For the past century, we've been relying on topical glaucoma medications as the first-line treatment of a disease that can lead to blindness. The standard of care for glaucoma that most of us were taught was to use topical medications first, exhaust them, then move on to interventions. Next came laser trabeculoplasty, and then filtering surgeries, like trabeculectomy and tube shunts, as a last resort. Now, new diagnostic and therapeutic options are giving us good reasons to reevaluate our protocols. Over the years, more and more studies have revealed the shortcomings of topical glaucoma medications, which include issues of toxicity, compliance, adherence, and even efficacy.
THE CASE FOR CHANGING THE TREATMENT PARADIGM
Toxicity and Compliance
Stacking topical glaucoma drops, which has historically been the standard of care, is fraught with problems of toxicity and compliance. Documented side effects include hyperemia, preorbital fat atrophy, ocular surface disease, and iris hypochromia.1 Medication stacking often instigates dry eye disease (DED),2,3 as Figure 1 illustrates. In turn, these symptoms may contribute to medication noncompliance and nonadherence.4,5
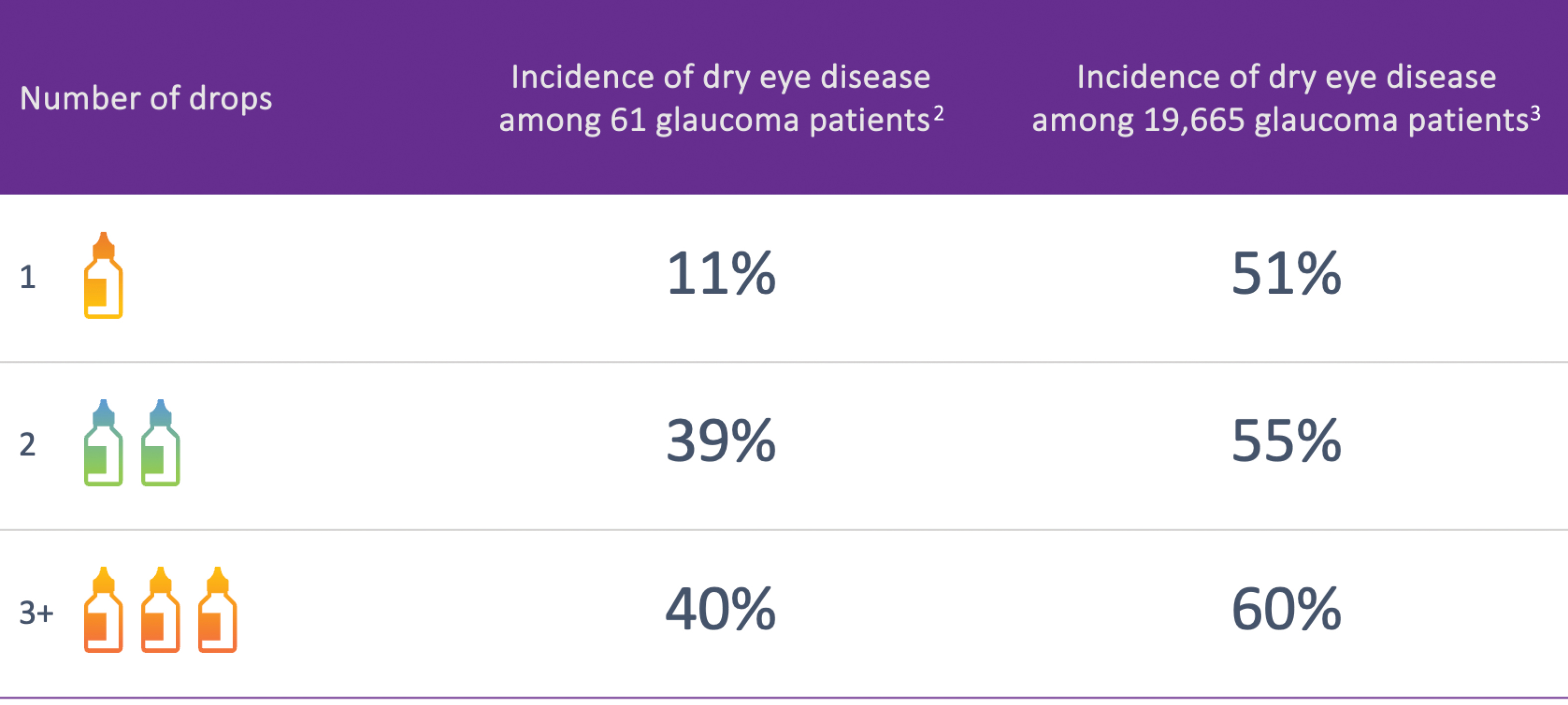
Figure 1. Studies show a direct correlation between the number of glaucoma medications used and the incidence and severity of dry eye
disease.2,3 (Data adapted from Rossi et al, Eur J Ophthlamol, 2009; and Erb et al, Graefes Arch Clin Exp Ophthalmol, 2008.)
Courtesy of I. Paul Singh, MD.
Prescription medication nonadherence is an issue in and of itself and likely contributes to the disease’s progression. In a retrospective cohort study of health insurance claims data, Nordstrom et al found that almost 50% of those who filled a topical glaucoma prescription discontinued the therapy within 6 months.1 Prescription renewals for the various drug categories in glaucoma over a period of several years decreased dramatically in every drug class.6 Should we be taking the burden of adherence out of patients' hands?
Efficacy of IOP Control
Equally important is the question of how effectively these topical pharmaceuticals control IOP. Is it stabilized for 24 hours, 7 days a week? Recent data indicate that the practice of medication stacking is not very effective in lowering IOP after the initial drop7 (Figure 2), or in preventing blindness. Neelakantan et al found that the addition of a third and fourth glaucoma medication produced a relatively poor cumulative probability of success, including safety outcomes.8 In the United States, Malihi et al conducted a longitudinal study to determine the trend toward blindness in patients with open-angle glaucoma between 1965 and 2009. Despite treatment with topical glaucoma medications, patients’ probability of disease progression leading to blindness in at least 1 eye was 13.5%, and the average time to blindness was 4.8 years.9
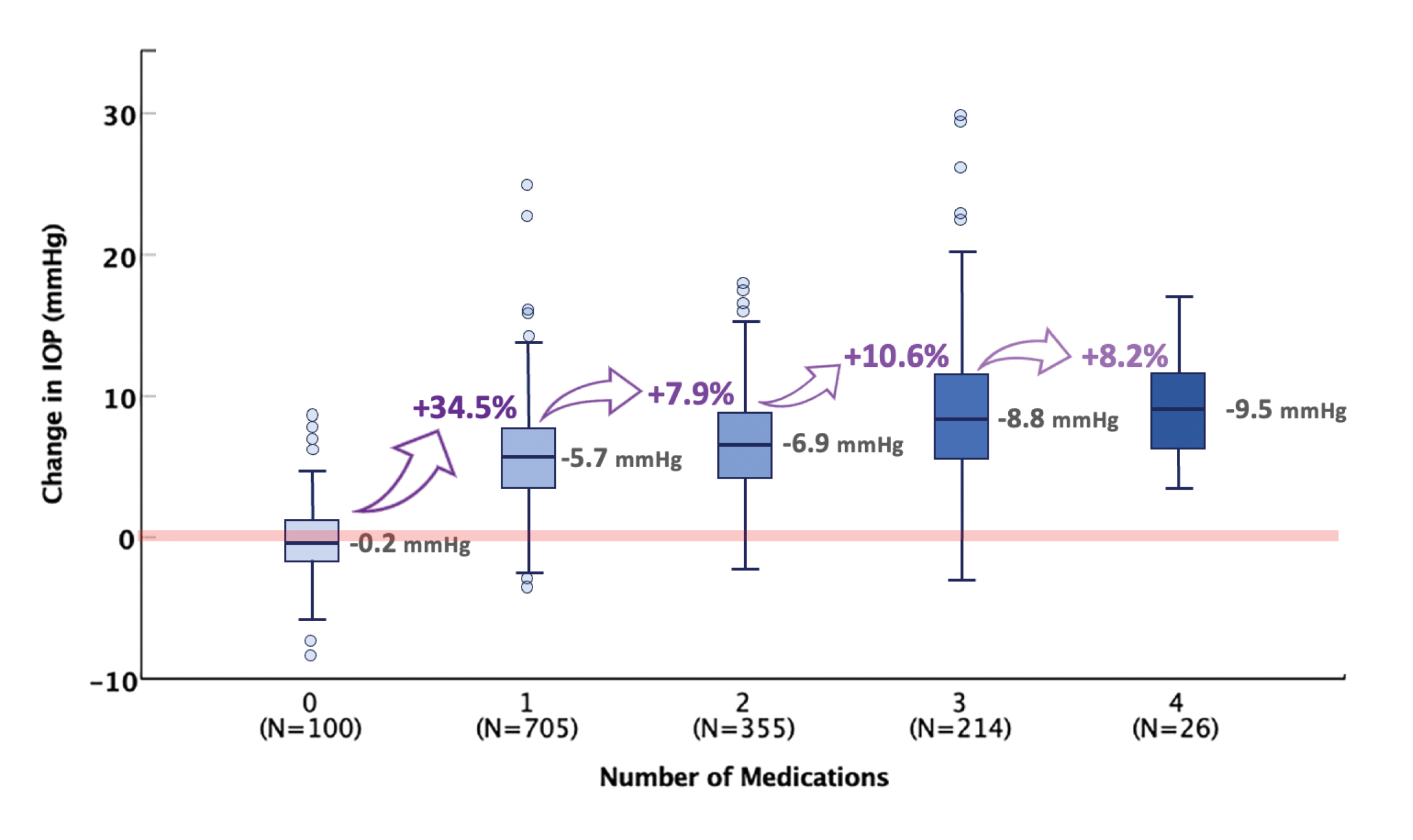
Figure 2. The first glaucoma medication is often the most impactful in reducing IOP. Subsequent medications have less effect. (Adapted from: Johnson TV et al. Am J Ophthalmol. 2020.7)
Courtesy of Iqbal "Ike" K. Ahmed, MD
In Europe, Peters et al conducted a retrospective study of patients who received glaucoma treatment for 20 years. By the conclusion of the study, more than one-third of the patients were blind in one eye, and 13% were blind in both eyes.10 I consider these data an indictment against our standard treatment paradigm. Together, these findings suggest that we should change our approach to both evaluating and treating glaucoma.
A New Model: Interventional Glaucoma Care
Some of our glaucoma colleagues, such as Iqbal “Ike” Ahmed, MD, in the United States, have been leading change in our field, especially in the surgical management of glaucoma. Dr. Ahmed describes interventional glaucoma as a proactive approach to treating the disease, in which clinicians regularly screen for glaucoma, actively monitor patients’ disease, and institute procedural interventions earlier in the treatment paradigm, with the goal of keeping patients’ vision stable over the long term whilst considering topical medications as supplemental only (Figure 3).
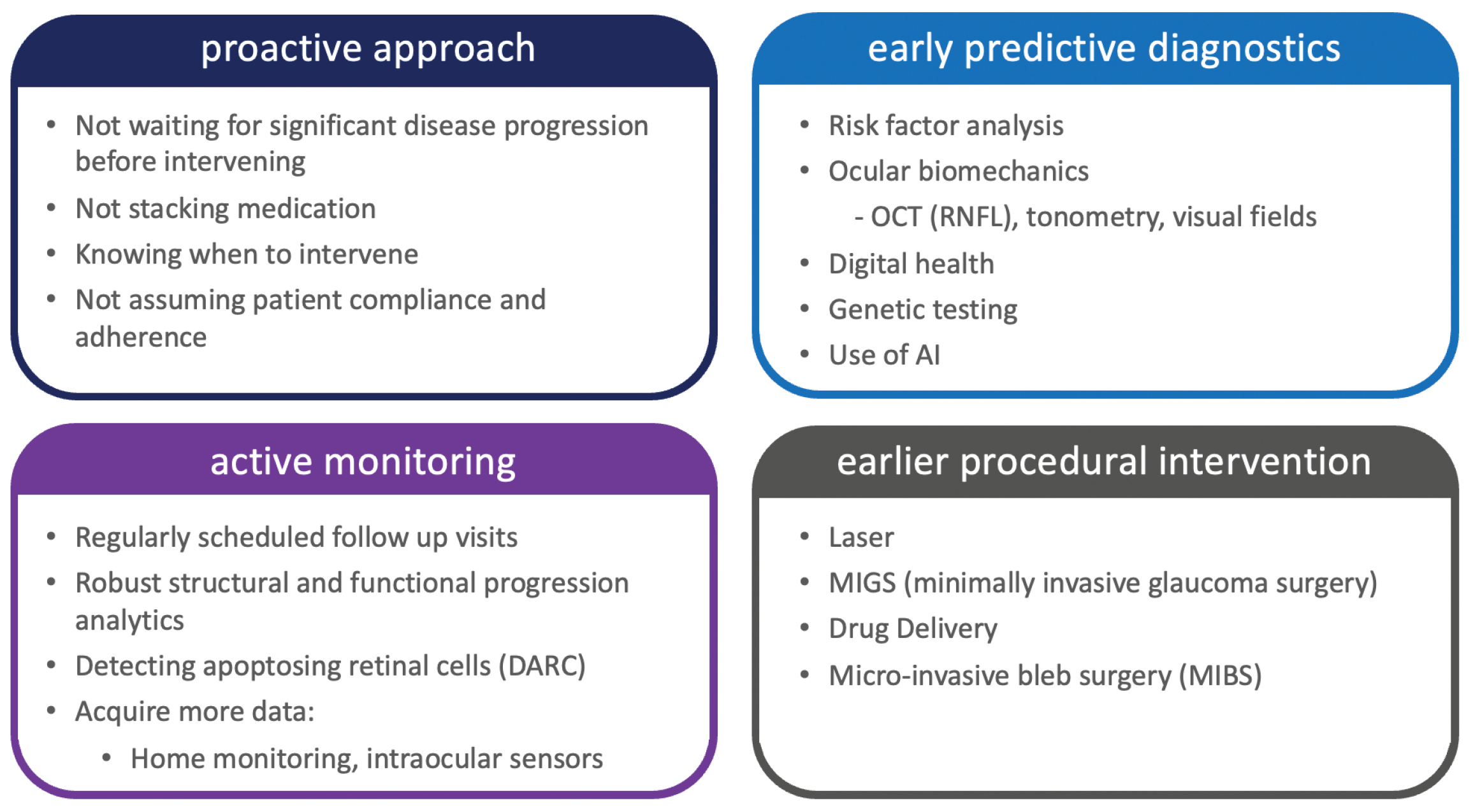
Figure 3. The differentiators of interventional glaucoma, as outlined by Iqbal "Ike" K. Ahmed, MD, of the John A. Moran Eye Center in Utah, USA.
A new era of early predictive diagnostics is emerging, in which technology will enable the detection of glaucoma at a very early stage and actively monitor the disease’s progression. No longer should we wait for a noticeable decline in vision; now, we can intervene quickly to adequately control a patient’s glaucoma.
We are already seeing the tip of the iceberg of interventional glaucoma with the introduction of laser trabeculoplasty and procedural pharmaceuticals, which are increasingly being adopted as first-line therapies for newly diagnosed open-angle glaucoma patients.11 Minimally invasive glaucoma surgery (MIGS) is increasingly introduced at fairly early stages, especially in combination with cataract surgery, with encouraging results.12 Additionally, filtering surgery with stents like XEN® Gel Stent (AbbVie/Allergan) and the PRESERFLO™ Ab-Externo Miroshunt (Santen) can provide IOP reduction with less risk and more standardization than trabeculectomy and tube shunts.
CONCLUSION
Well-run clinical trials have shown the safety, efficacy, and cost savings of interventional alternatives to topical pharmaceutical glaucoma management. Our clinicians and researchers will continue to amass scientific evidence favoring the use of procedural pharmaceuticals, MIGS, trabeculectomies, and other modalities for maintaining consistent IOP control. I foresee increased adoption of the interventional mindset to treat glaucoma more proactively, rather than waiting for the disease’s progression to force our hands.
To hear more from Dr. Katz, visit https://eyetube.net/spotlight/glaukos-international/glaukos-egs-symposium-2024-video-2
1. Alm A, Grierson I, Shields MB. Side effects associated with prostaglandin analog therapy. Surv Ophthalmol. 2008;53 Suppl1:S93-105.
2. Rossi GC, Tinelli C, Pasinetti GM, et al. Dry eye syndrome-related quality of life in glaucoma patients. Eur J Ophthalmol. 2009;19(4):572-579.
3. Erb C, Gast U, Schremmer D. German register for glaucoma patients with dry eye. I. Basic outcome with respect to dry eye. Graefes Arch Clin Exp Ophthalmol. 2008;246(11):1593-1601.
4. Baudouin C. Detrimental effect of preservatives in eyedrops: implications for the treatment of glaucoma. Acta Ophthlmol. 2008;86(7):716-726.
5. Stringham J, Ashkenazy N, Galor A, Wellick SR. Barriers to glaucoma medication compliance among veterans: dry eye symptoms and anxiety disorders. Eye Contact Lens. 2018;44(1):50-54.
6. Nordstrom BL, Friedman DS, Mozzaffari E, et al. Persistence and adherence with topical glaucoma therapy. Am J Ophthlamol. 2005;140(4):598-606.
7. Johnson TV, Jampel HD. Intraocular pressure following prerandomization glaucoma medication washout in the HORIZON and COMPASS trials. Am J Ophthalmol. 2020;216:110-120.
8. Neelakantan A, Vaishnav HD, Iyer SA, Sherwood MB. Is addition of a third or fourth antiglaucoma medication effective? J Glaucoma. 2004;13(2):130-136.
9. Malihi M, Filho ERM, Hodge DO, Sit AJ. Long-term trends in glaucoma-related blindness in Olmsted County, Minnesota. Ophthalmology. 2014;121(1):134-141.
10. Peters D, Bengtsson B, Heijl A. Lifetime risk of blindness in open angle glaucoma. Am J Ophthalmol. 2013;156(4):724-730.
11. Gazzard G, Konstantakopoulou E, Garway-Heath D, et al. Six-year results of primary selective laser trabeculoplasty versus eye drops for the treatment of glaucoma and ocular hypertension (LiGHT trial). Ophthalmology. 2023;130(2):139-151.
12. Samuelson TW, Sarkisian SR Jr, Lubeck DM, et al; iStent inject Study Group. Prospective, randomized, controlled pivotal trial of an ab interno implanted trabecular micro-bypass in primary open-angle glaucoma and cataract: two-year results. Ophthalmology. 2019;126(6):811-821.
INDICATIONS FOR USE: The iStent inject® is intended to reduce intraocular pressure safely and effectively in patients diagnosed with primary open-angle glaucoma, pseudo-exfoliative glaucoma or pigmentary glaucoma. The iStent inject® can deliver two (2) stents on a single pass, through a single incision. The implant is designed to stent open a passage through the trabecular meshwork to allow for an increase in the facility of outflow and a subsequent reduction in intraocular pressure. The device is safe and effective when implanted in combination with cataract surgery in those subjects who require intraocular pressure reduction and/or would benefit from glaucoma medication reduction. The device may also be implanted in patients who continue to have elevated intraocular pressure despite prior treatment with glaucoma medications and conventional glaucoma surgery. CONTRAINDICATIONS: The iStent inject® System is contraindicated under the following circumstances or conditions: • In eyes with primary angle closure glaucoma, or secondary angle-closure glaucoma, including neovascular glaucoma, because the device would not be expected to work in such situations • In patients with retrobulbar tumor, thyroid eye disease, Sturge-Weber Syndrome or any other type of condition that may cause elevated episcleral venous pressure. WARNINGS/PRECAUTIONS: • For prescription use only. • This device has not been studied in patients with uveitic glaucoma. • Do not use the devices if the Tyvek® lid has been opened or the packaging appears damaged. In such cases, the sterility of the device may be compromised. • Due to the sharpness of certain injector components (i.e. the insertion sleeve and trocar), care should be exercised to grasp the injector body. Dispose of device in a sharps container. • iStent inject® is MR-Conditional; see MRI Information below. • Physician training is required prior to use of the iStent inject® System. • Do not re-use the stent(s) or inserter, as this may result in infection and/or intraocular inflammation, as well as occurrence of potential postoperative adverse events as shown below under “Potential Complications.” • There are no known compatibility issues with the iStent inject® and other intraoperative devices (e.g., viscoelastics) or glaucoma medications. • Unused product & packaging may be disposed of in accordance with facility procedures. Implanted medical devices and contaminated products must be disposed of as medical waste. • Due to the sharpness of certain injector components (i.e. the insertion sleeve and trocar), care should be exercised to grasp the injector body • The surgeon should monitor the patient postoperatively for proper maintenance of intraocular pressure. If intraocular pressure is not adequately maintained after surgery, the surgeon should consider an appropriate treatment regimen to reduce intraocular pressure.
INDICATION FOR USE: The iStent inject® W, is intended to reduce intraocular pressure safely and effectively in patients diagnosed with primary open-angle glaucoma, pseudo-exfoliative glaucoma or pigmentary glaucoma. The iStent inject® W, can deliver two (2) stents on a single pass, through a single incision. The implant is designed to stent open a passage through the trabecular meshwork to allow for an increase in the facility of outflow and a subsequent reduction in intraocular pressure. The device is safe and effective when implanted in combination with cataract surgery in those subjects who require intraocular pressure reduction and/or would benefit from glaucoma medication reduction. The device may also be implanted in patients who continue to have elevated intraocular pressure despite prior treatment with glaucoma medications and conventional glaucoma surgery. CONTRAINDICATIONS: The iStent inject® W System is contraindicated under the following circumstances or conditions: • In eyes with primary angle closure glaucoma, or secondary angle-closure glaucoma, including neovascular glaucoma, because the device would not be expected to work in such situations. • In patients with retrobulbar tumor, thyroid eye disease, Sturge-Weber Syndrome or any other type of condition that may cause elevated episcleral venous pressure. WARNINGS/PRECAUTIONS: • For prescription use only. • This device has not been studied in patients with uveitic glaucoma. • Do not use the device if the Tyvek® lid has been opened or the packaging appears damaged. In such cases, the sterility of the device may be compromised. • Due to the sharpness of certain injector components (i.e. the insertion sleeve and trocar), care should be exercised to grasp the injector body. Dispose of device in a sharps container. • iStent inject® W is MR-Conditional; see MRI Information below. • Physician training is required prior to use of the iStent inject® W System. • Do not re-use the stent(s) or injector, as this may result in infection and/or intraocular inflammation, as well as occurrence of potential postoperative adverse events as shown below under “Potential Complications.” • There are no known compatibility issues with the iStent inject® W and other intraoperative devices. (e.g., viscoelastics) or glaucoma medications. • Unused product & packaging may be disposed of in accordance with facility procedures. Implanted medical devices and contaminated products must be disposed of as medical waste. • The surgeon should monitor the patient postoperatively for proper maintenance of intraocular pressure. If intraocular pressure is not adequately maintained after surgery, the surgeon should consider an appropriate treatment regimen to reduce intraocular pressure. • Patients should be informed that placement of the stents, without concomitant cataract surgery in phakic patients, can enhance the formation or progression of cataract. ADVERSE EVENTS: Please refer to Directions For Use for additional adverse event information. CAUTION: Please reference the Directions For Use labelling for a complete list of contraindications, warnings and adverse events.
© 2024 Glaukos Corporation. Glaukos, iStent inject® and iStent inject® W are registered trademarks of Glaukos Corporation. PM-EU-0308


