
Glaucoma surgery is evolving. Today, a variety of procedures can be used to lower patients’ IOP, but some procedures are more invasive than others. It's important to therefore start with the least invasive treatment and work your way up—like a staircase of surgical treatments—to more invasive options as and when necessary.
The iStent inject® W (Glaukos) and other microsurgical devices offer a great opportunity for patients to reduce their IOP with less risk of complications compared to conventional incisional techniques.1,2 When performed early in the disease state, the iStent technologies can control IOP in the long term,3-6 which may save many patients with glaucoma from having a bigger operation further down the line.
SUITABLE PATIENTS
The iStent inject® W is an important device to consider for many patients, including those with cataracts who use at least one IOP-lowering medication. If you are doing an operation inside the eye for anyone with elevated IOP and functional symptoms that disturb their daily quality of life, it makes sense to do something for the glaucoma at the time of surgery. For these cases, I always consider the iStent inject® W.
The device is also great for patients who struggle with drops. Although IOP-lowering drops are effective, studies suggest that 50% to 90% of patients do not adhere to their drop schedules as prescribed.5,7-10 This could be, for example, because they experience side effects such as irritation and redness or because of the associated cost (Figures 1 and 2). If undertreated for any reason, the disease can progress. Additionally, now that longer-term data are available for the iStent technologies3,6,11,12 I also consider the device for patients who are well controlled on only one drop. When I can put in a trabecular micro-bypass and alleviate the drop burden, that can be life-changing for patients.
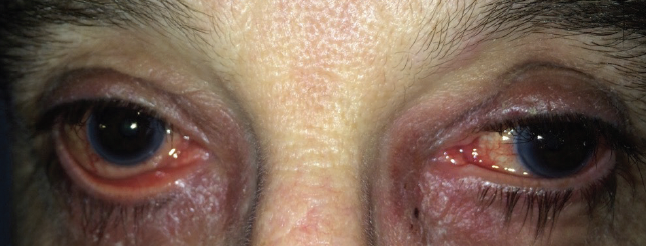
Figure 1. Side effects of glaucoma topical treatment. In this patient, the glaucoma is preperimetric and asymptomatic.

Figure 2. Polypharmacy in a patient with glaucoma.
A LOOK AT COST UTILITY
My real-world experience shows that the iStent inject® W is not only clinically effective, but it's likely cost-effective as well, what is in line with the results from published studies on iStent inject®.13-16 The cost utility of the device includes not only its cost on day 1 but how it translates into benefits for the patient over the next 5 to 10 years. For patients who no longer need IOP-lowering drops or those who require fewer drops because they're well treated with the iStent inject® W, there are fewer trips to the doctor and pharmacy. This benefits not only the patient but the whole health care system as well.
Keeping patients’ IOP stable on fewer or no drops also creates more capacity for clinicians to help other patients who are higher risk and may need more invasive surgery. From my point of view, there's added benefit to offering the iStent inject® W beyond its effect on individual patients and the teams that look after them because it can translate to clinical and cost benefits for the entire health care system.
AN EMPHASIS ON EARLY INTERVENTION
A lot of patients with glaucoma will require cataract surgery because the treatment of glaucoma itself can often accelerate cataract formation.17 When counseling these patients about cataract surgery, it is important to make them aware that surgical options are available to them. Early intervention can be important.
I counsel patients at the beginning of their glaucoma treatment that surgical options like the iStent inject® W (Figures 3 and 4) are available so that they get used to the idea that we may add a minimally invasive procedure at the end of their cataract surgery. I explain to them that there is a minimal impact on the procedure, and studies have shown that implantation of iStent inject® has a similar safety profile to that of cataract surgery alone.2

Figure 3. iStent introducer and view of the drainage angle.
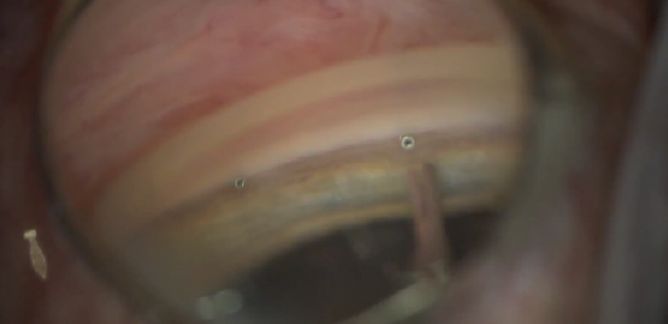
Figure 4. Two iStent inject® are visible in situ.
I think it's important to maintain equity of access to this treatment because it has the potential to improve quality of life and help with disease progression control in a wide range of patients.18,19 It is our role as practitioners to make sure we educate all suitable patients about the iStent inject® W rather than waiting for the internet-savvy patient who has researched treatments to bring it up. Many patients who would never come across such treatment themselves can benefit significantly from the iStent inject® W.
Appropriate early intervention has the potential to reduce patients' burden of eye drops. This is important in the modern glaucoma era. Patients welcome drop-independent treatment for glaucoma, and a trabecular micro-bypass device like the iStent inject® W may offer that possibility. Appropriate and timely intervention also reduces the need for more invasive surgeries, which carries significant increased risks. There is no doubt that our rate of filtering surgery has reduced because of trabecular micro-bypass procedures. Also, the burden of postoperative management is different with early intervention. The eye recovers much more quickly after a minimally invasive procedure, and postoperative care (eg, healing response and the need for intensive antiinflammatory treatments) is not as labor-intensive. It can also have a positive response for the ocular surface. As we know, ocular surface disease (OSD) affects up to 30% of the general and up to 60% of the population with glaucoma5,19-23 The latter group is much more at risk of the annoying and uncomfortable feelings of gritty, dry eyes and redness. Reducing the drop burden can help alleviate the symptoms of dry eye disease.
LASTING RESULTS
Most sentinel clinical trials look for a 20% reduction in IOP from baseline. In my experience, patients achieve this with the iStent inject® W. In fact, some patients have a much more dramatic drop in pressure. Additionally, the IOP-lowering effect is sustainable. Clinical trial results that are just coming out indicate the benefits can extend well beyond 6 to 12 months, in some cases going out 5 to 7 years and beyond.3,6,11,12,24,25
The iStent inject® W offers additional IOP-lowering benefits on top of the cataract procedure.2 The IOP-lowering from the cataract procedure itself can wear off after approximately 2 years,8 whereas the iStent technologies provide sustained reduction.3,4,6 For patients with more controlled disease over the long term, there are fewer clinic visits and they achieve better vision. I don’t think it's an understatement to say that the iStent inject® W can be life-changing for some patients.
CONCLUSION
Patients with glaucoma can be categorized into three groups according to the risk of disease progression (ie, low, medium, and high risk). For patients who are lower risk, the goal should be good IOP control over the longer term with minimal disruption and impact on their quality of life. Early intervention with a device such as the iStent inject® W can provide patients with an excellent opportunity to achieve this goal.
The iStent inject® W has patient benefits in terms of quality of life, staff benefits in terms of delivering a high level of care and patient experience, and overall health care system benefits in terms of reduced patient burdens. It aids our ability to provide good treatment pathways for low-, medium-, and high-risk patients. Offering the iStent inject® W is an easy and sustainable way to provide customized care and deliver better results within the same financial envelope.
1. Watson PG, Jakeman C, Ozturk M, Barnett MF, Barnett F, Khaw KT. The complications of trabeculectomy (a 20-year follow-up). Eye. 1990;4(3):425-438.
2. Samuelson TW, Sarkisian SR Jr, Lubeck DM, et al. Prospective, randomized, controlled pivotal trial of an ab interno implanted trabecular micro-bypass in primary open-angle glaucoma and cataract: two-year results. Ophthalmology. 2019;126(6):811-821.
3. Hengerer FH, Auffarth GU, Conrad-Hengerer I. iStent inject trabecular micro-bypass with or without cataract surgery yields sustained 5-year glaucoma control. Adv Ther. 2022;39:1417-1431.
4. Salimi A, Watt H, Harasymowycz P. Long-term outcomes of two first-generation trabecular micro-bypass stents (iStent) with phacoemulsification in primary open-angle glaucoma: eight-year results. Eye and Vision. 2021;8(1):1-12.
5. Fechtner RD, Godfrey DG, Budenz D, Stewart JA, Stewart WC, Jasek MC. Prevalence of ocular surface complaints in patients with glaucoma using topical intraocular pressure-lowering medications. Cornea. 2010; 29(6):618-621.
6. Ziaei H, Au L. Manchester iStent study: long-term 7-year outcomes. Eye. 2021;35:2277-2282.
7. Li G, Akpek EK, Ahmad S. Glaucoma and ocular surface disease: more than meets the eye. Clin Ophthalmol. 2022:16;3641-3649.
8. Nordstrom BL, Friedman DS, Mozaffari E, Quigley H, Walker AM. Persistence and adherence with topical glaucoma therapy. Am J Ophthalmol. 2005;140(4):598-606.
9. Tsai JC, McClure CA, Ramos SE, Schlundt DG, Pichert JW. Compliance barriers in glaucoma: a systematic classification. J Glaucoma. 2003;12(5):393-398.8.
10. Skalicky SE, Goldberg I, McCluskey P. Ocular surface disease and quality of life in patients with glaucoma. Am J Ophthalmol. 2012;153(1):1–9.e2.
11. Healey PR, Clement CI, Kerr NM, et al. Standalone iStent trabecular micro-bypass glaucoma surgery: a systematic review and meta-analysis. J Glaucoma. 2021;30(7):606-620.
12. Cantor L, Lindfield D, Ghinelli F, et al. Systematic literature review of clinical, economic, and humanistic outcomes following minimally invasive glaucoma surgery or selective laser trabeculoplasty for the treatment of open-angle glaucoma with or without cataract extraction. Clin Ophthalmology. 2023;17:85-101.
13. Schweitzer C, et al. A cost-utility analysis of the iStent inject® trabecular mi¬cro-bypass system plus cataract surgery in patients with mild-to-moderate open-angle glaucoma in France. Paper presented at the: ESCRS Annual Meeting; September 14-18, 2019; Paris.
14. Garcia-Feijoo J, Teus M. Cost-effectiveness analysis of iStent inject® implantation for the treatment of open-angle glaucoma in Spain. Paper presented at the: ESCRS Annual Meeting; September 14-18, 2019; Paris.
15. Belda JI. Budget impact analysis of the IStent inject® implant for open angle glaucoma treatment in Spain. Paper presented at the: ESCRS Annual Meeting; September 14-18, 2019; Paris.
16. Bucholz PM. A budget-impact analysis of the iStent inject® trabecular micro-bypass system vs trabeculectomy for the treatment of glaucoma from a German payer perspective. Poster presented at the: ESCRS Annual Meeting; September 14-18, 2019; Paris.
17. Agis Investigators. The advanced glaucoma intervention study, risk of cataract formation after trabeculectomy. Arch Ophthalmol. 2001;119(12):1771-1779.
18. Samuelson TW, Singh IP, Williamson BK, et al. Quality of life in primary open-angle glaucoma and cataract: an analysis of VFQ-25 and OSDI from the iStent inject® pivotal trial. Am J Ophthalmol. 2021;229:220-229.
19. Schweitzer JA, Hauser WH, Ibach M, et al. Prospective interventional cohort study of ocular surface disease changes in eyes after trabecular micro-bypass stent(s) implantation (iStent or iStent inject) with phacoemulsification. Ophthalmology and Therapy. 2020;9:941-953.
20. Prum BE Jr, Rosenberg LF, Gedde SJ, et al. Primary open-angle glaucoma preferred practice pattern((R)) guidelines. Ophthalmology. 2016; 123(1):41–111.
21. Leung EW, Medeiros FA, Weinreb RN. Prevalence of ocular surface disease in glaucoma patients. J Glaucoma. 2008;17(5):350-355.
22. Dana R, Bradley JL, Guerin A, et al. Estimated prevalence and incidence of dry eye disease based on coding analysis of a large, all-age United States health care system. Am J Ophthalmol. 2019;202:47-54.
23. Farrand KF, Fridman M, Stillman IO, Schaumberg DA. Prevalence of diagnosed dry eye disease in the United States among adults aged 18 years and older. Am J Ophthalmol. 2017;182:90-98.
24. Mansberger SL, O Gordon M, Jampel H, et al. Reduction in intraocular pressure after cataract extraction: the Ocular Hypertension Treatment Study. Ophthalmology. 2012;119(9):1826-1831.
25. Ferguson TJ, Mechels KB, Dockter Z, et al. iStent trabecular microbypass stent implantation with phacoemulsification in patients with open-angle glaucoma: 6-year outcomes. Clin Ophthalmol. 2020;14:1859-1866.
iStent inject® W IMPORTANT SAFETY INFORMATION
INDICATION FOR USE: The iStent inject® W, is intended to reduce intraocular pressure safely and effectively in patients diagnosed with primary open-angle glaucoma, pseudo-exfoliative glaucoma or pigmentary glaucoma. The iStent inject® W, can deliver two (2) stents on a single pass, through a single incision. The implant is designed to stent open a passage through the trabecular meshwork to allow for an increase in the facility of outflow and a subsequent reduction in intraocular pressure. The device is safe and effective when implanted in combination with cataract surgery in those subjects who require intraocular pressure reduction and/or would benefit from glaucoma medication reduction. The device may also be implanted in patients who continue to have elevated intraocular pressure despite prior treatment with glaucoma medications and conventional glaucoma surgery. CONTRAINDICATIONS: The iStent inject® W System is contraindicated under the following circumstances or conditions: • In eyes with primary angle closure glaucoma, or secondary angle-closure glaucoma, including neovascular glaucoma, because the device would not be expected to work in such situations. • In patients with retrobulbar tumor, thyroid eye disease, Sturge-Weber Syndrome or any other type of condition that may cause elevated episcleral venous pressure. WARNINGS/PRECAUTIONS: • For prescription use only. • This device has not been studied in patients with uveitic glaucoma. • Do not use the device if the Tyvek® lid has been opened or the packaging appears damaged. In such cases, the sterility of the device may be compromised. • Due to the sharpness of certain injector components (i.e. the insertion sleeve and trocar), care should be exercised to grasp the injector body. Dispose of device in a sharps container. • iStent inject® W is MR-Conditional; see MRI Information below. • Physician training is required prior to use of the iStent inject® W System. • Do not re-use the stent(s) or injector, as this may result in infection and/or intraocular inflammation, as well as occurrence of potential postoperative adverse events as shown below under “Potential Complications.” • There are no known compatibility issues with the iStent inject® W and other intraoperative devices. (e.g., viscoelastics) or glaucoma medications. • Unused product & packaging may be disposed of in accordance with facility procedures. Implanted medical devices and contaminated products must be disposed of as medical waste. • The surgeon should monitor the patient postoperatively for proper maintenance of intraocular pressure. If intraocular pressure is not adequately maintained after surgery, the surgeon should consider an appropriate treatment regimen to reduce intraocular pressure. • Patients should be informed that placement of the stents, without concomitant cataract surgery in phakic patients, can enhance the formation or progression of cataract. ADVERSE EVENTS: Please refer to Directions For Use for additional adverse event information. CAUTION: Please reference the Directions For Use labelling for a complete list of contraindications, warnings and adverse events.
© 2023 Glaukos Corporation. Glaukos, iStent®, iStent inject® and iStent inject® W are registered trademarks of Glaukos Corporation. PM-EU-0232




