What are some of the biggest changes in glaucoma surgical management over the past 10 years?
Several new surgical treatments have been introduced under the category of MIGS procedures. The goal of MIGS is to increase the aqueous outflow while minimally disrupting the ocular anatomy. This group of procedures has a better safety profile and faster visual recovery than conventional filtering surgeries, and they can be used in different stages of the disease. At one end of the spectrum, newer bleb-forming procedures as well as canaloplasty and gonioscopy-assisted transluminal trabeculotomy procedures can be beneficial when the outflow system no longer functions properly. At the other end, trabecular micro-bypass procedures such as iStent inject® W (Glaukos) provide an overall safety profile similar to cataract surgery alone,1 giving surgeons the confidence to intervene surgically much earlier in the disease pathway.
What place does trabecular micro-bypass surgery have in the management of glaucoma?
One major benefit of trabecular micro-bypass surgery is that it can be performed at the time of cataract surgery, when the surgeon is already entering the eye. Not only does the surgical risk decrease, but so does the patient’s inconvenience. Patients with mild to moderate open-angle glaucoma (OAG) controlled with medical topical treatments are ideal candidates for trabecular micro-bypass surgery.1,2 The subsequent IOP decrease can lead to a reduction in the number of topical hypotensive medications, at the clinician’s discretion, reducing patients’ out of pocket costs and the inconvenience of the medical treatment, as well as improving their quality of life.3
Patients with early to moderate glaucoma who are noncompliant or intolerant to topical drops are also ideal cases for a trabecular micro-bypass surgery. Evidence shows comparable efficacy and safety of iStent technology in primary open-angle glaucoma (POAG) and pigmentary or pseudoexfoliative glaucoma.4
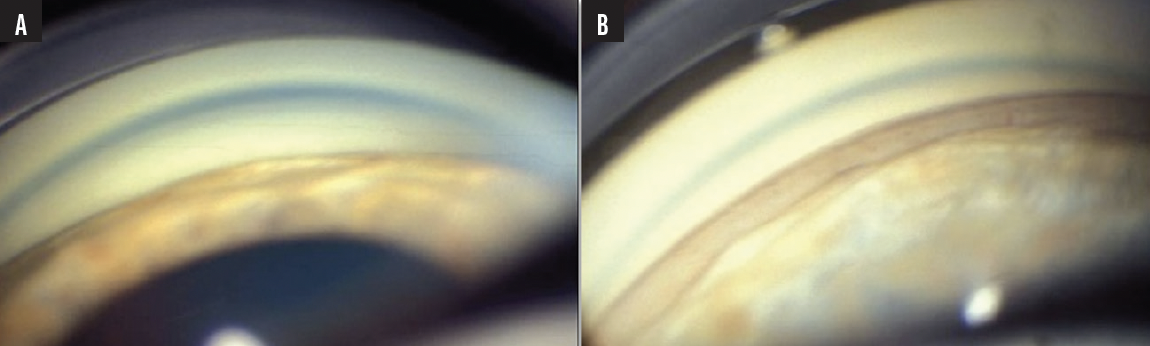
Figure. Gonioscopy examination before cataract surgery showing angle closure with a circumferential apposition of the iris against the trabecular meshwork but no peripheral angle synechia (A) and a large opening of the angle with clear visibility of the trabecular meshwork and the scleral spur in the inferior and nasal quadrants of the angle, making an implantation of iStent inject® W possible (B).
What has been your experience with iStent inject® W?
Although the iStent inject® W is indicated for mild to moderate POAG—and most of my experience is in these patients—occasionally I have used the device in more complex open-angle cases. Several clinical studies have evaluated the efficacy and safety of some trabecular micro-bypass procedures, particularly the iStent, in other forms of more complex open-angle and angle-closure glaucoma.2,5

In my opinion, the safety profile of the iStent inject® W makes it an appealing option in patients with progressing OAG with a high risk of complications from filtering surgeries (central or paracentral visual field defects, highly myopic eyes, poor general conditions, anticoagulants and antiaggregant treatments). The efficacy may be less than a trabeculotomy, but the safety profile is much better, and this makes it a valuable choice when there is a need to avoid complications.
I have also had good results in patients with appositional angle closure who do not have peripheral angle synechia. Following cataract surgery and subsequent opening of the irido-corneal angle, studies have shown a greater IOP reduction after iStent implantation compared to that provided by the cataract extraction alone.5,6
Clinical case
A 78-year-old man, initially treated with a topical hypotensive treatment for an established POAG that subsequently developed a progressively narrowing angle, which became crowded as a result of an enlarging cataract, presented to my clinic. He had an IOP of about 25 mm Hg with a bi-therapy of prostaglandin analogs and beta-blockers in 2023, as well as progression of visual field defects. The patient had several cardiovascular diseases, with some anticoagulants and antiaggregant treatments that could not be stopped.
We wanted to significantly reduce the IOP with a simple one-step surgery and to avoid the need for a subsequent filtering surgery. Some studies have shown that a combined cataract surgery with iStent implantation could potentially be more effective than cataract surgery alone to reduce the IOP and/or the hypotensive medications in this type of patient. So, we decided to do a preoperative gonioscopy examination just after the lens extraction; the goal, if possible, was to implant two iStent inject W devices (see Figure 1). This was done without technical difficulties.
The patient's IOP is now between 12 and 15 mm Hg with the same hypotensive medications and without visual field defects progression.
1. Samuelson TW, Sarkisian SR Jr, Lubeck DM, et al. Prospective, randomized, controlled pivotal trial of an ab interno implanted trabecular micro-bypass in primary open-angle glaucoma and cataract: two-year results. Ophthalmology. 2019;126(6):811-821.
2. Kahale F, Chanbour W, El Zein L, Brenner J, You JY, Melki S. Phacoemulsification with and without iStent: a systematic review and meta-analysis of comparative studies. Ophthalmic Res. 2023. doi: 10.1159/000531077. Online ahead of print.
3. Schweitzer JA, Hauser WH, Ibach M, et al. Prospective interventional cohort study of ocular surface disease changes in eyes after trabecular micro-bypass Stent(s) implantation (iStent or iStent inject) with phacoemulsification. Ophthalmol Ther. 2020;9(4):941-953.
4. Ferguson TJ, Swan R, Ibach M, Schweitzer J, Sudhagoni R, Berdahl JP. Trabecular microbypass stent implantation with cataract extraction in pseudoexfoliation glaucoma. J Cataract Refract Surg. 2017;43(5):622-626.
5. Salimi A, Abu-Nada M, Harasymowycz P. Matched cohort study of cataract surgery with and without trabecular microbypass stent implantation in primary angle-closure glaucoma. Am J Ophthalmol. 2021;224:310-320.
6. Hernstadt DJ, Cheng J, Htoon HM, Sangtam T, Thomas A, Sng CCA. Case series of combined iStent implantation and phacoemulsification in eyes with primary angle closure disease: one-year outcomes. Adv Ther. 2019;36(4):976-986.
iStent inject® W IMPORTANT SAFETY INFORMATION
INDICATION FOR USE: The iStent inject® W, is intended to reduce intraocular pressure safely and effectively in patients diagnosed with primary open-angle glaucoma, pseudo-exfoliative glaucoma or pigmentary glaucoma. The iStent inject® W, can deliver two (2) stents on a single pass, through a single incision. The implant is designed to stent open a passage through the trabecular meshwork to allow for an increase in the facility of outflow and a subsequent reduction in intraocular pressure. The device is safe and effective when implanted in combination with cataract surgery in those subjects who require intraocular pressure reduction and/or would benefit from glaucoma medication reduction. The device may also be implanted in patients who continue to have elevated intraocular pressure despite prior treatment with glaucoma medications and conventional glaucoma surgery. CONTRAINDICATIONS: The iStent inject® W System is contraindicated under the following circumstances or conditions: • In eyes with primary angle closure glaucoma, or secondary angle-closure glaucoma, including neovascular glaucoma, because the device would not be expected to work in such situations. • In patients with retrobulbar tumor, thyroid eye disease, Sturge-Weber Syndrome or any other type of condition that may cause elevated episcleral venous pressure. WARNINGS/ PRECAUTIONS: • For prescription use only. • This device has not been studied in patients with uveitic glaucoma. • Do not use the device if the Tyvek® lid has been opened or the packaging appears damaged. In such cases, the sterility of the device may be compromised. • Due to the sharpness of certain injector components (i.e. the insertion sleeve and trocar), care should be exercised to grasp the injector body. Dispose of device in a sharps container. • iStent inject® W is MR-Conditional; see MRI Information below. • Physician training is required prior to use of the iStent inject® W System. • Do not re-use the stent(s) or injector, as this may result in infection and/or intraocular inflammation, as well as occurrence of potential postoperative adverse events as shown below under “Potential Complications.” • There are no known compatibility issues with the iStent inject® W and other intraoperative devices. (e.g., viscoelastics) or glaucoma medications. • Unused product & packaging may be disposed of in accordance with facility procedures. Implanted medical devices and contaminated products must be disposed of as medical waste. • The surgeon should monitor the patient postoperatively for proper maintenance of intraocular pressure. If intraocular pressure is not adequately maintained after surgery, the surgeon should consider an appropriate treatment regimen to reduce intraocular pressure. • Patients should be informed that placement of the stents, without concomitant cataract surgery in phakic patients, can enhance the formation or progression of cataract. ADVERSE EVENTS: Please refer to Directions For Use for additional adverse event information. CAUTION: Please reference the Directions For Use labelling for a complete list of contraindications, warnings and adverse events.
Glaukos®, iStent®, iStent inject®, iStent inject® W are registered trademarks of Glaukos Corporation. All rights reserved. ©2023 PM-EU-0266


