Sponsored by ZEISS

DR CATRIONA A HAMER
Lecturer in Optometry (Honorary Researcher at the Royal Eye Infirmary, Plymouth)
Dr Catriona Hamer completed her PhD at Plymouth University before beginning a lectureship there in 2015. She is part of a small team of academics who worked to achieve accreditation of the new Optometry programme established there. She has worked across several modules within the programme. Catriona’s research areas include assessment and correction of corneal astigmatism in cataract surgery as well as the relationship between visual correction and stability in older adults. She has presented and published work in Europe and the USA.

DR PHILLIP J BUCKHURST
Associate Professor (Senior Lecturer) in Optometry
Dr Phillip Buckhurst completed his PhD at Aston University before moving to Plymouth University in 2011 as Associate Professor. He was part of a team of academics who designed and developed the Plymouth University Optometry programme. Phillip’s research interests are mainly focused on the assessment of intraocular lenses and ocular aberrations. He is an active researcher in clinical and laboratory-based research and has been involved in numerous multicentre clinical trials from design to delivery.

DR HETAL BUCKHURST
Associate Professor (Senior Lecturer) in Optometry
In 2011, Hetal joined the inaugural team of academics tasked with the setup and development of the Optometry BSc (Hons) programme at Plymouth University. As part of her role she has developed and taught several modules across the three years of the programme. Hetal’s research areas are mainly related to scleral and ocular rigidity in humans and its possible links with myopia. She has presented her research internationally in USA, Japan and Europe and has authored a number of papers on the subject. In 2014, Hetal was awarded the prestigious College of Optometrist fellowship grant to further support her research.
Over the last century, the progressive improvement in cataract surgery has evolved the operation beyond the mere replacement of the clouded crystalline lens. There is an expectation by both practitioner and patient that emmetropia will be achieved post-operatively. Despite the common practice to correct residual refractive error during cataract surgery, there are challenges in predicting the post-operative outcomes and correction required. The main source of variability arises from the surgically induced corneal shape change that results in surgical induced astigmatism (SIA).
It is generally accepted that the incision causes a flattening effect along the orientation in which it is applied. If the incision is placed oblique to the steepest corneal meridian then there is a subsequent torsional effect on the pre-existing astigmatism. These curvature changes result in significant alterations to the magnitude and orientation of the principal meridians post-surgically.1-3 The impact of such changes is amplified in individuals with lower levels of astigmatism. As surgeons aim to correct ever-lower levels of astigmatism the influence of SIA is becoming more significant.
Accurate assessment of SIA has been a key component in the improvement of the surgical technique. Incision characteristics such as, size and location can alter magnitude and orientation of the SIA. It is well established that the magnitude of SIA assessed after surgery can be reduced by decreasing the size of the incision,4-7 using a temporal location rather than a superior location8-12 and using suture-less incision, at the cornea.13-15 However, recent work has disputed the effect of wound size on SIA for incision <3mm in size.4,16-20 In the papers that found statistical significance, the difference reported would not be considered clinically significant.
If the size and orientation of SIA is predictable, pre-surgical consideration of these clinical variables will allow the surgeon to plan a more accurate correction of the pre-existing corneal astigmatism by one of two main options. Firstly, in low levels of astigmatism (<1.00DC), the choice of incision size and location could be used to reduce pre-existing low levels of astigmatism, or at the very least, avoid increasing it. Secondly, in higher levels of astigmatism (>1.00DC) when implanting toric IOLs, the surgeon can attempt to correct the full post-operative astigmatism. This latter approach has led to the development of the wide range of commercially available toric IOL calculators.
There are two main difficulties with assessing corneal astigmatism change. The first is that there are two components – the magnitude and the axis (orientation) of the astigmatism. In addition to this, the axis data is circular in nature but runs from 0 – 180. Therefore, assessment of the data cannot be carried out with standard mathematic conventions. In order to avoid these difficulties, the most common solution is to combine the data, using vectors to describe the induced astigmatic change. This method is widely known as the surgically induced refractive change (SIRC) formula1 and has been commonly used for calculating SIA. The analysis in SIRC is based upon assessment of corneal change in the form of vector magnitude differences. Despite its popularity, the formula is an over-simplification of post-operative SIA changes as it disregards axis. Applying trigonometry, Jaffe and Clayman21 and later Holladay et al1 developed a series of equations that could calculate both the magnitude and the orientation of SIA.21-24 Despite their popularity, these formulae still provide a global generalisation of post-operative SIA changes.
The polar method is an alternative method of analysis that describes the shape change across two perpendicular directions and hence summarise the flattening and the torsional effect of an incision.22,25 A more recent trend for analysis and displaying SIA results has been to utilise a double angle polar plot to describe the magnitude and axis of the corneal change.26
Principals of SIA calculations
Vector Calculation of SIA
All results are converted into rectangular Fourier form of mean spherical equivalent (MSE) and Jackson Cross-Cyl vectors representing the cylindrical power and axis as a combined vector for analysis (Figure 1a).27,28
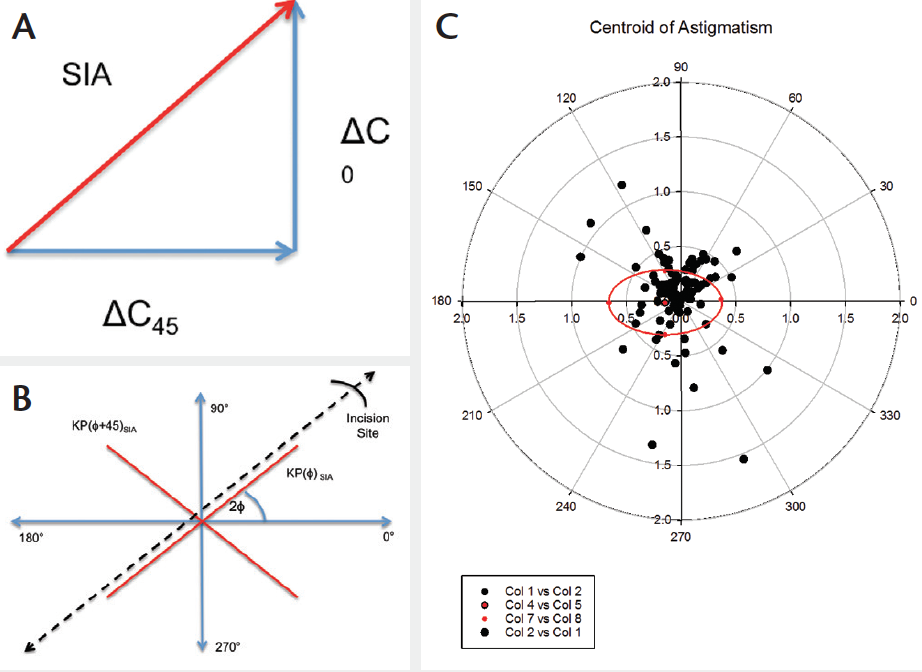
Figure 1: Example of three models of SIA calculations: a. Vector calculation. b is Polar method. c. Double angle polar plot with the centroid and standard deviation marked
Polar Calculation of SIA
This method calculates the change in power along a specific reference orientation. Polar analysis describes the power change both parallel and perpendicular to this orientation.22 Hence using this method, you can examine the flattening effect along the incision meridian and any resultant torsion (Figure 1b).
Double Polar Plots and Centroid of Astigmatism
A double Polar plot can be used to illustrate the differences between the pre and post-operative corneal astigmatic powers. The mean difference can be assessed by examining the centroids of each of these plots (Figure 1c).26
Sources of Error
The accurate prediction of SIA is of significant clinical value and there is still much research that is required to address the fundamental assumptions in current SIA calculation. The following section discusses the potential sources of error that are often overlooked.
Corneal Stability
At present there is significant ambiguity concerning the time taken for stabilisation of the corneal shape following surgery. It has been reported to stabilise after just 2 weeks,29 however some reports indicate change in the SIA up to a year later.30,31 Nevertheless, it is important to realise that the corneal shape will continue to change with time and age irrespective of surgery. The steepest corneal meridian can rotate from a vertical position (with the rule) towards a horizontal orientation (against the rule)32 and as such the surgeon may be chasing a moving target, confusing the matter further.
Accuracy of Keratometry
It is commonly assumed that the keratometry readings upon which SIA is calculated are free from error. Yet, most keratometry validation papers report a small level of error in the repeatability of readings especially when describing astigmatism, which are significant when compared to the low levels of SIA reported in modern surgeries.33-36 Mean test-to-test variability of instruments has been reported as 0.20 - 0.46D.37,38 Such variation will no doubt affect the accuracy of the calculated SIA. This may contribute to the conflicting reports on SIA with sub-3mm incisions discussed earlier.
Standard values for SIA
Surgically induced astigmatism is widely reported on and the general trends and influences of the incision type and size are well established. However, there is poor agreement on the specific resultant value induced by specific incision size (e.g. 3mm, see Table 1). Multiple other factors, such as location, surgical technique and delivery system of the lens may all have a significant influence. Using an assumed SIA without calculating it for the specific surgeon will add to the errors in the predictive calculations.
Assessing astigmatic axis
Due to the circular nature of the astigmatic axis, the data is difficult to interpret and assess. This has led to the axis being overlooked or confused in the vector calculations used to determine SIA. Furthermore, SIA is often expressed as a single value; by oversimplifying the data in this manner, much of the information regarding corneal change is lost. It is generally assumed that the vector change in SIA is equal for both an off-axis and on-axis incision: the SIA with an off-axis incision is assumed to translate to torsion rather than simple magnitude reduction. The effect of orientation is further complicated by the observation that incisions placed “with the rule” have a larger flattening effect when compared to those placed “against the rule”.
Biomechanics
Corneal biomechanical properties vary across the population.39-41 There is a negative correlation between pre-surgical hysteresis and SIA, with high corneal hysteresis associated with lower SIA.42 Studies have shown corneal hysteresis can significantly change following surgery, with high corneal hysteresis associated with lower SIA. These changes have been noted to recover with time, although the recovery time varies with incision type, location and size.43-45 This will in turn affect the SIA reading, depending upon the time after surgery the cornea was assessed.
The Calculations
One of the fundamental assumptions upon which all SIA calculations are based, is the fact that the resultant optical power produced by the interaction of the cornea and an incision is similar to that of a thin optical lens system. This is contrary to the reality of the biological and dynamic nature of the corneal tissue.
Although there has been extensive research in the dioptric power change that occurs to the cornea post-surgically,3,8,22,26,46,47 to date no studies have examined the predictability and consistency of the corneal axis changes. Toric calculators utilising SIA values predict that the steepest corneal meridian will move away from incision site. However, unpublished work from our team found that when 144 corneas (n = 144) were examined pre-and post- cataract surgery, an equal amount moved towards the incision, not away from the incision.38
If we acknowledge the many sources of error, we can work to minimise their influence on SIA and it’s use in the future. There are a few simple considerations that could be used to improve the accuracy and reduce the influence of some errors.
Hints and Tips
1. Keratometry readings
By repeating readings, quantifying the repeatability error and paying closer attention to the axis in relation to head and eye position can improve the accuracy of keratometry. Using the average of multiple readings and even instruments will help to increase the accuracy, compared to a single reading. Also, if the instrument error is quantified, the SIA readings can be adjusted to create a more accurate assessment of actual change. The influence of axis error can be greater than that of magnitude in the vector calculations and hence, careful head positioning and eye tracking will help to minimise the chance of rotation between readings.
2. Assessing axis position
The spread of the data should be assessed when calculating average SIAs, paying particular attention to the axis changes and direction changes. This will give a better understanding of the influence of the incisions. Looking at an average magnitude alone will offer limited information. In the future better understanding of biomechanics will help produce better predictors of corneal shape change and healing.
3. When using a Toric IOLs place your incisions “on-axis”
Toric IOL calculators use simple vector analysis to determine the location of the post-operative steepest meridian. These calculators will always predict that the post-operative steepest meridian position moves away from the incision location (when the incision is placed off-axis). However, the torsional effects of incisions are not this predictable and hence this method of analysis can lead to the misalignment of toric IOLs. Therefore, where possible a clear corneal incision should be placed along the steepest meridian (“on axis”) so that the oblique calculation is not required.
1. Holladay JT, Cravy TV, Koch DD. Calculating the Surgically Induced Refractive Change Following Ocular Surgery. J. Cataract Refract. Surg. Sep 1992;18(5):429-443.
2. Naeser K. Combining refractive and topographic data in corneal refractive surgery for astigmatism. Acta Ophthalmol. (Copenh.). Dec 2012;90(8):768-772.
3. Alpins N. Astigmatism analysis by the Alpins method. J. Cataract Refract. Surg. Jan 2001;27(1):31-49.
4. Yu YB, Zhu YN, Wang W, Zhang YD, Yu YH, Yao K. A comparable study of clinical and optical outcomes after 1.8, 2.0 mm microcoaxial and 3.0 mm coaxial cataract surgery. International journal of ophthalmology. 2016;9(3):399-405.
5. Dewey S, Beiko G, Braga-Mele R, Nixon DR, Raviv T, Rosenthal K. Microincisions in cataract surgery. J. Cataract Refract. Surg. Sep 2014;40(9):1549-1557.
6. Ale JB. Surgically induced astigmatism of small incision cataract surgery. Nepalese journal of ophthalmology : a biannual peer-reviewed academic journal of the Nepal Ophthalmic Society : NEPJOPH. Jul-Dec 2012;4(2):343.
7. Wei YH, Chen WL, Su PY, Shen EP, Hu FR. The influence of corneal wound size on surgically induced corneal astigmatism after phacoemulsification. J. Formos. Med. Assoc. May 2012;111(5):284-289.
8. Yoon JH, Kim KH, Lee JY, Nam DH. Surgically induced astigmatism after 3.0 mm temporal and nasal clear corneal incisions in bilateral cataract surgery. Indian J. Ophthalmol. Jun 2014;62(6):753.
9. Altan-Yaycioglu R, Akova YA, Akca S, Gur S, Oktem C. Effect on astigmatism of the location of clear corneal incision in phacoemulsification of cataract. J. Refract. Surg. May 2007;23(5):515-518.
10. Ozyol E, Ozyol P. Analyses of surgically induced astigmatism and axis deviation in microcoaxial phacoemulsification. Int. Ophthalmol. Jun 2014;34(3):591-596.
11. Visser N, Ruiz-Mesa R, Pastor F, Bauer NJ, Nuijts RM, Montes-Mico R. Cataract surgery with toric intraocular lens implantation in patients with high corneal astigmatism. J. Cataract Refract. Surg. Aug 2011;37(8):1403-1410.
12. Reddy B, Raj A, Singh VP. Site of Incision and Corneal Astigmatism in Conventional SICS versus Phacoemulsification. Ann. Ophthalmol. Sep 20 2007;39(3):209-216.
13. Linebarger EJ, Hardten DR, Shah GK, Lindstrom RL. Phacoemulsification and modern cataract surgery. Surv. Ophthalmol. 1999;44(2):123-147.
14. El-Kasaby HT, McDonnell PJ, Deutsch J. Videokeratography: a comparison between 6 mm sutured and unsutured incisions for phacoemulsification. Eye (London, England). February 1 1995;9 ( Pt 6):719-721.
15. Mendivil A. Frequency of induced astigmatism following phacoemulsification with suturing versus without suturing. Ophthalmic Surg. Lasers. May 1997;28(5):377-381.
16. Sethi HS, Saluja K, Naik MP. Comparative analysis of coaxial phacoemulsification with 2.2- and 2.8-mm clear corneal incisions. Int. Ophthalmol. Jan 31 2017.
17. Febbraro JL, Wang L, Borasio E, et al. Astigmatic equivalence of 2.2-mm and 1.8-mm superior clear corneal cataract incision. Graefes Arch. Clin. Exp. Ophthalmol. Feb 2015;253(2):261-265.
18. Luo L, Lin H, He M, Congdon N, Yang Y, Liu Y. Clinical evaluation of three incision size-dependent phacoemulsification systems. Am. J. Ophthalmol. May 2012;153(5):831-839 e832.
19. Chang SW, Su TY, Chen YL. Influence of ocular features and incision width on surgically induced astigmatism after cataract surgery. J. Refract. Surg. Feb 2015;31(2):82-88.
20. Klamann MK, Gonnermann J, Maier AK, Torun N, Bertelmann E. Smaller incision size leads to higher predictability in microcoaxial cataract surgery. Eur. J. Ophthalmol. Mar-Apr 2013;23(2):202-207.
21. Jaffe NS, Clayman HM. The Pathophisiology of Corneal Astigmatism after Cataract Extraction. Trans. Am. Acad. Ophthalmol. Otolaryngol. 1975;79:615-630.
22. Naeser K. Assessment and statistics of surgically induced astigmatism. Acta Ophthalmol. (Copenh.). May 2008;86 Suppl 1(thesis1):5-28.
23. Alpins NA. Vector analysis of astigmatism changes by flattening, steepening, and torque. J. Cataract Refract. Surg. Dec 1997;23(10):1503-1514.
24. Alpins NA. A new method of analyzing vectors for changes in astigmatism. J. Cataract Refract. Surg. Jul 1993;19(4):524-533.
25. Cravy TV. Calculation of the change in corneal astigmatism following cataract extraction. Ophthalmic Surg. Jan 1979;10(1):38-49.
26. Holladay JT, Moran JR, Kezirian GM. Analysis of aggregate surgically induced refractive change, prediction error, and intraocular astigmatism. J. Cataract Refract. Surg. Jan 01 2001;27(1):61-79.
27. Thibos LN, Wheeler W, Horner D. Power vectors: An application of Fourier analysis to the description and statistical analysis of refractive error. Optom. Vis. Sci. Jun 1997;74(6):367-375.
28. Hill W. Expected effects of surgically induced astigmatism on AcrySof toric intraocular lens results. J. Cataract Refract. Surg. 2008;34(3):364-367.
29. Masket S, Tennen DG. Astigmatic stabilization of 3.0 mm temporal clear corneal cataract incisions. J. Cataract Refract. Surg. 1996;22(10):1451-1455.
30. Pfleger T, Skorpik C, Menapace R, Scholz U, Weghaupt H, Zehetmayer M. Long-term course of induced astigmatism after clear corneal incision cataract surgery. J. Cataract Refract. Surg. Jan 1996;22(1):72-77.
31. Ermiş SS, Inan UU, Oztürk F. Surgically induced astigmatism after superotemporal and superonasal clear corneal incisions in phacoemulsification. J. Cataract Refract. Surg. May 31 2004;30(6):1316-1319.
32. Hayashi K, Hayashi H, Hayashi F. Topographic analysis of the changes in corneal shape due to aging. Cornea. Sep 1995;14(5):527-532.
33. Dehnavi Z, Khabazkhoob M, Mirzajani A, Jabbarvand M, Yekta A, Jafarzadehpur E. Comparison of the Corneal Power Measurements with the TMS4-Topographer, Pentacam HR, IOL Master, and Javal Keratometer. Middle East African Journal of Ophthalmology. Apr-Jun 2015;22(2):233-237.
34. Viswanathan D, Kumar NL, Males JJ, Graham SL. Comparative analysis of corneal measurements obtained from a Scheimpflug camera and an integrated Placido-optical coherence tomography device in normal and keratoconic eyes. Acta Ophthalmol. (Copenh.). Dec 14 2014.
35. Hamer CA, Buckhurst H, Purslow C, Shum GL, Habib NE, Buckhurst PJ. Comparison of reliability and repeatability of corneal curvature assessment with six keratometers. Clin. Exp. Optom. Nov 2016;99(6):583-589.
36. Asgari S, Hashemi H, Jafarzadehpur E, Mohamadi A, Rezvan F, Fotouhi A. OPD-Scan III: a repeatability and inter-device agreement study of a multifunctional device in emmetropia, ametropia, and keratoconus. Int. Ophthalmol. Feb 8 2016.
37. Goggin M, Patel I, Billing K, Esterman A. Variation in surgically induced astigmatism estimation due to test-to-test variations in keratometry. J. Cataract Refract. Surg. Oct 2010;36(10):1792-1793.
38. Hamer C, Buckhurst H, Shum G, Purslow C, Buckhurst PJ. Analysis and Correction of Corneal Astigmatism in Modern Pseudophakia. Plymouth, UK: Optometry, School of Health Professions, Plymouth University; 2016.
39. Lazreg S, Mesplié N, Praud D, et al. Comparison of corneal thickness and biomechanical properties between North African and French patients. J. Cataract Refract. Surg. Mar 2013;39(3):425-430.
40. Elsheikh A, Wang D, Rama P, Campanelli M, Garway-Heath D. Experimental assessment of human corneal hysteresis. Curr. Eye Res. Mar 2008;33(3):205-213.
41. McMonnies CW. Assessing Corneal Hysteresis Using the Ocular Response Analyzer. Optom. Vis. Sci. Mar 2012;89(3):E343-E349.
42. Denoyer A, Ricaud X, Van Went C, Labbé A, Baudouin C. Influence of corneal biomechanical properties on surgically induced astigmatism in cataract surgery. J. Cataract Refract. Surg. Aug 2013;39(8):1204-1210.
43. Kucumen RB, Yenerel NM, Gorgun E, et al. Corneal biomechanical properties and intraocular pressure changes after phacoemulsification and intraocular lens implantation. J. Cataract Refract. Surg. Dec 2008;34(12):2096-2098.
44. Kandarakis A, Soumplis V, Karampelas M, et al. Response of corneal hysteresis and central corneal thickness following clear corneal cataract surgery. Acta Ophthalmol. (Copenh.). Sep 2012;90(6):526-529.
45. Alio JL, Agdeppa MC, Rodriguez-Prats JL, Amparo F, Pinero DP. Factors influencing corneal biomechanical changes after microincision cataract surgery and standard coaxial phacoemulsification. J. Cataract Refract. Surg. Jun 2010;36(6):890-897.
46. Wei Y-H, Chen W-L, Su P-Y, Shen EP, Hu F-R. The influence of corneal wound size on surgically induced corneal astigmatism after phacoemulsification. J. Formos. Med. Assoc. May 2012;111(5):284-289.
47. Ozyol E, Ozyol P. The relation between superior phacoemulsification incision and steep axis on astigmatic outcomes. Int. Ophthalmol. Dec 2012;32(6):565-570.





