In the spring, I sat down with my colleagues Patricio G. Schlottmann, MD; Paulo-Eduardo Stanga, MD; and Daniel Ting, MD, PhD, to discuss the future of geographic atrophy (GA) therapy, to examine the role of various imaging modalities in following patients with GA, and to review evidence-based approaches for making the diagnosis and determining the prognosis for future lesion growth. The discussion, which can be read here, was productive—and also demonstrated that further discussions are needed to grasp the breadth of GA practice patterns and to explore topics we were unable to address within the scope of our roundtable.
Thus, I invited three more of my European colleagues—Jordi Monés, MD, PhD, from Spain; Sobha Sivaprasad, MD, from the United Kingdom; and Giovanni Staurenghi, MD, from Italy—to join me for a discussion about how GA affects the lives of our patients, how we orient new patients to GA, and how we discuss the value of follow-up with GA patients who have been in our clinics for years. We also reviewed whether AREDS supplementation is a viable treatment strategy given the latest data on the topic. We hope you find insights from our roundtable useful.
If you prefer, you can listen to the roundtable discussion as a series of podcasts on New Retina Radio. Or, if you’re a visual learner, watch videos of our discussions on Eyetube. References to the videos can be found below. The article you are about to read is adapted from that set of discussions and has been edited for brevity and clarity.
In the middle of this print piece, you’ll see a breakdown of a survey Retina Today sent to ophthalmologists outside of the United States who manage GA in their clinics. Interestingly, the survey results compare practice patterns among retina specialists and comprehensive ophthalmologists. My interpretation of a handful of the survey’s questions is included herein. Hopefully these survey results (and my responses to them) help us better understand how providers outside of the United States are addressing GA in real-world settings.
— Anat Loewenstein, MD, MHA
Anat Loewenstein, MD, MHA: When GA robs patients of their vision, it also robs them of their independence. How do you see GA progression negatively affecting the independence of your patients?
Giovanni Staurenghi, MD: Various characteristics dictate how GA will affect a patient’s independence. For example, if the patient has foveal involvement, their central vision will be worse. If a patient has reticular pseudodrusen (RPD), they will likely have difficulty with dark and light adaptation. Patients often experience depression and anxiety when an activity linked to independence (eg, driving) is taken from them.
Sobha Sivaprasad, MD: I have also observed that patients who are caregivers for spouses with life-threatening illnesses have a sense of anxiety that they themselves will be unable to care for loved ones. We should remember that caregiving goes both ways with GA patients: they sometimes require caregivers and at other times, they themselves are the caregivers.
Jordi Monés, MD, PhD: I have seen my patients lose self-esteem when they lose the ability to perform activities that defined their lives, such as reading, driving, self-care, or staying physically active. I have also seen them feel misunderstood about how GA has affected their day-to-day lives.
Patients with remaining vision often experience inconsistent or spotty vision, and family members sometimes fail to understand how patients can see one thing but not another. “Come on, Mom,” I have heard family members say. “You can see the words on this paper, so surely you can drive.”
When retina specialists recognize and validate the experience of patients with objective measurements and explain to family members or caretakers how GA has affected the patient’s vision, two things may occur: patients will feel understood, and caretakers will better understand the subjective experience of their loved one.
Prof. Loewenstein: GA results in more than just vision loss: it places patients at risk for other health-related complications. Which health risks might be associated with GA?
Prof. Sivaprasad: Patients who cannot see well are at higher risk for falls. Recent research from the United States shows that falls were associated with approximately 3 million visits to an emergency department, nearly 1 million hospitalizations, and approximately 32,000 deaths among those older than 65.1 Moreover, the older the patients became, the more likely they were to fall, with nearly 14% of patients over 85 experiencing a fall within the previous year.1
Patients in my clinic have reported difficulty with distinguishing between medications and have also reported difficulty with reading medication labels. This is an obvious problem for the population we treat, as many of our GA patients are diagnosed with comorbid conditions for which they take medication.
Survey Results
In the summer of 2025, Retina Today conducted a survey of 308 ophthalmologists who practice outside the United States. A little more than half of the respondents (53%) were comprehensive ophthalmologists, with the rest of the respondents comprising retina specialists.
From the survey, I have selected the responses that I think shed the most useful light on the current landscape of geographic atrophy (GA) treatment outside of the United States. The charts speak for themselves; in case you wanted further interpretation, you can see my commentary below each question.
—Anat Loewenstein, MD, MHA
WHAT PERCENTAGE OF YOUR PATIENT BASE IS PRESENTING WITH GEOGRAPHIC ATROPHY?
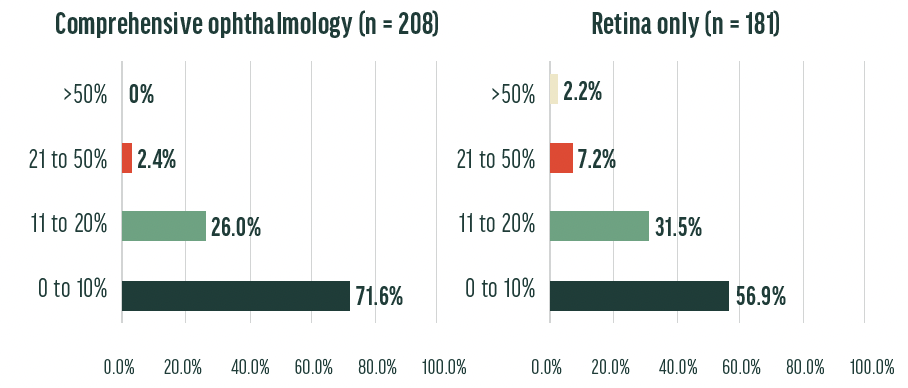
Interpretation: Perhaps unsurprisingly, retina specialists see a higher percentage of patients with GA in practice, with nearly 41% of retina specialists reporting that GA composes more than 11% of their patient population; in comprehensive ophthalmology clinics, this percentage was only 28%. Still, a considerable percentage of comprehensive ophthalmology physicians see patients with GA in their clinics. As you read in the roundtable discussion, providers differ on whether they prefer GA patients stay with their primary eye care provider or be referred promptly. Regardless of that practice preference, these results make clear that patients with GA can indeed be found in comprehensive ophthalmology clinics.
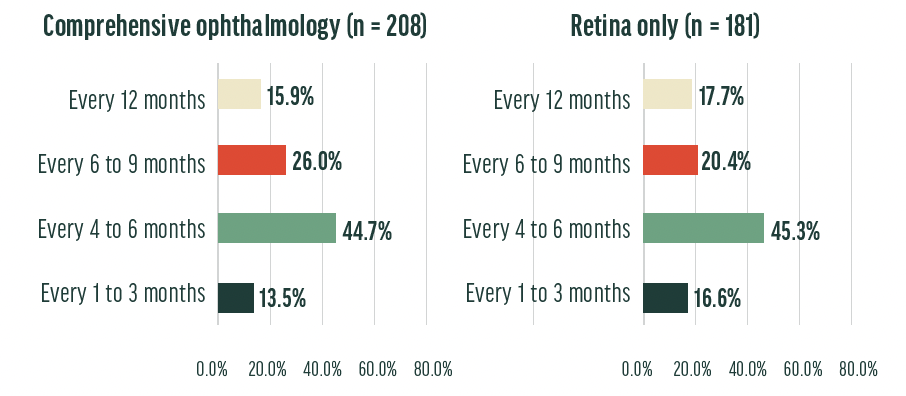
Interpretation: Retina specialists and comprehensive ophthalmologists have a similar distribution of preferences for follow-up patterns, with approximately 45% of both groups preferring an interval of 4 to 6 months. It is difficult to know, however, whether the survey respondents tailor their follow-up regimens to the patient’s specific presentation. For example, do providers follow early GA at different intervals than they do long-term GA? Further discussions similar to the roundtable in this piece, and further surveys similar to the one we’re discussing now, are needed to learn more.
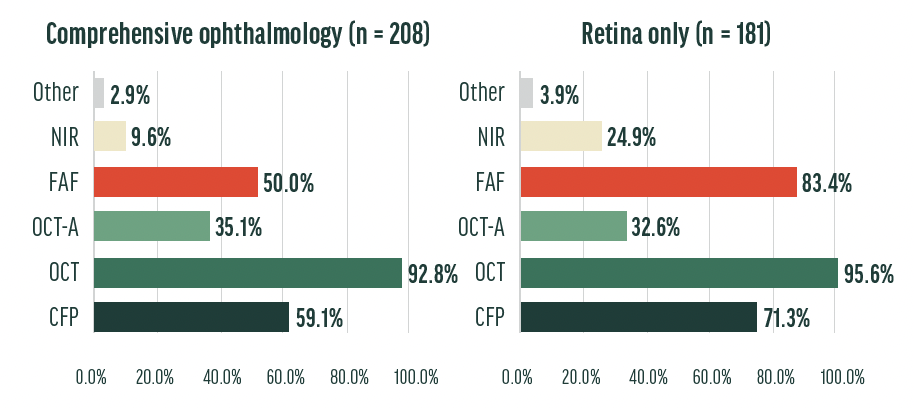
Interpretation: OCT imaging is the most popular imaging modality for both retina-only and comprehensive ophthalmology practices, which is perhaps unsurprising given the ease of using OCT, the ubiquity of OCT equipment, and the intuitive interpretation of OCT images. Higher percentages of retina-only clinics use fundus autofluorescence, color fundus photography, and near infrared reflectance to monitor GA compared with comprehensive ophthalmology clinics. These differences could be for any number of reasons: comprehensive ophthalmology clinics may lack the access to these modalities that retina-only clinics have, or comprehensive ophthalmology providers may not be as trained in interpreting results of these modalities.

Interpretation: More than a quarter of retina only providers discuss complement inhibition therapy with their patients, compared to approximately 17% of comprehensive ophthalmology providers. This may be due to familiarity with complement inhibitor data. On the other hand, comprehensive ophthalmology clinics were more likely to discuss photobiomodulation treatment with their patients, reflecting an embrace of this technology by those providers. Smoking cessation and AREDS vitamin supplementation were recommended at similar rates among the two groups; diet modifications and reduced sunlight exposure were also recommended at similar rates, albeit at rates lower than smoking cessation and AREDS supplementation. The biggest takeaway: lifestyle modification recommendations are popular among eye care providers who see GA patients.
New Geographic Atrophy Patients
Prof. Loewenstein: New patients who have GA present to our clinics from a variety of avenues. Prof. Staurenghi, how do new patients present to your clinic?
Prof. Staurenghi: Patients with GA may present with undiagnosed disease, arrive having received a diagnosis from a non–retina specialist, or be referred during the intermediate stage of age-related macular degeneration (AMD) for monitoring, subsequently progressing to an advanced stage, which may manifest as neovascular AMD, GA, or both. Historically, referrals to the retina clinic for patients with intermediate AMD or GA were uncommon. In recent years, however, there has been a notable increase in referrals of patients with intermediate AMD or suspected GA. This rise in patient volume has significantly altered the clinical profile of my patient population.

Prof. Monés: It’s notable how different clinics have different means of building a patient population. Many of the patients in my practice present directly to our clinic rather than via a referral avenue. In some cases, GA patients are former intermediate AMD patients who have been under our care for a while and have converted to GA. In other instances, they are new GA patients who present directly to our retina clinic.
Prof. Sivaprasad: We rely on a registry at my clinic, but also rely on referrals from optometrists, who provide routine eyecare in the United Kingdom.
Prof. Staurenghi: Patients themselves are a source of referral. When a patient from my clinic with intermediate AMD or GA knows a friend or family member with the same condition, they refer them to my practice. These patient-to-patient referrals have helped us build a robust GA and intermediate AMD patient population.
Prof. Loewenstein: When is the best time for a referral network to refer a patient to your clinic?
Prof. Sivaprasad: Smart minds in our field disagree on the answer to this question. For my practice in the United Kingdom, which does not have access to complement inhibition therapy, the best answer is for referring optometrists to closely monitor patients with intermediate AMD and GA until I can provide a pharmacologic intervention.
Prof. Monés: I prefer that my referral network send in all patients with suspected intermediate AMD or GA. As an expert in the field, I feel I am best equipped to assess and diagnose patients, and I don’t feel comfortable relying on the diagnosis of a provider who doesn’t share my background or who doesn’t have access to the same imaging modalities that I have in my clinic.
Further, as a provider who monitors clinical trials in GA, I can connect interested patients who fit enrollment criteria with clinical trials sites. Also, if there is a special access program to complement inhibition, then a complement inhibitor can be imported. As such, I advise my referral network to send me all patients with suspected or diagnosed intermediate AMD and GA.
I recognize that my clinic is unique in that I do not operate within a national public health service, which affords me greater time to speak with patients about their condition, educate them about pathogenesis, and answer their pressing questions. Providers in other health care settings—especially those which are resource limited—will have a different experience than I do.
WATCH THESE DISCUSSIONS ON EYETUBE
Prof. Staurenghi: I also prefer that patients be referred too early rather than too late. As I practice in a referral center, many of the individuals I evaluate have already progressed beyond intermediate AMD. Earlier referral allows us to integrate them into our care pathway sooner, enabling more effective counseling and monitoring.
Prof. Monés raises an excellent point regarding confidence in one’s diagnostic abilities. In my experience, some patients referred with presumed intermediate AMD or GA are ultimately found to have a different condition. The importance of establishing an accurate differential diagnosis is a key argument in favor of timely referral.
Prof. Loewenstein: Patients with intermediate AMD or GA could have a wide range of vision quality they present with. In general, how functional is your new patients’ vision?
Prof. Monés: It’s difficult to pick an average vision quality because the spectrum of presentations is so wide. Some patients present with obviously decreased vision but preserved best corrected visual acuity (BCVA), which is not uncommon in GA.
Whenever I assess patients whose BCVA doesn’t match their subjective description of vision, I tell them that I believe that their vision is compromised even if their BCVA says otherwise. This validates the patient’s experience. I also find that asking careful questions reveals the degree to which the patient experiences visual dysfunction. Asking about how quickly their eyes adapt to a dark room, for example, may reveal more specific information than merely asking, “How well do you think you see?”
Prof. Staurenghi: In my clinic, I see many GA patients with highly dysfunctional vision. This is because they have waited too long to present for an evaluation and examination. These patients often describe themselves as blind, and I work hard to remind them that they are not blind, but rather that they have low vision, and that individuals with low vision can still lead productive lives.
LISTEN TO THESE CONVERSATIONS ON NEW RETINA RADIO
Want to listen to the long-form conversations from which this roundtable was adapted? Head to New Retina Radio on your preferred podcast platform.
Prof. Loewenstein: With new patients, I emphasize that lifestyle interventions are wise, which is particularly true of smokers. Do you advise lifestyle interventions?
Prof. Sivaprasad: I, too, emphasize smoking cessation. Smoking is among the list of modifiable risk factors linked to AMD, and cessation could play a role in slowing disease progression. The time of diagnosis is also when we introduce an easily digested prognosis to the patient and a caregiver, if they have one with them. If necessary, I will direct the patient to a low vision clinic, which can connect them with social services and offer practical advice for navigating their new life.
Prof. Monés: I provide some top-line take-home points for patients with newly diagnosed GA. I educate patients that GA is a slow, progressive disease. That means that while their disease will grow, it will do so slowly, which means that in many instances patients will not lose all their vision. I inform them about clinical trials for which they could be eligible, and let them know that, in some circumstances, complement inhibition treatments used in the United States could be an option.

I also educate them that neovascular AMD and GA can coexist and emphasize that I will monitor them for signs of neovascular AMD. Like Prof. Sivaprasad, I educate caregivers if they are in the room as well, as a patient’s support system should understand the disease course as well as the patient understands it.
Breaking Down the Data on AREDS Vitamins in Geographic Atrophy
In 2025, Keenan et al1 published findings from a post hoc analysis of the Age-Related Eye Disease Study (AREDS) and the AREDS2 study, which were a pair of multicenter, randomized, placebo-controlled studies assessing the efficacy of oral micronutrient supplements in patients with age-related macular degeneration (AMD). Patients in AREDS and AREDS2 were randomly assigned to receive any one of several formulations of oral antioxidant supplements.
AREDS Findings. Among eyes with noncentral GA in AREDS (n = 208), progression toward the central macula was significantly slower among those who took antioxidant supplements compared with those who did not; specifically, those who took AREDS antioxidant supplements progressed toward the central macula at a rate of 50.7 μm/year, compared with 72.9 μm/year among those who did not take any supplements (P = .012).
AREDS2 Findings. Patients with noncentral GA in AREDS2 who were randomly assigned to receive AREDS antioxidants without beta-carotene (n = 325) experienced slower progression toward the central macula when they also consumed AREDS supplementation that included lutein/zeaxanthin compared with those who did not consume lutein/zeaxanthin (80.1 μm/year vs 114.4 μm/year, respectively; P = .011).
Eyes With Any Geographic Atrophy. When eyes with any GA (ie, not just noncentral GA) were assessed, no significant changes in the rate of progression were observed in AREDS or AREDS2.
1. Keenan TDL, Agrón E, Keane PA, et al; Age-Related Eye Disease Study Research Group; Age-Related Eye Disease Study 2 Research Group. Oral antioxidant and lutein/zeaxanthin supplements slow geographic atrophy progression to the fovea in age-related macular degeneration. Ophthalmology. 2025;132(1):14-29.
Legacy Geographic Atrophy Patients
Prof. Loewenstein: Patients with GA who have been in our clinics for a long time are treated differently than those who are presenting for the first time with GA. What are some of the ways in which these patients are different?
Prof. Monés: These patients have experience with reading imaging results, as (at least in my clinic) we educate patients about how imaging results depict GA. As many of my colleagues have observed, sharing imaging results with patients aids in educating them about their disease, and helps them grasp what is (for some patients) a complex concept.
Prof. Loewenstein: Do you follow the fellow eye in unifocal GA patients who have been under your care for a long time? If so, how closely?
Prof. Monés: At the very least, I assess my legacy GA patients every 6 months, and I always examine their non-GA fellow eye. Performing OCT imaging and fundus autofluorescence imaging in both eyes every 6 months ensures that I do not miss any progression that a single-imaging approach or non-imaging examination would not depict. Remember, our GA patients are at risk for conversion to neovascular AMD. We must remain vigilant specialists who detect disease early.
Prof. Sivaprasad: At present, we do not monitor a patient’s fellow eye more than annually. Even then, the monitoring is via virtual consultation, not through an in-person examination.
Prof. Loewenstein: How do you convince legacy GA patients that they should follow-up with your clinic? Right now, we cannot provide treatments that restore lost vision.
Prof. Staurenghi: I educate my patients that GA may not be the only retinal condition for which they need treatment or monitoring. We must remember that GA and neovascular AMD can co-exist in the same eye. The incidence rate of choroidal neovascularization or macular neovascularization in eyes with GA is nearly 14% within 4.1 years among GA patients.2 Further, I am routinely becoming aware of new clinical trials for GA treatments, and if patients return to my clinic for examination, I can assess their eligibility for such studies. To that end, I ask that patients return every 6 months to my clinic for follow-up and monitoring.
Follow-up appointments with legacy GA patients also allow providers to interact with caregivers. Opportunities to explain to caregivers how a patient’s prognosis will playout in practical terms can help them plan for the future.
Prof. Loewenstein: What technology do we lack that might be useful for educating our legacy GA patients?
Prof. Sivaprasad: It would be interesting to see how patients would react to a virtual reality simulation of potential vision loss. Patients with intermediate AMD might better understand the stakes of potential GA if they could “experience” in virtual reality the consequences of progressed disease; the same could be said of legacy patients with GA and good vision. However, I worry that such technology could also induce anxiety. The last thing we want to do is frighten patients.
One resource that is easy to overlook because it is analog in nature: support groups. I refer GA patients to the United Kingdom’s Macular Society, which facilitates discussion among peer groups. At these discussions, patients learn from one another how best to navigate the new realities dictated by their eye health. They can also get connected with low-vision specialists.
Prof. Loewenstein: I would love to see an AI-driven depiction of a patient’s likely progression based on natural history studies. If I could prognosticate with reasonable likelihood that a patient’s GA will, for example, progress to the fovea within 36 months, I could better prepare them for how their life will change and could tailor treatment advice to the specifics of their case. Providers in nations that have limited or no access to complement inhibitor treatments could use this tool to build a list of patients who could potentially benefit the most from an approved treatment, which could speed the rate of adoption of these treatments if and when they become available.
Are AREDS Vitamins an Answer for Some Patients?
Prof. Loewenstein: Keenan et al recently found that some formulations of AREDS supplements significantly slow GA growth among patients with noncentral GA.3 However, these findings also concluded that no difference existed among all GA patients, which is to say the differences in progression were no longer statistically significant when patients with central GA were included in the study population. How do we interpret these findings clinically, particularly in places where complement inhibitors are difficult to acquire or unavailable? [Editors’ note: To zoom in on this study, see the sidebar Breaking Down the Data on AREDS Vitamins in Geographic Atrophy].
Prof. Sivaprasad: Although a post hoc analysis should be approached with caution, I will say that these findings made me reconsider the degree to which I discuss AREDS vitamins with my patients. I also discuss the value of a Mediterranean diet with them, as the link between nutrition and ocular health is becoming clearer each year.
Prof. Monés: I live and practice in Spain, where the Mediterranean diet is quite common. AREDS supplementation is so useful in nations without easy, affordable access to the Mediterranean diet. Still, even though these foods are easily accessed in my home country, I prescribe AREDS supplementation because the burden is low and the upside is high. The cost-benefit ratio, in my estimation, makes supplement recommendation very easy.
Prof. Staurenghi: At least in Italy, I do not have access to the AREDS supplement formulation evaluated by Keenan et al., as it would be classified as a pharmaceutical product. Unfortunately, the formulation available here is offered at a different dosage, and in the absence of clinical trial data specific to this product, it is not possible to make evidence-based recommendations to patients. Moreover, attempting to replicate the AREDS dosage by combining multiple tablets may result in excessive intake of certain components compared with the original US formulation.
Prof. Loewenstein: I think we agree that the data from Keenan et al intrigue us, but it seems like more research could be performed.
Prof. Sivaprasad: It would be great if we could run a single randomized prospective clinical trial, but such a study is cost prohibitive, in part because it would require an endpoint approximately 6.5 years after initiation. Observational studies are perhaps the best we can do for now.
Prof. Loewenstein: Even if a prospective clinical trial were less expensive, it would still require a third-party organization such as the US National Institutes of Health or EURETINA to fund, organize, and execute such a study. In today’s economic climate, that is likely unfeasible.
Thank you to all three of you for participating in this discussion. Our colleagues will hopefully find it useful.
1. Hu A, Teneralli RE, Rodriguez R, et al. Falls and fractures in patients with geographic atrophy: a US claims data analysis. Adv Ther. 2025;42(5):2234-2247.
2. Kataoka K, Gale R, Li X, et al. Simultaneous GA and CNV/MNV: incidence, characteristics, and treatments. Graefes Arch Clin Exp Ophthalmol. 2025;263(5):1197-1212.
3. Keenan TDL, Agrón E, Keane PA, et al; Age-Related Eye Disease Study Research Group; Age-Related Eye Disease Study 2 Research Group. Oral antioxidant and lutein/zeaxanthin supplements slow geographic atrophy progression to the fovea in age-related macular degeneration. Ophthalmology. 2025;132(1):14-29.








