I am an ophthalmologist and the founder and medical director of Augenzentrum Harburg (AZH), a high-volume private eye center in Hamburg, Germany. My focus is on cataract, vitreoretinal, and minimally invasive glaucoma surgery (MIGS). I initially used the iStent (Glaukos) when I started performing MIGS in 2019. However, seeking a more significant impact on IOP, I transitioned to alternative MIGS options over time.
My introduction to the OMNI Surgical System (Figure; Sight Sciences, Inc.) occurred in 2023, when I visited Syril Dorairaj, MD, at the Mayo Clinic in Florida, USA. Immediately, I found the OMNI Surgical System very straightforward to use. The learning curve for me was much shorter compared to other MIGS devices, and I could be confident I was treating the correct structures. Since then, I have been using the OMNI Surgical System regularly in my practice, and I’ve led events in Germany sharing my experiences on its use and clinical adoption to facilitate the growing interest in this MIGS approach.

Figure. The OMNI Surgical System.
Unique Action and Surgical Flexibility
What attracted me to the OMNI device is its unique mode of action, which targets three critical structures involved in aqueous humor outflow: the trabecular meshwork (TM), Schlemm’s canal, and the collector channels. Unlike other MIGS devices that bypass only one structure, OMNI works to restore the eye’s natural, physiological outflow pathway. Yet, the device doesn’t just bypass outflow resistance, it works to restore the natural outflow pathways, providing a physiological solution for IOP reduction without leaving an implant in the eye.
One important aspect of OMNI is that the ab-interno approach avoids disrupting the conjunctiva. This is a significant advantage, especially for patients in early stages of the disease, because it preserves the ocular surface for future surgical options, including traditional filtering procedures (trabeculectomies or tube-shunt surgery). This flexibility means I can tailor the treatment to individual patients’ needs, by either performing the procedure as a standalone or combining it with cataract surgery. The OMNI procedure’s broad applicability helps me address glaucoma early and effectively across diverse patient profiles.
Easy to Implement
I am currently using the Ergo series of OMNI, and I find it to have improved ergonomics alongside the intuitive control of previous iterations. What stands out is the adaptability of the procedure, which allows me to tailor my surgical approach for each patient (e.g., a 360º canaloplasty vs 180º, or a 360º trabeculotomy vs 180º, or no trabeculotomy).
Sight Sciences provides a comprehensive training program that I consider to be absolutely vital to achieving surgical success with the OMNI device. This program ensures that surgeons not only learn the technique, but also gain confidence in performing the procedure safely, efficiently, and effectively.
From my experience, one of the key ways surgeons can shorten the learning curve with OMNI is to schedule the first procedures in quick succession after training. This approach allows them to apply their newly acquired skills immediately to reinforce the learning and practice and refine their technique. It also enables them to observe the clinical results first-hand, which builds confidence in the efficacy offered by OMNI.
Efficacy in the Clinic, Supported by Data
Since I began using OMNI, I’ve observed excellent efficacy in lowering IOP. On average, I achieve an IOP reduction of 6 to 9 mm Hg, or approximately 25% to 35%, with the OMNI Surgical System, and in many cases, IOP stabilizes in the low teens. This is typically accompanied by a reduction of one to two medications. Also, the procedure has demonstrated a strong safety profile, with minimal to no complications and a quick recovery for my patients.
In 2023, Greenwood et al published the results of a prospective, multicenter trial conducted in the US to evaluate the long-term (3 year) IOP control and medication usage of patients who underwent MIGS with the OMNI Surgical System in combination with cataract surgery (the GEMINI study).1 Out of 66 patients, 78% achieved a reduction in IOP of ≥20%, and approximately 74% of the patients had stopped using glaucoma medications by 36 months. Ocular adverse events in the first 12 months of the GEMINI study were relatively uncommon, mild, and transient, and only four were considered related to the procedure or the device—one case of dry eye at 18 months that resolved, and three cases of posterior capsular opacification reported at 24 months. Only one eye required a tube shunt at 30 months postoperatively.
Similarly, earlier this year, Radcliffe et al published the results of a retrospective analysis of patients from the IRIS® Registry of patients with mostly moderate-to-severe primary open-angle glaucoma (POAG) who were treated with standalone canaloplasty and trabeculotomy using the OMNI Surgical System (n = 230 eyes in 196 patients).2 Mean IOP was 22.1 mm Hg at baseline and ranged from 15.1 to 16.7 mm Hg (P < 0.0001) at all postoperative time points. Mean glaucoma medications at baseline were 2.1 and ranged from 1.1 to 1.8 over 36 months. The authors concluded that canaloplasty and trabeculotomy performed as a standalone MIGS treatment provide clinically and statistically significant reductions in IOP out to 36 months. (For a summary of the original IRIS Registry Study,3 which was a real-world comparison of the results of three MIGS procedures with cataract surgery versus cataract surgery alone at 24 months, see the sidebar, The Iris Registry Study.)
The IRIS® Registry Study
- The study encompassed 109,745 glaucomatous eyes of 77,391 patients with glaucoma
- 9,000 eyes of 6,632 patients received a MIGS procedure plus cataract surgery; the remainder received just cataract surgery
- Patients were divided into two cohorts based on their baseline IOP: those with >18 mm Hg and those with ≤18 mm Hg
- OMNI performed better than the other interventions in both groups of patients (Figure)
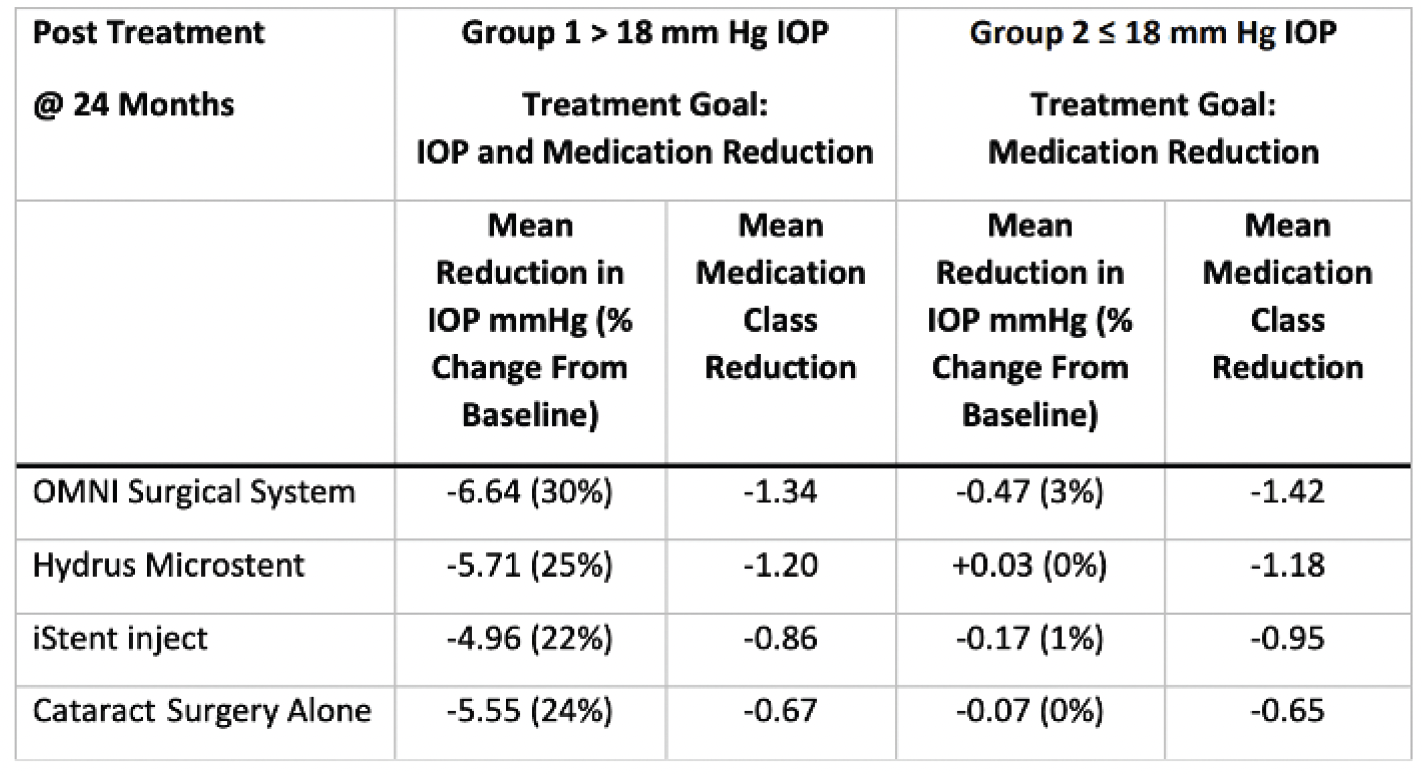
Mbagwu M, Chapman R, Garcia K, et al. Ab interno minimally invasive glaucoma surgery combined with cataract surgery and cataract surgery alone: IRIS® registry study. AJO International. 2024;100015, ISSN 2950-2535. https://www.sciencedirect.com/science/article/pii/S2950253524000157. Accessed June 19, 2025.
Conclusion
I am pleased to have found a flexible MIGS procedure that is easy to perform, is effective in its ability to lower both IOP and medication usage for patients, and has significant data supporting its long-term efficacy and safety. My patients are consistently pleased with their results, and I share their satisfaction. And, my peers with whom I’ve discussed various MIGS options appreciate that they can choose to use the OMNI either combined with cataract surgery or as a standalone procedure in pseudophakic patients. It has wide utility within the clinical armamentarium.
1. Greenwood MD, Yadgarov A, Flowers BE, et al. 36-month outcomes from the prospective GEMINI study: canaloplasty and trabeculotomy combined with cataract surgery for patients with primary open-angle glaucoma. Clin Ophthalmol. 2023;17:3817–3824.
2. Radcliffe NM, Harris J, Garcia K, et al. Standalone canaloplasty and trabeculotomy using the omni surgical system in eyes with primary open-angle glaucoma: a 36-month analysis from the American Academy of Ophthalmology IRIS® Registry (Intelligent Research in Sight). Am J Ophthalmol. 2025:271:436-444.
3. Mbagwu M, Chapman R, Garcia K, et al. Ab interno minimally invasive glaucoma surgery combined with cataract surgery and cataract surgery alone: IRIS® registry study. AJO Internationl. 2024;1(2):1-10.
Important Product Information
This information is intended solely for healthcare professionals located in the UK and EU. Patients should direct questions to their healthcare professional.
INDICATIONS FOR USE: The OMNI Surgical System is indicated for the catheterization and transluminal viscodilation of Schlemm’s canal and the cutting of trabecular meshwork to reduce intraocular pressure in adult patients with open-angle glaucoma.
For important product information including cautions and adverse events, please refer to the full instructions for use available at omnisurgical.com/international
OMNI and the Sight Sciences logo are registered trademarks of Sight Sciences, Inc.
All other copyrights and trademarks are owned by their respective owners.
©2025 Sight Sciences, Inc. 07/25





