A 67-year-old White woman with a family history of glaucoma (father) underwent uneventful cataract surgery in her right eye. A 22.50 D IOL was implanted, and the refractive target was emmetropia. The standard postoperative treatment included guttae chloramphenicol 0.5% four times a day for 2 weeks and guttae dexamethasone 0.1% four times a day for 4 weeks.
At the 4-week postoperative visit with her local optician, the patient’s IOP was 41 mm Hg OD and 20 mm Hg OS. There were no signs of infection or active inflammation. The patient was referred to the hospital eye service, where topical treatment with guttae latanoprost once a day, guttae apraclonidine 1% three times a day, fixed combination guttae dorzolamide and timolol 2%/0.5% twice a day, and oral acetazolamide 250 mg three times a day was initiated. The iridocorneal angle was fully open upon gonioscopy; the visual acuity was 6/7.5 OU, and the central corneal thickness was 539 µm OD and 542 µm OS. The visual field test (Humphrey 24-2) and the retinal nerve fiber layer on OCT showed no clinically significant abnormalities in either eye. Diagnosis of steroid response was made based on the clinical information.
After 1 week, IOP in the right eye decreased to 25 mm Hg, but the patient reported significant side effects with the oral acetazolamide, including an extreme sense of fatigue and loss of appetite, which could not be tolerated. Oral acetazolamide was stopped and guttae brimonidine 0.2% twice a day in the right eye was added to the topical treatment course.
At 3 weeks, IOP in the right eye increased to 29 mm Hg despite the use of four glaucoma agents. The right conjunctiva was hyperaemic with signs of ocular surface disease potentially linked to the multiple topical drug therapy. The patient reported discomfort and a subjective deterioration of vision in her right eye. At that time, the patient was offered surgical treatment, including ab-interno viscocanaloplasty/trabeculotomy with OMNI.
The patient elected to undergo 360º viscocanaloplasty and 180º trabeculotomy inferiorly (Figures 1 and 2). No significant intraoperative complications were encountered.
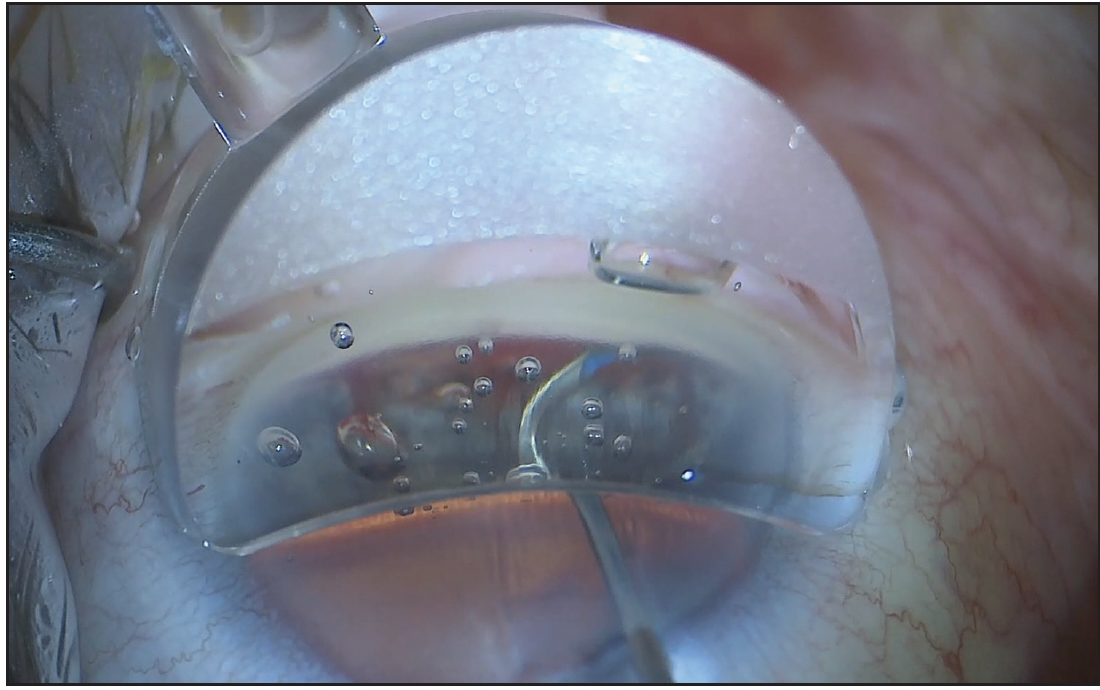
Figure 1. An inferonasal trabeculotomy was performed.
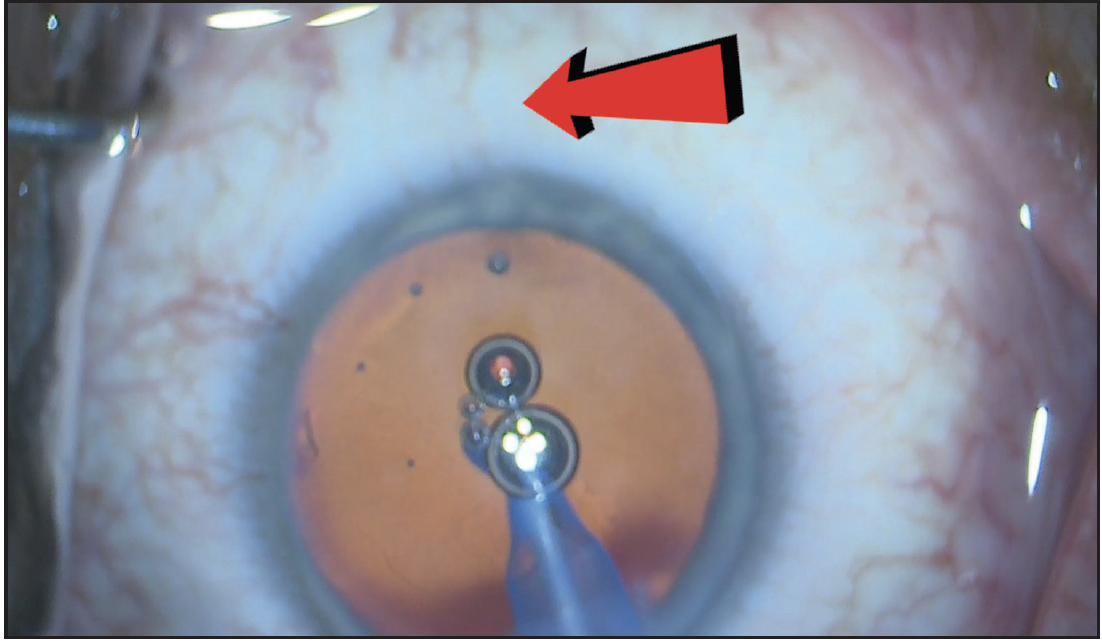
Figure 2. Blanching of the episcleral vessels (red arrow) is seen during irrigation/aspiration, confirming improved drainage and decreased resistance of the collector channels.
On day 1 postoperative, IOP in the right eye was 19 mm Hg. The presence of red blood cells was noticed, and there was minimal inflammation in the anterior chamber. The patient expressed good comfort, and visual acuity in the right eye was 6/12. Topical treatment included guttae chloramphenicol 0.5%, guttae dexamethasone 0.1% four times a day, guttae dorzolamide and timolol, and guttae apraclonidine 1%.
Two weeks later, IOP in the right eye was 17 mm Hg with no anterior chamber cell activity. Visual acuity in that eye was 6/7.5. Guttae dexamethasone 0.1% was reduced to three times a day for 1 week, then twice a day for 2 weeks, and finally once a day for 2 weeks. Guttae apraclonidine 1% was stopped, but guttae dorzolamide and timolol were continued.
By 8 weeks postoperative, the IOP in the right eye was 15 mm Hg. There were no signs of inflammation, and the patient reported good comfort and stable visual acuity. At this stage, the only topical drug in use was guttae dorzolamide and timolol, which was discontinued. Consecutive 4-week checks revealed right eye IOPs of 18 mm Hg with no topical treatment, good comfort, and stable visual acuity.
This case shows a significant steroid response following uneventful cataract surgery. Steroid responses may present with different magnitude. About 5% of healthy adults develop an IOP increase of more than 15 mm Hg following the use of topical steroids. People with glaucoma or a family history of glaucoma are at higher risk for developing a significant steroid response. The mechanism of the response involves both intra- and extracellular changes in the trabecular meshwork. These are usually reversible but may lead to trabecular sclerosis, sustained high IOP, and, if left uncontrolled, glaucomatous damage.
Targeting the trabecular meshwork addresses the pathophysiological mechanism and therefore treats increased IOP at its root. Ab-interno viscocanaloplasty/trabeculotomy with OMNI is an effective and minimally invasive treatment for steroid-induced glaucoma and can significantly reduce both IOP and the need for glaucoma drops.
IMPORTANT PRODUCT INFORMATION:
INDICATIONS FOR USE: The OMNI® Surgical System is indicated for the catheterization and transluminal viscodilation of Schlemm’s canal and the cutting of trabecular meshwork to reduce intraocular pressure in adult patients with open-angle glaucoma. For important safety information including contraindications, warnings, precautions and adverse events, please visit omnisurgical.com
Surgeons are paid consultants of Sight Sciences
This information is intended solely for the use of ophthalmic clinicians - Patients should contact their eyecare professional.
OMNI, Sight Sciences, and the Sight Sciences logo are registered trademarks of Sight Sciences, Inc. All other trademarks are the property of their respective owners.
© 2023 Sight Sciences, Inc 6/23 OM-2915-OUS.v1 CE2797
