CASE PRESENTATION
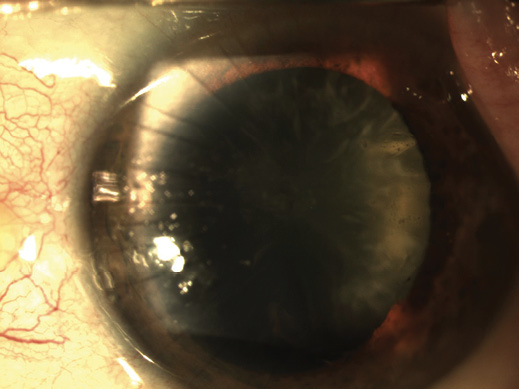
Figure 1. The patient has a history of multiincision RK. The cataract in his left eye exhibits nuclear sclerotic, cortical, and posterior subcapsular changes.
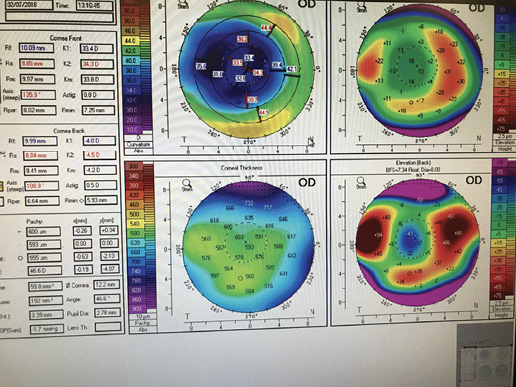
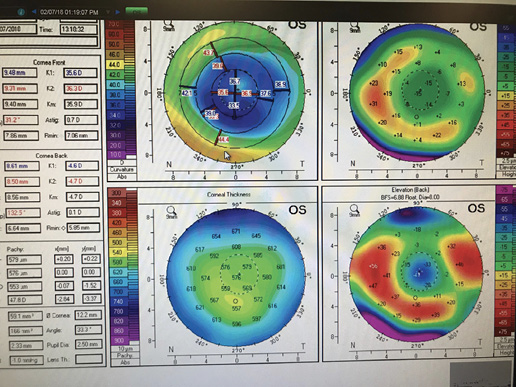
Figure 2. Measurements obtained with the Pentacam (Oculus Optikgeräte) for the patient’s right (A) and left (B) eyes.
—Case prepared by Audrey R. Talley Rostov, MD

DOUGLAS A. KATSEV, MD
To provide the overall best long-term quality of vision to patients who have compromised corneal architecture, I rely heavily on a lens optic that does not add aberrations. Specifically, I have found Bausch + Lomb’s line of Advanced Optics (AO) IOLs to work well in these abnormal eyes. Extensive lifestyle profiling of patients greatly assists with IOL selection. For me, the only premium option in these cases is the Crystalens AO. This IOL can extend patients’ depth of field and decrease their dependence on glasses without inducing aberrations.
Before entering the OR, it would be important to discuss with this patient his potentially limited visual outcome secondary to previous eye surgery. He should have a say in whether to consider a premium IOL.
If applicable, I would instruct the patient to discontinue contact lens wear—1 week for soft lenses and 1 week per decade of use for rigid gas permeable lenses. It would also be important to discuss visual fluctuations with the patient preoperatively. Performing biometry and keratometry (K) in the morning and again in the afternoon would help identify migration of the K values. For IOL power calculations, I favor the Hoffer Q formula but consider all data. I would target a refraction of -0.50 to -0.75 D. At present, I do not use intraoperative aberrometry.
I would avoid using a femtosecond laser in this case. Instead, I would carefully create the clear corneal incision manually and avoid excessive stromal hydration in an effort not to disrupt the RK scars. I would use lower overall flow, vacuum, and ultrasound power than my usual levels and match the sizes of those entry wounds to that of the instruments. Surgeon control will be key in this case. At the conclusion of surgery, I would suture the cataract incisions to ensure a watertight closure.
Although post-RK patients are some of my most grateful, that might not be the case here with one procedure; PRK or a piggyback IOL might be needed to achieve the desired refractive result. It would be important to counsel the patient that his early postoperative vision will not be his final result; in a post-RK eye, vision can take weeks to stabilize after cataract surgery.

SHERI ROWEN, MD
In the past, when RK patients presented for cataract evaluation, I used to feel like I was throwing linguine against the wall and hoping something would stick. Today, with new diagnostics and formulas, there is a much better chance of success despite an unstable cornea. Another thing that will help in this case is hindsight. The K readings in the patient’s right eye are extremely flat—about 3.00 D flatter than in his left eye. Normally, this would have been a setup for a hyperopic outcome, but the surgeon was able to compensate and achieve excellent near vision in the patient’s right eye. The K readings are now approximately 33.00 D OD and 37.00 D OS, but the central cornea might well be flatter than what can be measured.
My first step would be to find out the power of the IOL implanted in the patient’s right eye and to determine how close the outcome would have been with the formula I plan to use. I would keep in mind the necessity of adding at least 2.00 D to any power an older traditional IOL formula provides to avoid residual hyperopia in a postmyopic RK eye. I therefore routinely use the ASCRS and Holladay 2 formulas to guide IOL power selection in post-RK eyes. I would try the ORA System (Alcon) in this case, but I would expect image quality to be insufficient in an eye with so many RK cuts. It would be perfectly appropriate to select a monofocal IOL and to target distance vision in the patient’s left eye for a monovision outcome. That said, I have been able to achieve remarkable outcomes in multifocal corneas using the Crystalens platform IOLs. The plano aspheric optics of the Crystalens AO, Trulign Toric Posterior Chamber IOL (Bausch + Lomb), or the enVista IOL (Bausch + Lomb) do not add significant internal aberrations, so any of these implants would be an appropriate option in this case. My preference would be the Crystalens AO.
The chances of not encountering one of 32 RK cuts while making the clear corneal incision with a keratome are about nil, so I would manually create a scleral or very posterior limbal incision in an effort not to split an RK incision. Even so, I would be prepared to repair a split RK incision with interrupted 10-0 nylon sutures; in my experience, the eye will heal without additional astigmatism. I never use a femtosecond laser for corneal incisions in post-RK eyes, but I have used the technology for the capsulotomy and lens fragmentation in this setting.
When implanting a Crystalens, I always insert a capsular tension ring to stabilize the refractive outcome and to prevent fibrosis and Z syndrome. I would avoid a multifocal or extended depth of focus IOL in this case, owing to the significant corneal aberrations and because reentry into the eye to address any dysphotopsia could prove quite difficult. When it becomes commercially available in the United States, the Light Adjustable Lens (RxLAL, RxSight) will be a great monofocal lens option for correcting residual refractive error.


TIM SCHULTZ, MD; AND H. BURKHARD DICK, MD, PhD
Because of the highly irregular cornea, we would use the ray-tracing Okulix software on the Pentacam to calculate the IOL power. With this software, we could adjust the optical zone to the regular central part of the cornea. Otherwise, the irregular outer zone would be overrepresented in the calculation. We would also use the Verion Image Guided System (Alcon [former ORA system]) and ORA System during surgery to confirm our preoperative measurements using the integrated RK adjustment module.1
For several reasons, our lens preference in this case would be the IC-8 IOL (AcuFocus). First, this pinhole implant can compensate for up to 1.50 D of astigmatism and corneal aberrations while reducing stray light. Second, it is possible to achieve good near vision with this IOL. Third, intraoperative complications such as corneal perforation are possible in this case, which would require suturing, and the IC-8 IOL would offer a large refractive landing area, forgiving of a deviation from the targeted refraction. Finally, our experience with this IOL in highly aberrated eyes has been positive thus far.2
Another option for this patient would be the RxLAL. With this technology, the surgeon could easily adjust the IOL power and astigmatic correction after implantation within a range of 2.00 to 2.50 D. We would avoid an extended depth of focus or multifocal IOL in this case.
In post-RK eyes, we use a femtosecond laser (Catalys Precision Laser System, Johnson & Johnson Vision) to perform the capsulotomy and lenticular fragmentation, but the settings must be adjusted. We decrease the grid spacing of the fragmentation as much as possible to facilitate lens removal (aspiration only). For the capsulotomy, we increase the pulse energy to 8 μJ to lessen the risk of blocked laser shots. So as to be prepared for an incomplete cut, we recommend the dimple-down maneuver and the use of trypan blue ophthalmic solution (VisionBlue, Dutch Ophthalmic). We also instill a high-viscosity OVD such as Healon5 (Johnson & Johnson Vision) to stabilize the anterior chamber, especially in cases of corneal perforation.
In eyes that have eight or 16 RK cuts, the side and main cataract incisions can be positioned between the RK cuts. In this case, we would create the main incision in the posterior limbus. We like the Blumenthal single-use knife (Geuder) for a bimanual approach. We would make the sideport incisions at the 10- and 2-o’clock positions.
During intraoperative aberrometry, we recommend the use of Healon (Johnson & Johnson Vision) because it has the same refractive index as balanced salt solution.

PRIYANKA SOOD, MD
One thing going for this patient is that he has already undergone cataract surgery on one eye, and those outcomes can be used when planning cataract surgery on the second eye. I would try to obtain the biometry results from the first cataract surgeon and use that information to assist with IOL power calculations. If that information were not available, I would rely on the IOL Calculator for Eyes With Prior RK that is available on the ASCRS website. I would aim for -0.50 D because these eyes can have a tendency toward hyperopic fluctuations. I would plan to use an aspheric IOL to offset the typically increased spherical aberration of a post-RK cornea.
Given how tight the 32 RK incisions are in this case, the safest approach would be a scleral tunnel incision. Trypan blue dye would be useful because posterior subcapsular and cortical changes and the RK scars will make visualizing the anterior capsule more challenging.
Although a femtosecond laser could be used to make the capsulotomy and soften the lens, I would not use it to create the corneal incisions. I would also avoid overfilling the anterior chamber with balanced salt solution and OVD because the RK cuts will be prone to opening under minimal pressure. As a precaution, I would have sutures readily available during surgery. I would not use intraoperative aberrometry in this case because the technology’s utility has not been demonstrated for post-RK eyes.
Postoperatively, post-RK patients tend to experience fluctuating refractive results because of the transient swelling and concurrent flattening of the central cornea. It would therefore be important to inform this patient preoperatively that up to 3 months may elapse before his vision stabilizes and to explain that any adjustments (PRK, piggyback IOL, or an IOL exchange) will have to wait until then.

WHAT I DID: AUDREY R. TALLEY ROSTOV, MD
I used the ASCRS online calculator and targeted a postoperative refraction of -2.00 D. As part of informed consent, I counseled the patient that he should expect to wear glasses full-time after surgery.
I used the Lensar Laser System (Lensar) to perform the capsulotomy and fragment the lens. I used a keratome to create posterior limbal cataract incisions and performed bimanual cataract surgery. As I had anticipated, areas of the laser capsulotomy were incomplete because of the RK incisions, but I was nevertheless able to remove the capsulotomy easily with capsulorhexis forceps (MicroSurgical Technology). Hydrodelineation, hydrodissection, and phacoemulsification proceeded without complication, although some of the RK incisions splayed open temporally.
I selected an enVista IOL (model MX60) for its zero spherical aberration, and the lens was easily implanted in the capsular bag. Multiple 10-0 nylon sutures were needed to close the incision sites, and I applied ReSure Sealant (Ocular Therapeutix) to further secure the wounds.
Although the patient had induced cylinder secondary to the multiple sutures immediately after surgery, her final postoperative refraction was -2.00 +1.00 × 005º when I removed the sutures 4 weeks later.
1. Canto AP, Chhadva P, Cabot F, et al. Comparison of IOL power calculation methods and intraoperative wavefront aberrometer in eyes after refractive surgery. J Refract Surg. 2013;29(7):484-489.
2. Schultz T, Dick HB. Small-aperture intraocular lens implantation in a patient with an irregular cornea. J Refract Surg. 2016;32(10):706-708.



