CASE PRESENTATION
A 63-year-old woman experienced varicella zoster virus (shingles) V1 distribution of the trigeminal nerve in 2016 that scarred the central cornea in the right eye, leaving her with irregular hyperopic astigmatism in that eye. Visual acuity decreased further thereafter because of the formation of a nuclear sclerotic cataract.
The patient was referred for cataract surgery. On examination, UCVA was +5.25 -5.00 x 92º = poor-quality 20/30-2. Retinoscopy showed a distorted red reflex. A slit-lamp examination found central corneal scarring without significant vascularization (Figure 1). Corneal sensation was mildly reduced, and mild to moderate nuclear sclerosis was apparent. Tomography demonstrated irregular astigmatism (Figure 2).
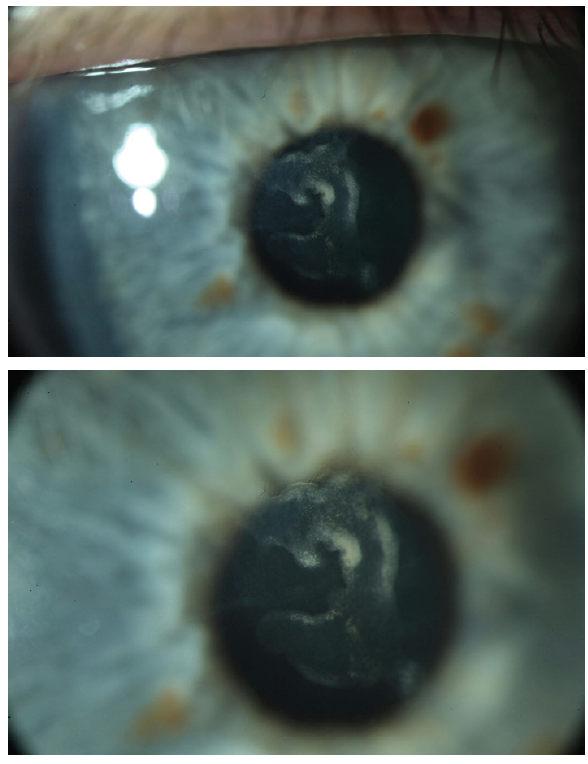
Figure 1. Central scarring of the anterior corneal stroma (top, bottom). The scarring does not involve the posterior 400 μm of the corneal stroma.
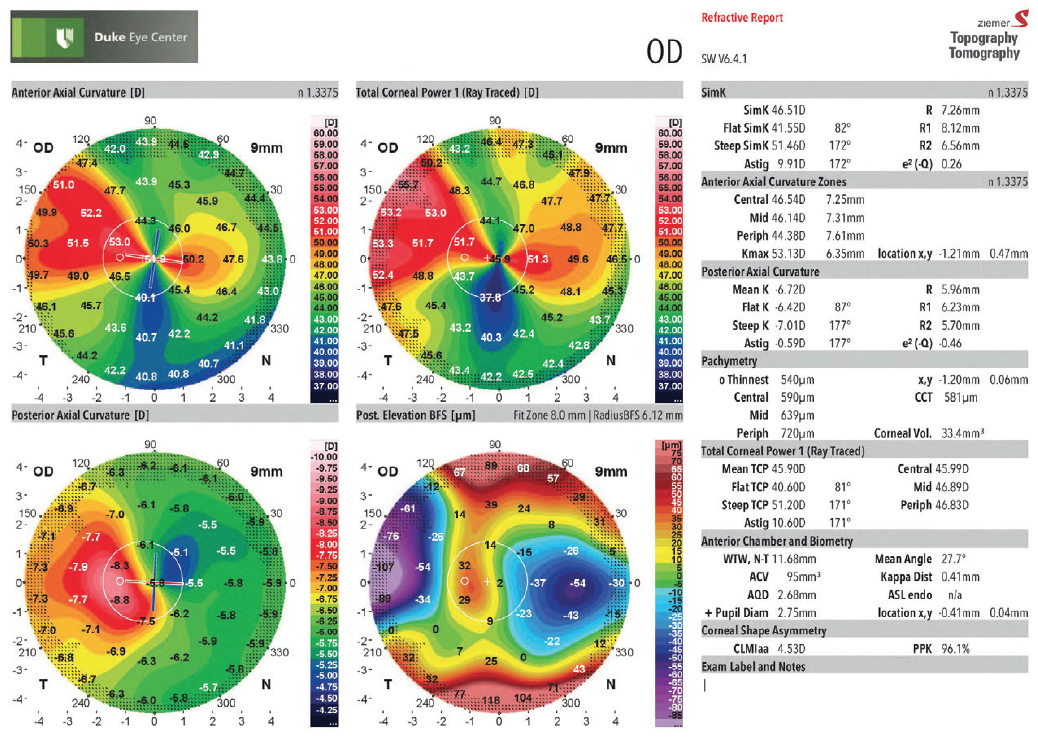
Figure 2. The patient’s right-eye tomography (Galilei G4, Ziemer) demonstrates asymmetric and irregular astigmatism.
Multiple attempts at fitting a scleral contact lens (Synergeyes VS, Synergeyes) achieved a poor-quality UCVA of 20/30-. Although the patient felt that she could wear this lens, she expressed concern that she will become intolerant of it as she ages. She is otherwise healthy and has a robust life expectancy.
How would you proceed?
—Case prepared by Alan N. Carlson, MD

TOBIAS H. NEUHANN, MD, FEBOS-CR
It would be helpful to know if the scar has remained quiet since 2016. If so, I would consider treating the corneal scar with surface ablation using a high-speed 1,050-Hz excimer laser system with reliable eye tracking. My first step would be to obtain topography and aberrometry measurements as well as high-resolution swept-source OCT scans of the corneal epithelium and stroma. OCT would determine whether performing a phototherapeutic keratectomy (PTK) is a reasonable strategy.
If the scar is sufficiently shallow, as I think is the case here, traditional PTK is an option. The procedure is technically easy, but this patient will likely require several PTK treatments and measurements, which will be time-consuming and not very precise.
Another option is aberrometry-guided transepithelial PTK using proprietary software from Schwind eye-tech-solutions (not available in the United States). This unique software calculates the depth of treatment and the amount of irregular hyperopic astigmatism to allow PTK/PRK treatment to be performed in one step. If the thickness of the epithelium is irregular, the epithelium should be manually removed. In addition to my typical medical regimen after PTK, I would prescribe antiviral topical and systemic therapy to reduce the possibility of viral inflammation.
Three to 6 months postoperatively, cataract surgery could be planned if the patient wished, but the IOL power calculation will be challenging. My inclination would therefore be to implant a RxLAL (RxSight).

FRANCIS W. PRICE JR, MD
Not presented in this case are the depth of the scar, whether this patient has experienced recurrent corneal inflammation, and her quality of vision 1 year after the episode of shingles. If corneal inflammation has been neither persistent nor recurrent, then an important question is whether she was happy with or could tolerate the visual acuity she had 1 year after the outbreak, as that would presumably be her visual acuity without the cataract. If she found her visual acuity at least tolerable, then cataract surgery and implantation of a toric lens could be performed. The patient would have residual glare and somewhat decreased visual acuity, but her life could continue with minimal disruption. If, however, this patient has experienced either persistent or recurrent episodes of inflammation, as often happens after herpes zoster ophthalmicus (HZO), then she needs to receive treatment with topical corticosteroids until the inflammation subsides, and she may well require topical steroid therapy indefinitely. After the eye has been quiet for at least 1 year, surgery may be considered.
If the eye has been totally quiet for the past few years, and she was always unhappy with the distortion of her vision, then deep anterior lamellar keratoplasty (DALK) may be offered. The patient may require treatment with a low-dose topical steroid indefinitely because the case presentation suggests some vascularization that is not currently significant. After all keratoplasty sutures are removed, residual refractive error may be fine-tuned with cataract surgery.
High regular astigmatism is an issue because it could have contributed to the poor quality of vision and anisometropia with glasses, making it difficult for the patient to evaluate the quality of her vision after the viral outbreak. Although the cataract clouds the issue somewhat, if her vision with a scleral lens was too distorted from corneal haze and irregularity, it is probably safe to assume that the problems were caused by the corneal scarring. The only way to fix that would be with a graft, especially if the scarring extends deep into the midstroma. Excimer laser surgery is contraindicated in eyes with herpes simplex virus infections because the intervention can cause severe reactivation. I am not sure that excimer laser surgery would reactivate HZO, but I would err on the side of caution. Discussing with the patient the option of receiving a graft, the time required for healing, and the possible need for her to undergo another procedure to treat residual astigmatism may yield a clearer picture of how badly the scarring is affecting her.
I have found that zigzag incisions for the sidecuts in DALK offer somewhat faster healing, more secure wounds, and a surprisingly lower risk of stromal rejection episodes.1,2
A final note is that placing a corneal graft in eyes with a history of HZO raises concern about decreased sensation and can be problematic because the cornea is already neurotrophic. If the patient developed exposure or ulceration of the cornea, a lateral tarsorrhaphy might be required, so she should be made aware of that possibility before surgery.

KHOR WEI BOON, MBBS, FRCS(ED), FAMS
Given this patient’s poor quality of vision, I would offer her the option of anterior lamellar keratoplasty to remove the central corneal scar. My preference would be hemi-automated lamellar keratoplasty (HALK).3,4
HALK is indicated for anterior to midstromal scars in corneas that are steep or thin or have an irregular surface. In this technique, manual lamellar dissection is performed with a sharp crescent blade to a point below the scar, followed by transplantation of an anterior cap of corneal tissue prepared with a microkeratome by an eye bank. Sharp lamellar dissection produces a smooth stromal bed that, when coupled with the smooth stromal surface of the graft, minimizes the risk of interface scarring. HALK avoids the main complication of DALK, namely perforation of the Descemet membrane. The lamellar graft can also be exchanged readily if keratitis and scarring recur.
In this instance, I would perform the transplantation after the herpes zoster keratitis had been quiescent for 6 months. After transplantation, I would instruct the patient to continue using oral acyclovir 400 mg twice a day as prophylaxis (off-label use) for at least the first year, until the topical steroids could be tapered and eventually stopped at 9 to 12 months. Selective suture removal for astigmatic control could begin at 6 months. Finally, when the corneal astigmatism was reasonably low (ideally ≤ 2.00 D) and stable for 3 months, I would perform phacoemulsification and lens implantation to remove the cataract and correct any spherical error.

WHAT I DID: ALAN N. CARLSON, MD
Drs. Neuhann, Price, and Khor offer excellent advice on this case of a patient progressing toward cataract surgery complicated by scarring from herpes zoster viral keratitis. The scarring from the herpes zoster viral keratitis fortunately remains stable. There has been some epithelial remodeling from hyperplasia, leaving a 200-µm depression and nearly 10.00 D of against-the-rule asymmetric corneal astigmatism. Contact lens correction would likely require a scleral lens. Although PTK using a masking agent is an option, treatment to this depth can make postoperative recovery poorly predictable, and it is likely to result in late stromal scarring, even with the application of mitomycin C.
An important aspect of patient care is managing patient expectations. Meeting the expectations of most of the patients I see who are in situations similar to this one would likely require performing a lamellar corneal procedure prior to cataract surgery to achieve the desired outcome. Although I have been achieving the best results with DALK in cases such as this one, stromal dissection techniques have improved and can approach outcomes similar to DALK. The cataract and astigmatism would be addressed after corneal healing.
This particular patient is easygoing and happy with the visual acuity of the contralateral eye. That combination gives us the luxury of time, so I am monitoring both eyes while advances in technology and techniques continue. If her cataract were worse and her expectations higher, I would proceed with DALK followed by cataract surgery, as I did with a teenaged patient 2 years ago, as shown in the Video.
1. Price FW Jr, Price MO, Grandin JC, Kwon R. Deep anterior lamellar keratoplasty with femtosecond-laser zigzag incisions. J Cataract Refract Surg. 2009;35(5):804-808.
2. Gonzalez A, Price MO, Feng MT, Lee C, Arbelaez JG, Price FW Jr. Immunologic rejection episodes after deep lamellar keratoplasty: incidence and risk factors. Cornea. 2017;36(9):1076-1082.
3. Yuen LH, Mehta JS, Shilbayeh R, Lim L, Tan DT. Hemi-automated lamellar keratoplasty (HALK). Br J Ophthalmol. 2011;95(11):1513-1518.
4. Fuest M, Liu YC, Arundhati A, Li L, Tan D, Mehta JS. Long-term outcomes of hemi-automated lamellar keratoplasty. Clin Exp Ophthalmol. 2018;46(9):1017-1027.



