
In humans, iris pigmentation ranges from light brown to black, and, depending on how light scatters in the iris stroma through the Tyndall effect, the iris can appear blue, green, or hazel. Although the genetics of eye color is extremely complex, in the future it may be possible to permanently alter a person’s natural iris color in less than 1 minute with a noninvasive laser treatment designed to remove pigment through the vasculature.
AN UNMET NEED
In the aesthetics market, beauty trends often start as temporary solutions and advance to permanent fixes. Two prime examples are toupees and push-up bras, temporary solutions that were followed by the permanent surgical solutions of hair transplants and breast implants, respectively. In my opinion, permanent eye color change could be another popular trend in aesthetics.
Today, more than 26 million people worldwide wear colored contact lenses, and another 75 million have experimented with wearing them. Of those who have ever worn colored contacts, approximately 85% sought a transition from a dark to a light eye color.
Unfortunately, colored contacts have drawbacks. They require daily care, they often look unnatural, and they can cause ocular irritation and symptoms of dry eye. Iris implants provide the opportunity for patients to permanently change their eye color; however, significant complications with the implants have led to explantation in many cases.
Recent experience with a novel laser treatment developed and patented by Strōma Medical has been promising. The company has multiple patents in many countries and has several key patents pending. This noninvasive surgical procedure combines eye mapping, eye tracking, and a frequency-doubled Nd:YAG laser to capture, track, and change the color of the iris safely and effectively.
HOW IT WORKS
Eye color is determined by the pigment in the anterior iris. Iris color—and indeed any color—comprises three elements: hue, saturation, and value. Hue is the combination of primary colors (in this case, the underlying green or blue in the eye), saturation is the distance of the color from white on a spectrum, and value is brightness or light reflectance.
One of the unique features of the Strōma procedure is that it can produce a variety of color appearances, ranging from hazel to dark blue, by removing different amounts of iris pigment (Figure 1). The hue can be predicted and postoperative saturation and value controlled using a separate technology developed by Strōma.
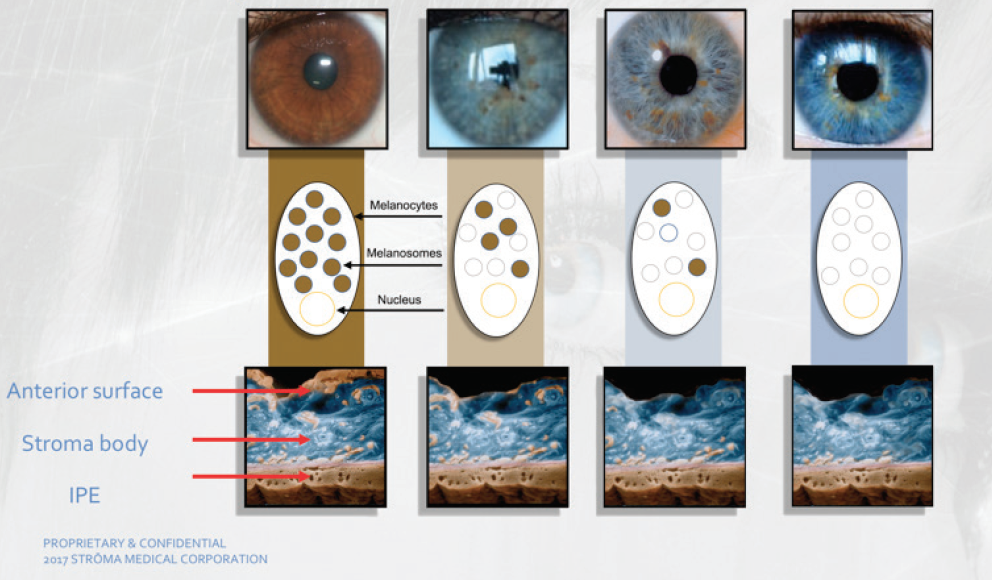
Figure 1. Eye color options available to patients who undergo the Strōma treatment. Abbreviation: IPE, iris pigment epithelium
Images courtesy of Strōma Medical
During the procedure, a frequency-doubled Nd:YAG laser (532-nm wavelength) is used to raise and lower the temperature of the anterior iris pigment a total of 15 times over a period of 30 seconds. The photothermolysis process initiated by the laser treatment stimulates the anterior iris pigment, and cytokine and macrophage responses to the treatment result in pigment removal through the vasculature, revealing the underlying gray stromal fibers of the iris (Figure 2). Depending on the amount of pigment removed, when light shines on the stromal fibers, a shade of blue, green, or hazel is reflected, giving the eye its blue, green, or hazel appearance. The patient leaves the laser suite with the same color irides, as it can take up to 3 weeks for the treatment to take effect.
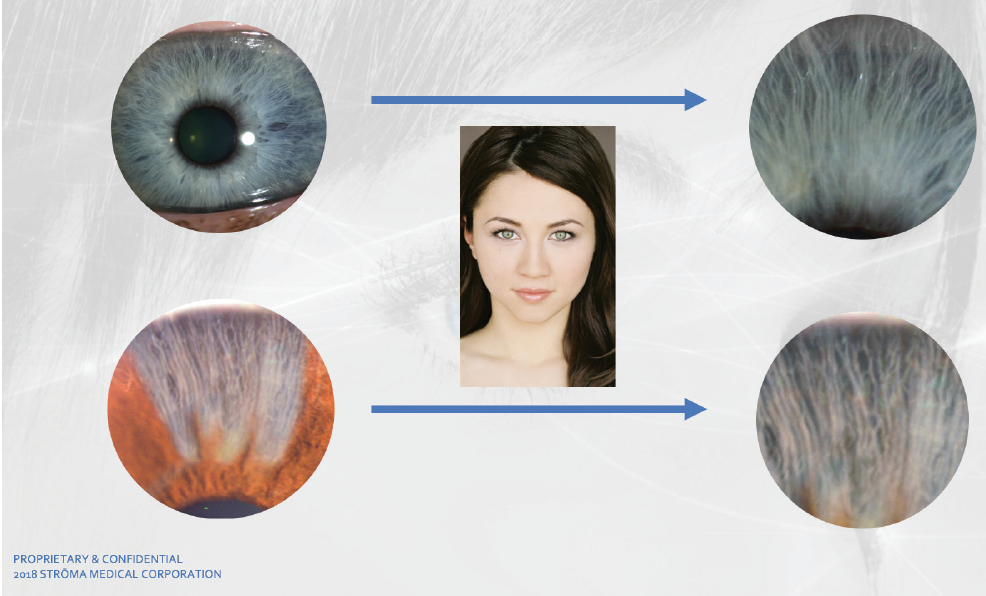
Figure 2. A naturally blue eye depicting the appearance of the gray stromal fibers (top row). Comparative results of pigment removal with the Strōma Laser System (bottom row).
From a safety standpoint, it is important to note that the pigment is carried away by the vascular system; it is not released into the anterior chamber, where it could clog the trabecular meshwork.
EARLY RESULTS
Significant laboratory and animal testing was completed prior to the first-in-human trial of this technology. To date, 10 years of animal data and 6 years of human data have demonstrated the safety and effectiveness of the Strōma procedure.
In human trials, the clinical process involved treating superior (3 to 4 clock hours) segments at first, and then full irides as well as altering the energy applied to the iris in order to minimize adverse events and to help define key parameters of success. As greater energy was introduced into the eye, more iris pigment was released through the vasculature.
During the first-in-human evaluation of the procedure, conducted in November 2018 at two sites in Central America, 14 patients in Costa Rica and 20 patients in Panama received treatment in only a pie-shaped portion of the iris equivalent to 3 or 4 clock hours (Figure 3). A full-iris trial was completed in March, with 30 patients receiving treatment of the whole iris. Initial results of both international trials are promising and will be reported once longer-term follow-up is complete.
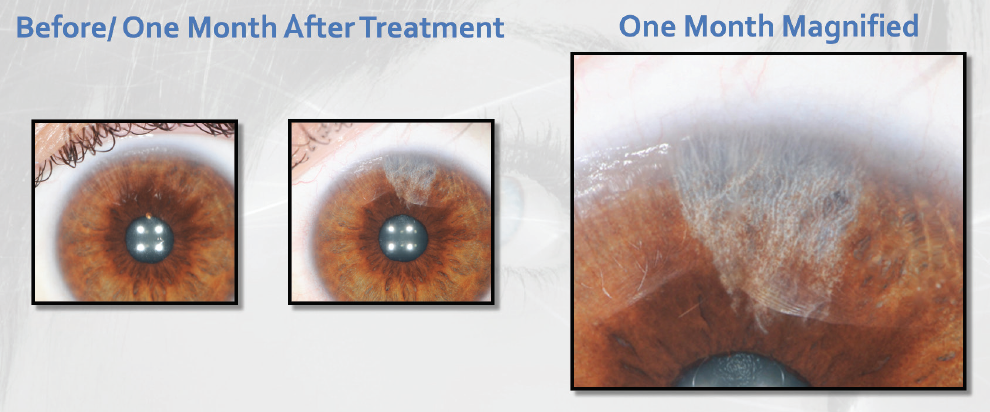
Figure 3. The same eye preoperatively and 1 month after sectoral iris treatment with the Strōma laser.
What can be shared at this time is the excellent safety profile demonstrated in the 64 patients who have been treated so far. Each patient went through rigorous testing, including an external eye examination; dry eye assessment; visual acuity, color vision, and contrast sensitivity testing; retinal health assessment, including assessment for age-related macular degeneration; glaucoma testing, including IOP, iridocorneal angle, and trabecular meshwork pigmentation; OCT imaging; assessment of atrophy and transillumination defects; pupillary health assessment, including size, shape, and reactivity; and measurements of endothelial loss and damage, crystalline lens health, anterior chamber cell and flare, and treatment efficacy, including saturation and hue.
Researchers have noted that, in addition to producing a change in iris color, the Strōma treatment can decrease IOP in treated eyes by a statistically significant amount.
CONCLUSION
Further clinical trials of this technology are planned. In the meantime, there is strong evidence from animal and international human studies to suggest that the Strōma treatment can safely, effectively, and permanently change iris color. The company has good multinational intellectual property coverage and a clear regulatory and clinical pathway outside the United States. In the next few years, I hope to implement this technology in my practice, as I believe that the Strōma procedure signifies an excellent opportunity to expand the ophthalmic surgery market.


