The TM is a complex 3D structure that plays a major role in IOP regulation. The majority of aqueous outflow (70% to 90%) occurs through the conventional trabecular outflow pathway. Aqueous humor passes through cell-lined beams of the TM before moving into the canal and collector channels. SC endothelial cells help to maintain the blood-aqueous barrier.
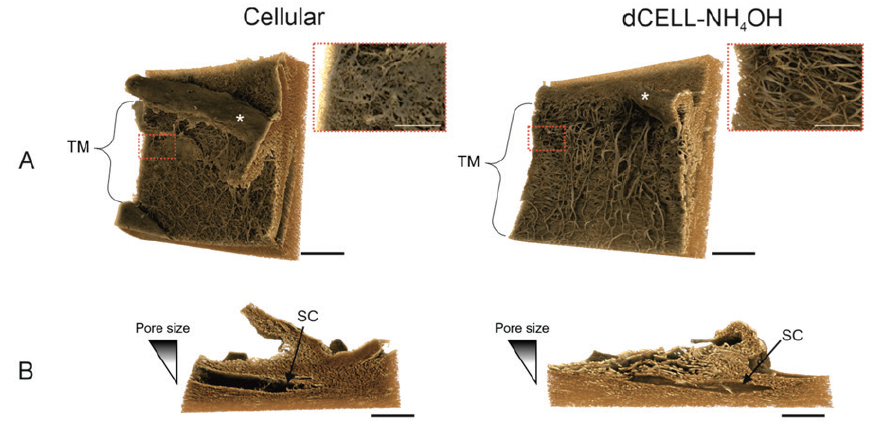
Figure 1. MicroCT images of cellular and decellularized human TM in front-on (A) and cross-sectional (B) orientations.
A noninvasive imaging tool called microcomputed tomography (microCT) has been developed to enable 3D reconstruction of the TM and help visualize the tissue’s architecture.1 Work done at the University of Liverpool depicts a healthy TM with cells following decellularization with ammonium hydroxide (Figure 1). With decellularization, the complex structure and decreasing porous nature of the TM are apparent.
Under normal physiological conditions, the uveal and corneoscleral meshwork do not offer resistance to outflow; instead, resistance to outflow is generated in the juxtacanicular TM–canal interface. Scanning electron microscopy, which helps depict the TM’s surface structure, shows clear physical alterations in structure with primary open-angle glaucoma. In a healthy TM, pores through which aqueous travels are visible, whereas in a glaucomatous TM, a dense plaque-like material obstructs outflow.
Beyond the TM
Aqueous outflow beyond the TM is a dynamic process. TM cells have baroreceptor properties, and fluctuations of these valve-like conduits help to open and close Schlemm’s canal. Using ultra high-resolution OCT, Johnstone et al2 showed how the TM distends and pulls on the collector channels ostia entrance in response to a pulse, causing the channels to open and close. The outflow pathway has similar lymphatic-like characteristics that allow drainage through valves into distal collector channels and vessels.
ELIOS Treatment in Detail
My colleagues and I used the ELIOS laser to create microchannels in donor human TM. Scanning electron micrographs (Figure 2) show the well-defined microchannels. The treatment is 120 µm in depth, and the Schlemm’s canal lumen space remains patent adjacent to and beyond the pore. Maintaining the natural physiology of aqueous drainage is key—and it is possible with the ELIOS laser.

Figure 2. Well-defined microchannels in the TM created by the ELIOS laser.
1. Crouch DJ, Sheridan CM, Behnsen JG, Bosworth LA. An optimized method to decellularize human trabecular meshwork. Bioengineering. 2022;9(5):194.
2. Johnstone M, Xin C, Tan J, et al. Aqueous outflow regulation – 21st century concepts. Prog Retin Eye Res. 2021;83:100917.



