME TORIC POWERS
For eyes that require sphere or cylinder powers outside the range provided by Zeiss, my go-to manufacturers are two German companies, HumanOptics and Oculentis. Both of these manufacturers have a wider range of IOLs of more extreme powers that can be individually manufactured for individual eyes and can be inserted through microincisions.
1. Goggin M, Patel I, Billing K, Esterman A. Variation in surgically induced astigmatism estimation due to test-to-test variations in keratometry. J Cataract Refract Surg. 2010;36(10):1792-1793.
2. Goggin M, Moore S, Zamora-Alejo K, Esterman A. Astigmatic neutrality in 1.9-mm coaxial microincision phacoemulsification. J Refract Surg. 2012;28(7):451-452.
3. Goggin M, Moore S, Esterman A. Outcome of toric intraocular lens implantation after adjusting for anterior chamber depth and intraocular lens sphere equivalent power effects. Arch Ophthalmol. 2011;129(8):998-1003.
4. Goggin M, Moore S, Esterman A. Toric intraocular lens outcome using the manufacturer’s prediction of corneal plane equivalent intraocular lens cylinder power. Arch Ophthalmol. 2011;129(8):1004-1008.
5. Beheregaray S, Goggin M, LaHood B. Astigmatic overcorrection and axis flip for targeting minimal remaining refractive astigmatism with toric intraocular lenses. J Cataract Refract Surg. 2018;44:109-110.
6. Koch DD, Jenkins RB, Weikert MP, Yeu E, Wang L. Correcting astigmatism with toric intraocular lenses: effect of posterior corneal astigmatism. J Cataract Refract Surg. 2013;39:1803-1809.
7. Goggin M, van Zyl L, Caputo S, Esterman A. Outcome of adjustment for posterior corneal curvature in toric intraocular lens calculation and selection. J Cataract Refract Surg. 2016;42:1441-1448.
8. Goggin M, Zamora-Alejo K, Esterman A, van Zyl L. Adjustment of anterior corneal astigmatism values to incorporate the likely effect of posterior corneal curvature for toric intraocular lens calculation. J Refract Surg. 2015;31:98-102.
9. LaHood BR, Goggin M, Esterman A. Assessing the likely effect of posterior corneal curvature on toric IOL calculation for IOLs of 2.50 D or greater cylinder power. J Refract Surg. 2017;33:730-734.
Keeping Up With Changing Technologies
By Detlef Holland, MD

Astigmatism is a common refractive error that increases with patient age. Approximately 30% of people older than 40 years have astigmatism of greater than 1.00 D, and this is true in patients presenting for cataract surgery as well.
For any patient with astigmatism greater than 0.75 D, we consider the implantation of toric IOLs. Additional corneal pathologies are not immediate criteria for exclusion. For instance, if early Fuchs endothelial dysplasia is diagnosed, a sulcus-fixed additive toric lens may be suitable.
We have used a number of toric IOL models, and our preferences have evolved over the years to take advantage of the wide range of technologies available in Europe. For years we have been using the AddOn Toric lens (1stQ Deutschland), a supplementary sulcus-fixated lens. This hydrophilic IOL has four haptics that provide high rotational stability and a square-edged optic that reduces the possibility of iris capture.
The standard toric monofocal IOL that we routinely use is the AT Torbi, which has a plate-haptic design and a bitoric optic. This IOL can be implanted easily through a limbal incision of 1.8 mm for astigmatism-neutral surgery, and it provides a high degree of rotational stability. During a period of 3 years, we observed postoperative rotation of this lens in only one patient with high myopia.
Another toric monofocal lens we have used more often recently is the Precizon Toric (model 565, Ophtec), which employs an innovative toric optic design. The transitional conic surface of the lens provides refractive resilience in the event of malpositioning. The transitional conic surface blends into the aspheric surface in all meridians, leading to a broader toric surface that makes the lens more tolerant to rotation and misalignment. In an ongoing study with this lens, our early results show good outcomes, comparable to those of other studies, and the lens exhibits tolerance to misalignment.
This year, in our laser cataract surgery cases, we will begin to use the toric version of the Femtis IOL (Oculentis), a lens that is held in place by four additional haptics that enclave the capsulotomy (see The Lens: Femtis Laser Lens, CRST Europe January 2017). In a multicenter study in which we participated, the Femtis showed excellent rotational stability, with less than 1° rotation in the first 6 months postoperative. In combination with the cyclotorsion control and the Intelliaxis marking system of the Lensar Laser System (Lensar), this lens should achieve good functional results for astigmatic correction in cataract surgery.
The Importance of Calculation
By Gilles Lesieur, MD

To achieve positive outcomes with monofocal and multifocal IOLs, it is not sufficient to address only spherical error. Patients now also expect correction of astigmatism to improve their quality of vision. Careful preparation, accurate surgery, and new technologies can help us achieve these desired results.
A number of steps are essential for success with toric IOLs:
- Analyze and calculate total astigmatism;
- Decide what amount of astigmatism you need to correct;
- Eliminate SIA;
- Place the IOL in the right position; and
- Ensure that the lens doesn’t rotate after surgery.
CALCULATE TOTAL ASTIGMATISM
Calculation of total astigmatism is the essential first step in preoperative analysis. Anterior corneal shape alone cannot predict the total power of astigmatism and the real axis of astigmatism. Normal anterior corneal astigmatism is +0.50 to +0.75 D at 90°, with the rule, balanced by against-the-rule posterior corneal astigmatism of -0.3 D at 90° (0.26 at 0.78 D).
The Gullstrand model predicts a constant relationship between anterior and posterior steep meridian, but Koch et al have shown that anterior corneal axis can be considerably different from posterior corneal axis in astigmatic patients, resulting in a different total corneal astigmatism.1
Scheimpflug imaging can provide total corneal astigmatism, but there can be imperfections due to instability of the corneal surface if meibomian gland dysfunction is present. The Barrett nomogram can provide good results with its algorithmic calculation of the posterior cornea, but it does not address outliers. Calculation of total keratometry (TK) is now available with the IOLMaster 700 (Carl Zeiss Meditec), and early data show promising outcomes. For the best results, treat any meibomian gland dysfunction and perform repeat measurements with three devices to be certain of the final TK.
DECIDE WHAT TO CORRECT
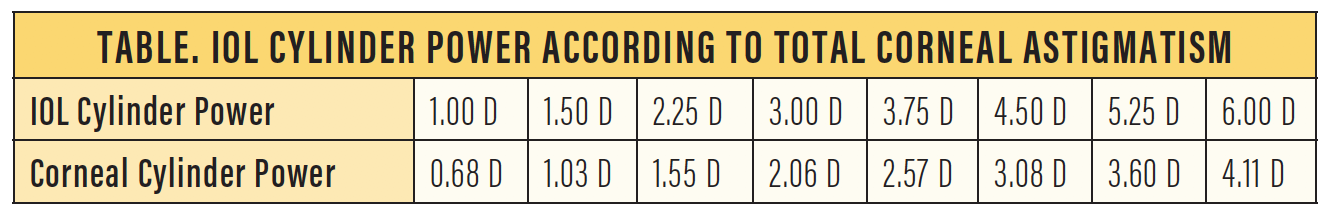
Anterior astigmatism decreases with age while posterior astigmatism remains stable. Therefore, for patients younger than 70 years, it is important to have a target of +0.25 D to +0.50 D at 90°. For patients older than 70, the target should be 0.0 D. You can use an online calculator with total astigmatism included (whether by direct measure or use of an algorithm) and adjust the results with the help of a nomogram like the one shown in the Table.
MINIMIZE SIA
Residual astigmatism diminishes the effect of a toric IOL, so it is essential to minimize or eliminate SIA. Surgeons have two options to achieve this: The first is to practice microincision cataract surgery (MICS), using an incision of less than 2 mm, and the second is to use a slightly larger incision (generally 2.2 to 2.4 mm) and include the resulting SIA in the lens power calculations.
In my experience, it is better to perform MICS. In a personal study of 40 toric IOL implantations using bimanual MICS, the final SIA was -0.05 D AT 85°, and patients achieved an average outcome of -0.04 D at 96° OD and -0.08 D at 79° OS.
ENSURE PROPER POSITIONING
To avoid mistakes in axis location due to cyclorotation when the patient is lying flat, the eye can be marked before surgery. Use of marking instruments is effective but time-consuming. Considering that up to 80% of cataract surgery cases have some degree of astigmatism, it is not possible to achieve an efficient workflow using these methods. Image-guided technology is therefore more appropriate. A printout from the IOLMaster 700 can be used to identify the 180° axis.
The capsulorhexis should be circular and covering the optic edge. In the future, it may be possible to fixate the IOL directly in the rhexis, as with the Femtis lens (Oculentis).
It is also vital to remove all OVD from the capsular bag and from behind the toric IOL to ensure postoperative stability.
AVOID POSTOPERATIVE ROTATION
I performed an analysis of 428 toric IOLs I implanted in 2016 (AT Torbi 709 M/MP). Overall, results were excellent, with 62.7% of eyes achieving 0.00 D of postoperative astigmatism and 92.6% achieving ±0.50 D (unpublished data; Figure).
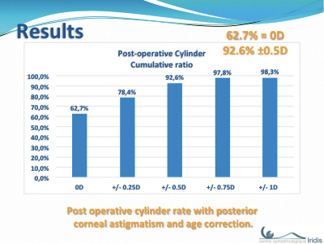
Figure: Postoperative cylinder with posterior corneal astigmatism and age correction.
In cases in which unexpected lens rotation occurred, these factors were noted:
- Large corneal diameter (greater than 12 mm);
- Vertical (ie, with-the-rule) astigmatism;
- No preponderance of myopic versus hyperopic eyes.
In an effort to understand the reasons for my findings, I came across a study assessing lens diameter and other ocular dimensions using high-resolution MRI imaging.2 The conclusions were as follows:
- Capsular bag size increases with corneal diameter (CD);
- Horizontal CD is greater than vertical CD; and
- Vertical capsular bag diameter is greater than horizontal.
Some companies have designed low-power IOLs with greater overall diameter and high-power lenses with smaller overall diameter, based on the conventional idea that long eyes will have a large capsular bag. Experience suggests that this is not always true.
Adapting the overall diameter of toric IOLs to the CD is a novel concept. If lens sizing were to be made available, it might be preferable to implant, for example, a toric IOL with an 11-mm overall diameter in eyes with a CD of 12 mm or less, and use a lens with greater overall diameter (11.4 mm or more) when the CD is greater than 12 mm. It would be important, however, to ensure that the larger diameter lenses could still be used in MICS, in order to avoid SIA. In addition, it would be interesting to include the lens thickness in the calculation of the possible capsular bag size.
1. Koch DD, Jenkins RB, Weikert MP, Yeu E, Wang L. Correcting astigmatism with toric intraocular lenses: effect of posterior corneal astigmatism. J Cataract Refract Surg. 2013;39(12):1803-1809.
2. Erb-Eigner K, Hirnschall N, Hacki C, Schmidt C, Asbach P, Findl O. Predicting lens diameter: ocular biometry with high-resolution MRI. Invest Ophthalmol Vis Sci. 2015;56(11):6847-6854.
Establishing a Well-Defined Procedure
Edoardo Ligabue, MD

I started using toric IOLs in 2005 and have gradually increased the percentage of these lenses that I have implanted over the years. My philosophy is simple: I always try to completely correct the refractive defect of the patient with regard to sphere and cylinder exactly as if I had to prescribe a pair of glasses. I perform around 1,000 laser cataract surgery procedures per year, and, because of the positive outcomes I’ve observed, over the past 5 years I have implanted a toric IOL in about 76% of these cases.
Today it is normal routine to calculate the ideal lens for each patient in order to reduce as much astigmatism as possible. Correcting astigmatism using a toric IOL provides better visual quality than glasses or contact lenses, and the improvement is more evident when brightness decreases in the evening and at night. A residual 0.50 D of astigmatism can generate the loss of 1 line of visual acuity, and for this reason I insist on always looking for the best possible option for astigmatism correction.
I correct astigmatism even if the residual refractive target is not emmetropia, for example if the patient wishes to remain 2.50 D myopic in order to read without glasses. In fact, near vision is much better if there is no residual astigmatism.
In the case of multifocal IOL implantation, the correction of astigmatism is absolutely essential. The presence of two or three foci for distance, intermediate, and reading accentuates any small visual interference, reducing visual quality.
Making the use of toric IOLs routine requires a well-defined procedure that I apply in my daily practice for all cataract surgery candidates. The procedure includes the following:
- Evaluation of tear film quality because it can affect IOL power calculation. If necessary, I perform preoperative therapy to improve it;
- Performing biometry and topography with two different devices such as the IOLMaster 700, Pentacam AXL (Oculus), or Sirius (CSO);
- Calculating the value and orientation of toric IOLs using a latest-generation formula that also considers posterior corneal curvature (eg, the Barrett formula or the Alcon toric IOL calculator);
- Choosing the value of toric IOL that leaves the least possible astigmatism, always with the rule. This choice is made to counteract the fact that, with aging, the cornea will tend toward against-the-rule astigmatism.
- Use of the Verion Image Guided System (Alcon), paying great attention to correct image acquisition;
- Performing all surgeries with a 2.2-mm temporal incision at 0° to 180° and a calculated SIA of about 0.10 D;
- Use of the LenSx femtosecond laser (Alcon) to guarantee a regular and perfectly centered capsulotomy in order to position the toric IOL in the best possible way;
During surgery a toric lens implant requires about 30 seconds more surgical time than other lens types. To simplify the procedure, I have a complete inventory of toric IOLs in order to always have the lens available and ready to be implanted without need to preorder the lens from the manufacturer. I always check the postoperative result after 7 days by performing aberrometry using OPD-Scan III (Nidek) to verify the correct alignment of the lens.
The Approach is More Important Than the Brand
By Majid Moshirfar, MD

For astigmatism correction at cataract surgery, I most often implant Tecnis toric IOLs (Johnson & Johnson Vision), but toric IOL models from Alcon and Bausch + Lomb are also excellent, and I do not recommend one platform over another. Most data available show similar outcomes with all of these IOL platforms regarding visual outcome, reduction of astigmatism, and rotational stability.
More important, then, is the approach to toric IOL implantation rather than the IOL brand itself. At a minimum, I measure tomography twice, with the Pentacam and Galilei G4 (Ziemer), in addition to assessing angle kappa and other typical IOL measurements with the Lenstar (Haag-Streit). All measurements are then repeated once more. If appropriate, a third set of measurements is taken, for example in patients who have had radial keratotomy. Appropriate adjustments are made for previous refractive surgery using the ASCRS calculators.
Immediately prior to surgery, with the patient sitting upright, the eye is marked at four cardinal directions rather than just two. Intraoperatively, precise incisions with predictable effects on cylinder are created. A small capsulorhexis (no larger than 5.0 mm) with 360° optic edge coverage is used to increase rotational stability. OVD is scrupulously removed at the end of surgery, especially from behind the IOL, which is accomplished by gently pressing posteriorly on the lens. The IOL is centered with respect to fixation and compared with angle kappa as measured previously. I believe centration carries added importance when implanting a toric multifocal lens.
Most patients have very good outcomes after toric IOL implantation with this procedure. In my experience, a low percentage of IOLs requires postoperative adjustment. For the few cases in which adjustment is needed, such as when there is a refractive surprise or an IOL rotates, I typically exchange the lens or rotate it. I also consider a corneal refractive procedure for appropriate candidates.
The future holds promise for improvements in toric IOLs. Initial reports of light-adjustable lenses show that they may outperform current platforms, after some temporary quality-of-life sacrifice due to the need for UV-light protection prior to lock-in. Femtosecond lasers may someday be able to etch toricity into existing IOLs. Optimistically, it may follow that correction of higher-order aberrations or surface irregularities could be addressed with postoperative IOL adjustments, just as surface-guided ablations do.
Occasionally a patient with a high level of regular astigmatism, well above what is correctable with current toric IOLs, seeks vision correction. Toric IOL placement may be followed by limbal relaxing incisions, toric small-incision lenticule extraction, or LASIK in such cases.
Finally, many of my colleagues prefer to correct corneal astigmatism with a corneal procedure rather than a toric IOL. Truly, it may be more accurate to implant a monofocal IOL, aim for slight myopia, and follow with a corneal refractive surgical procedure for ultimate correction later. n



