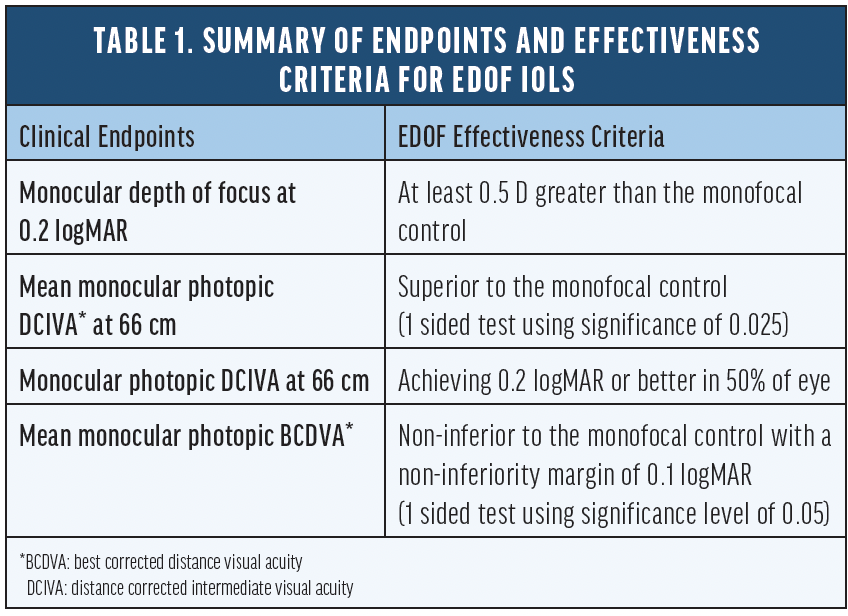
Introduction
Despite the significant advancement of presbyopia-correcting IOL technology, monofocal IOLs—which are designed to restore distance vision at a lower cost and are covered by medical insurance—remain the most implanted type of IOL worldwide. Recently, certain monofocal IOLs with modified aspheric optical profiles, reportedly designed to slightly extend the range of vision into intermediate distance, have been introduced to the market.1 It is important to understand the clinically meaningful benefit provided by these lenses versus other traditional monofocal IOLs, particularly given the associated additional cost.
Traditional Aspheric Monofocal IOLs
The optical design of monofocal IOLs has long been focused on providing high-quality distance vision. They were originally developed with spherical surfaces. Unlike the young human crystalline lens, which provides negative spherical aberration (SA)2 and can neutralize positive corneal SA,3 spherical IOLs introduce additional positive SA.4 Increased ocular SA reduces the clarity of an image as the aberrations cause light to deviate from the point of focus on the retina.4 The desire to provide sharply focused distance vision prompted the development of aspheric monofocal IOLs, which feature a gradual curvature reduction from center to the periphery, resulting in concomitant change in optical power from center to the peripheral portions of the optical zone.4 Today, we have a collection of aspheric lenses that are designed to neutralize all (e.g., TECNIS ZCB00* [Johnson & Johnson Vision], -0.27 μm SA), partial (e.g., AcrySof IQ SN60WF, Clareon SY60WF [both Alcon], -0.2 μm SA), or none (e.g., Akreos AO*, enVista MX60E* [both Bausch + Lomb], 0 μm SA) of the visual system’s naturally occurring corneal SA (+0.28 ± 0.09 μm for a 6-mm pupil).3 Different aspheric monofocal IOLs may be selected based on a patient’s corneal higher-order aberration profile to achieve higher-quality distance vision.
It should be noted that residual SA has the potential benefit of providing some depth of focus or tolerance to residual refractive error when it is not high enough to degrade retinal image quality. Patients with traditional monofocal IOLs, especially spherical or aspheric IOLs that do not fully neutralize corneal SA, have been reported to experience some intermediate vision.5-8
Modified Monofocal IOLs
Recently, a group of modified monofocal IOLs has emerged with optical designs aiming to extend the depth of focus slightly. However, Fernandez et al9 showed that none of these lenses are clinically proven to meet the extended depth-of-focus (EDOF) IOL standard as defined by the American National Standard Z80.35-2018 (Table 1).10 These lenses are marketed as modified or enhanced monofocal IOLs designed to extend the depth of focus. TECNIS Eyhance (Johnson & Johnson Vision), for example, is an FDA-approved monofocal IOL with a modified aspheric anterior surface which facilitates a steady increase in lens power within the central 1-mm diameter of the IOL optic. It was introduced as a level A modification of the TECNIS ZCB00 monofocal IOL, and did not require additional clinical study to verify that the modified optical design provided any additional benefit. Different studies have compared Eyhance to its parent lens, the TECNIS ZCB00, and showed various degrees of improvement in intermediate vision with mean value ranging from 0.09 to 0.2 logMAR.11-13 However, limited data is available comparing the range of vision between Eyhance and other traditional aspheric monofocal IOLs.
As mentioned above, having some intermediate vision with monofocal IOLs is not a novel concept nor a newly studied phenomenon. Rocha et al5,6showed residual SA with spherical IOLs and aspherical neutral IOLs could improve depth of focus. Bilateral implantation of AcrySof IQ monofocal IOLs has also been shown to provide intermediate vision of 0.2 logMAR.7,8The Clareon monofocal IOL is an innovation from Alcon with an advanced biomaterial to provide excellent optical clarity.14It shares a similar optical design as the AcrySof IQ monofocal IOL with -0.2 μm asphericity to partially compensate corneal SA, suggesting it may provide similar range of vision as the AcrySof IQ monofocal IOL. Blehm et al reported excellent distance vision and good intermediate vision (mean value of 0.16 and 0.23 logMAR at 80 cm and 66 cm, respectively) from a prospective study with the Clareon monofocal IOL.15
To better understand the visual performance of the Eyhance IOL versus an aspheric monofocal IOL other than TECNIS ZCB00, we conducted a large, non-interventional, single-center, multisurgeon, head-to-head study comparing visual outcomes in patients bilaterally implanted with either Clareon or Eyhance IOLs.16 Non-inferiority of the Clareon monofocal IOL relative to the Eyhance IOL was demonstrated in both BCDVA and DCIVA measured at 66 cm (Table 2) in an apples-to-apples comparison with both groups corrected to plano. A 2.5-ETDRS letter difference in DCIVA was observed between the two IOLs, which is well within the range of non-inferiority (<0.1 logMAR) and not considered to be a clinically meaningful difference. The binocular distance corrected defocus curve measurements were extremely similar from -3.00 D to +1.00 D defocus level (P > 0.05), suggesting comparable range of vision achieved by the two IOLs (Figure 1).
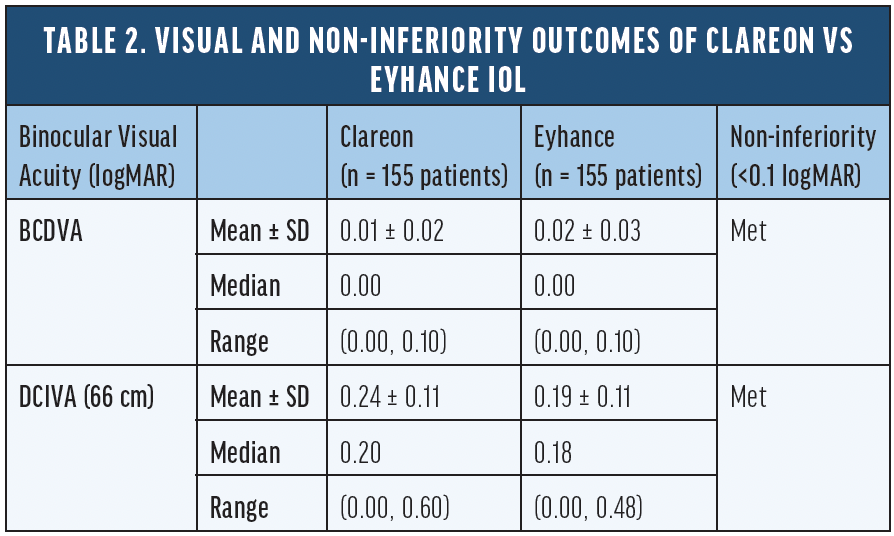
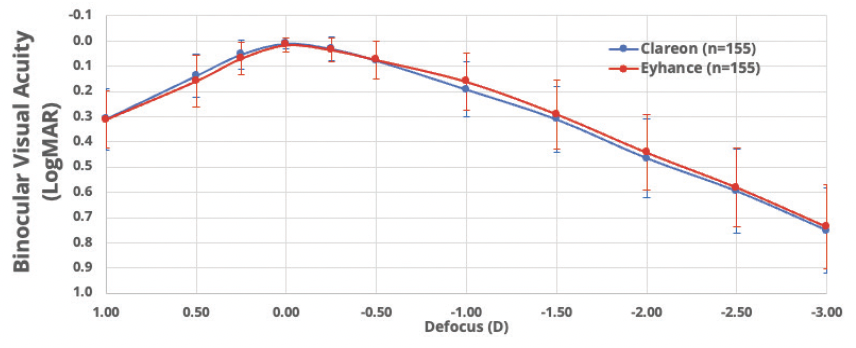
Figure 1. Binocular distance-corrected defocus curve of Clareon vs Eyhance IOL.
Additionally, it's crucial to underscore the importance of evaluating distance-corrected visual acuities at the relevant point of focus—distance, intermediate, or near—when comparing the visual performance of two IOLs. This approach allows us to rule out the influence of residual refractive error, ensuring a fair comparison of the visual benefits each IOL provides.
Summary
Monofocal IOLs remain an important option for cataract patients, typically providing excellent distance VA, but they also provide some intermediate vision depending on the optical design. The Eyhance IOL has been demonstrated to have slightly better intermediate VA compared to its parent IOL. However, this observation cannot be generalized to all monofocal IOLs. Our data conclude that the Clareon monofocal IOL can provide a comparable range of vision to the Eyhance IOL and has a similar potential to decrease spectacle wear at the intermediate range. The Clareon monofocal IOL may be an equally suitable choice with a relatively lower cost for surgeons who want to provide patients with the potential of some intermediate vision after cataract surgery. For patients who desire excellent vision from distance to intermediate and functional near vision with a low incidence of visual disturbance, non-diffractive EDOF IOLs should be considered. They are known to consistently deliver extended range of vision without the dependency on targeting strategies like mini-mono or monovision.7,8
Important Product Information - Clareon® Family of IOLs
Refer to the Directions for Use labeling for a complete list of indications, warnings and precautions
Alcon medical devices comply with the current legislation for Medical Devices. Exclusive use by healthcare professionals.
Alcon Portugal-Produtos e Equipamentos Oftalmológicos, Lda. NIPC.501 251 685
ANZ: ©2023 Alcon Laboratories Pty Ltd. AUS: 1800 224 153; Auckland NZ: 0800
©2023 Alcon Inc.
09/23 IMG-CLM-2300008
1. TECNIS Eyhance DFU.
2. Smith G, Cox MJ, Calver R, Garner LF. The spherical aberration of the crystalline lens of the human eye. Vision Res. 2001;41(2):235-243.
3. Wang L, Dai E, Koch DD, Nathoo A. Optical aberrations of the human anterior cornea. J Cataract Refract Surg. 2003;29(8):1514-1521.
4. Holladay JT, Piers PA, Koranyi G, van der Mooren M, Norrby NE. A new intraocular lens design to reduce spherical aberration of pseudophakic eyes. J Refract Surg. 2002;18(6):683-691.
5. Rocha KM, Soriano ES, Chamon W, Chalita MR, Nosé W. Spherical aberration and depth of focus in eyes implanted with aspheric and spherical intraocular lenses: a prospective randomized study. Ophthalmology. 2007;114(11):2050-2054.
6. Rocha KM, Gouvea L, Waring GO 4th, Haddad J. Static and dynamic factors associated with extended depth of focus in monofocal Intraocular Lenses. Am J Ophthalmol. 2020;216:271-282.
7. McCabe C, Berdahl J, Reiser H, et al. Clinical outcomes in a U.S. registration study of a new EDOF intraocular lens with a nondiffractive design. J Cataract Refract Surg. 2022;48(11):1297-1304.
8. Bala C, Poyales F, Guarro M, et al. Multicountry clinical outcomes of a new nondiffractive presbyopia-correcting IOL. J Cataract Refract Surg. 2022;48(2):136-143.
9. Fernández J, Rocha-de-Lossada C, Zamorano-Martín F, Rodríguez-Calvo-de-Mora M, Rodríguez-Vallejo M. Positioning of enhanced monofocal intraocular lenses between conventional monofocal and extended depth of focus lenses: a scoping review. BMC Ophthalmol. 2023;23(1):101.
10. American National Standard for Ophthalmics. ANSI Z80.35-2018: extended depth of focus intraocular lenses. 2018.
11. Auffarth GU, Gerl M, Tsai L, Janakiraman DP, Jackson B, Alarcon A, Dick HB; Quantum Study Group. Clinical evaluation of a new monofocal IOL with enhanced intermediate function in patients with cataract. J Cataract Refract Surg. 2021;47(2):184-191.
12. Mencucci R, Cennamo M, Venturi D, Vignapiano R, Favuzza E. Visual outcome, optical quality, and patient satisfaction with a new monofocal IOL, enhanced for intermediate vision: preliminary results. J Cataract Refract Surg. 2020;46(3):378-387.
13. Unsal U, Sabur H. Comparison of new monofocal innovative and standard monofocal intraocular lens after phacoemulsification. Int Ophthalmol. 2021;41(1):273-282.
14. Werner L, Thatthamla I, Ong M, et al. Evaluation of clarity characteristics in a new hydrophobic acrylic IOL in comparison to commercially available IOLs. J Cataract Refract Surg. 2019;45(10):1490-1497.
15. Blehm C, Hall B. Evaluation of visual outcomes and 3-month refractive stability of a new hydrophobic acrylic intraocular lens. Clin Ophthalmol. 2023;17:1859-1864.
16. Micheletti JM. Head-to-head comparison of two monofocal IOL. Presented at the annual meeting of the ASCRS. San Diego, CA. May 2023.





