The following case illustrates successful use of OMNI (Sight Sciences) to perform a 360º canaloplasty and 180º trabeculotomy in a patient with primary open-angle glaucoma (POAG).
A 52-year-old woman presented with POAG in both eyes, but the diagnosis was worse in the left. At presentation, IOP was 25 mm Hg OU, and corneal thickness was average. The introduction of topical therapy with a prostaglandin analogue achieved a decrease in IOP to 18 mm Hg in the left eye. A topical beta-blocker was added as a fixed-dose combination, and a further reduction in IOP to 16 mm Hg was achieved. The patient was monitored over the subsequent 4 years without any significant change in retinal nerve fibre layer (RNFL) thickness or visual field (Figure 1A). IOP in the left eye remained between 15 and 17 mm Hg during this period.
Six years after presentation, however, IOP in the left eye was 23 mm Hg despite apparently good adherence and substantial progression on visual field testing (Figure 1B). The mean deviation had declined from -2.39 dB in 2020 to -5.82 dB in 2022. There was paracentral involvement and evidence of significant progressive RNFL thinning on OCT. The visual acuity had also declined to 6/9, improving with pinhole to 6/6. An early nuclear sclerotic cataract was evident. In the left eye, a myopic shift with the spherical equivalent refraction was noted (-3.50 D of sphere in 2020 to -5.00 D of sphere in 2022). The patient was still driving due to the relatively well-preserved visual field in the right eye. Options for treatment including adding a third medication, performing selective laser trabeculoplasty (SLT), and performing trabeculectomy were discussed with the patient. The last was considered an option given the degree of progression, paracentral involvement, relatively young age of the patient, and low target IOP. The patient was not keen to take additional medication due to symptoms of dry eye disease. When the patient learned that SLT would probably not offer a good long-term option, she expressed the desire for more immediate and sustained IOP lowering.
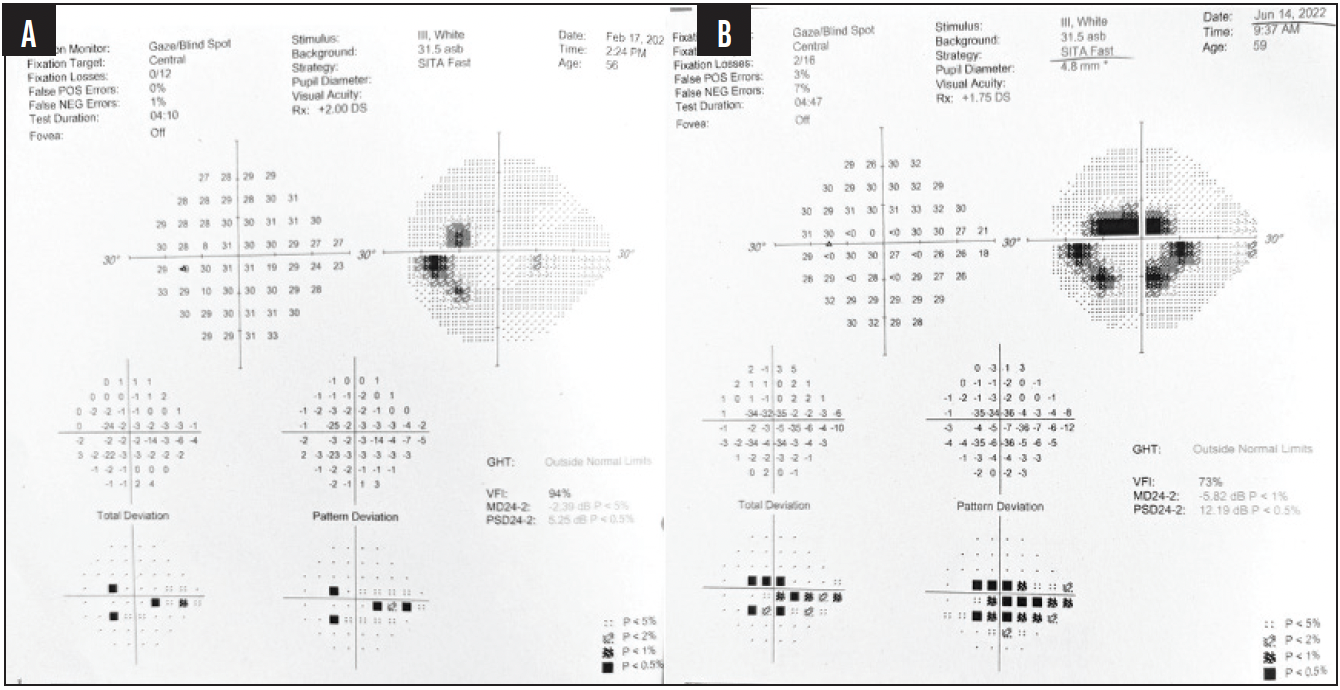
Figure 1. Visual field tests at ages 56 (A) and 59 (B) showing marked progression with paracentral involvement (tested with contact lenses).
We decided to combine cataract surgery with a canal-based MIGS procedure. The patient was well informed of the options and preferred to avoid the use of an implant-based treatment and the chance for a substantial IOP drop, which would be greater with a 360º treatment. Cataract extraction combined with 360º canaloplasty and 180º trabeculotomy was performed using the OMNI system (Figure 2).
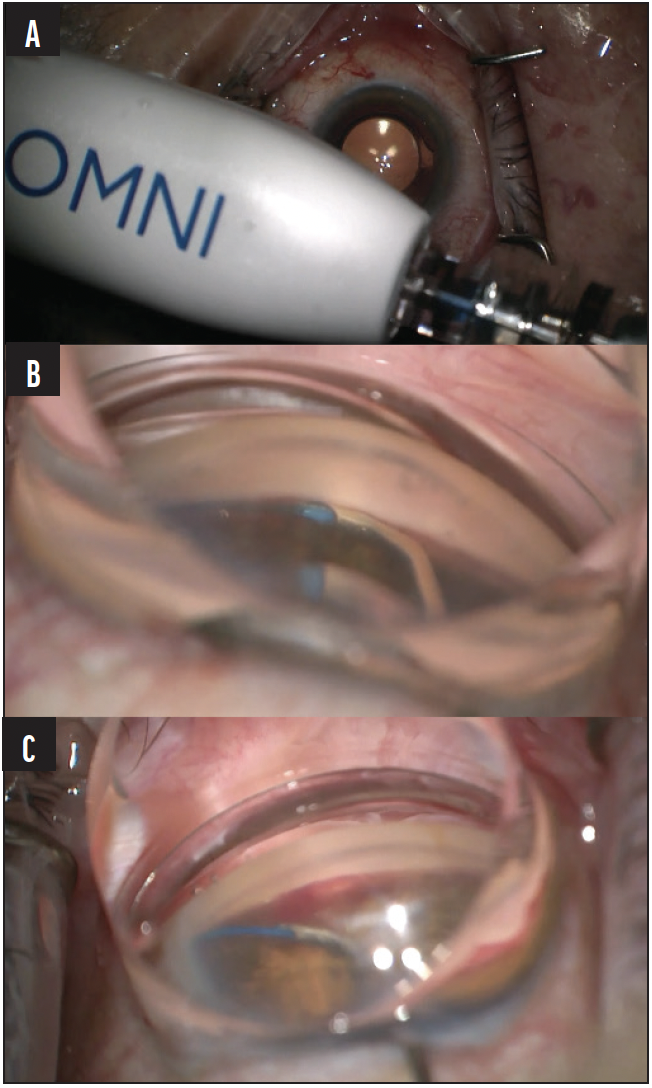
Figure 2. Photographs from the surgical video showing the OMNI device being primed with a cohesive OVD (A), entry of the OMNI catheter into Schlemm’s canal and canaloplasty (B), and trabeculotomy with withdrawal of the microcatheter and deroofing of Schlemm’s canal (C).
Postoperatively, the fixed-dose combination medication was continued to achieve a low target IOP with such a high preoperative IOP. Topical dexamethasone eye drops were prescribed twice hourly for 1 week and then four times per day for 4 weeks. On day 1, week 1, and month 1, IOP was 12 mm Hg, 11 mm Hg, and 13 mm Hg, respectively. The patient was still using topical dexamethasone. At 6 months, IOP in the left eye was 12 mm Hg, and it remained stable at the 12-month review, though the fixed-dose combination medication was continued. The patient’s UCVA improved to 6/6 with emmetropia targeted and achieved. The visual field remained stable.
It remains to be seen whether this patient might still require a filtering procedure in the future, but canaloplasty/trabeculotomy combined with phacoemulsification enabled safe removal of the cataract and a substantial reduction in IOP that was sustained at least in the medium term. The patient has subsequently undergone the same procedure in the right eye.
IMPORTANT PRODUCT INFORMATION:
INDICATIONS FOR USE: The OMNI® Surgical System is indicated for the catheterization and transluminal viscodilation of Schlemm’s canal and the cutting of trabecular meshwork to reduce intraocular pressure in adult patients with open-angle glaucoma. For important safety information including contraindications, warnings, precautions and adverse events, please visit omnisurgical.com
Surgeons are paid consultants of Sight Sciences
This information is intended solely for the use of ophthalmic clinicians - Patients should contact their eyecare professional.
OMNI, Sight Sciences, and the Sight Sciences logo are registered trademarks of Sight Sciences, Inc. All other trademarks are the property of their respective owners.
© 2023 Sight Sciences, Inc 6/23 OM-2915-OUS.v1 CE2797




