Postoperative outcomes play an integral role in the success of surgery with multifocal and trifocal IOLs and in producing happy patients. Today, we know that IOL decentration and angle kappa can influence postoperative outcomes. The topic of angle kappa can be challenging to understand, mainly because there are so many different terms to describe often theoretical optical axes and angles. In short, angle kappa is the angular separation between visual and pupillary axes (Figure 1), and its magnitude can impact postoperative outcomes and potential side effects with certain types of IOLs. Even that statement takes some background knowledge to understand. Instead, consider that we are talking about a mismatch between the center of an IOL, the pupil, and a line joining the fovea with a target of fixation.
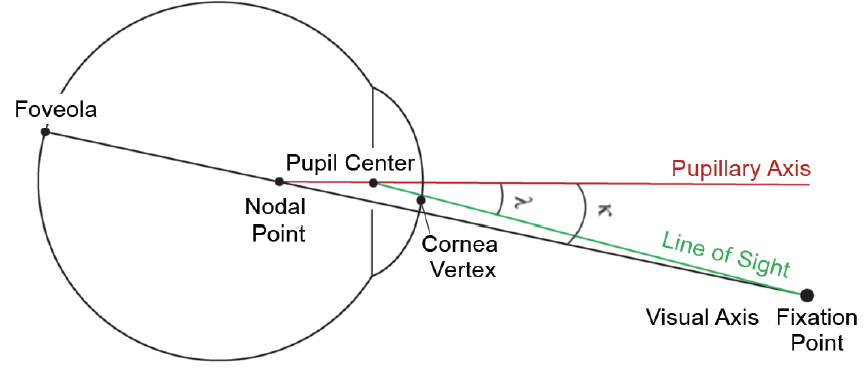
Figure 1. A depiction of angle kappa.
We recently conducted a study to determine the effect of decentration for the AT LISA tri 839MP (Carl Zeiss Meditec). Our results are presented in this article.
BACKGROUND
Decentration of any IOL can degrade image quality. The amount of decentration needed to impact vision for a monofocal IOL is quite extreme and usually the consequence of a surgical complication. A monofocal, neutral aspheric IOL is the most forgiving to decentration, with negative aspheric and spherical monofocal IOLs being slightly less immune. With a diffractive IOL, significant decentration can cause light to hit some of the rings at an unusual angle. In that situation, the pupil aperture can limit light to the rings, creating asymetrical diffractive patterns and unwanted visual symptoms.
Each diffractive optic has a different level of tolerability to decentration. In most eyes with minimal angle kappa, the two potential candidates for centration—pupil center and visual axis—are virtually superimposed and make positioning an easy decision. A higher angle kappa means that the center of the pupil does not fall on a line connecting the fovea to fixation. Therefore, the surgeon must decide between the more cosmetically appealing centration on the pupil and the potentially more functional centration on the visual axis. Moving the IOL too far from the pupil center creates problems with the pupillary aperture interfering with the diffraction process, and this has been blamed for giving patients waxy vision. In simple terms, with a high angle kappa, even when the lens is perfectly centered on either the pupil or the visual axis, a functional decentration could result, which can reduce image quality just the same as a decentered IOL in a more normal eye.
MEASURING ANGLE KAPPA, PLACING THE IOL
Angle kappa is difficult to define and even trickier to measure. A more modern and functional equivalent is the Chang-Waring chord (CWC), also known as chord mu. It is a two-dimensional vector measured across the corneal surface from the center of the pupil to the coaxially sighted, subject-fixated corneal light reflex. This measurement is possible with the IOLMaster 700 (Carl Zeiss Meditec). This device will also provide coordinates of the iris center, which relates to angle alpha and the center of the capsular bag, where an IOL will most likely settle.
Views have been divided on whether an IOL can stay positioned where it is placed or whether it goes where the capsule pushes it; however, there is clinical evidence that IOLs generally show long-term stability with decentration of 0.1 to 0.3 mm.1-3 There is no definite cutoff value of chord mu above which a diffractive multifocal IOL should not be used. Theoretical optical bench studies and in vivo testing indicate that if chord mu is greater than 0.6, 0.75, or 0.5 mm, respectively, patients with a diffractive lens are more likely to experience noticeable visual phenomena.3-5 Therefore, proper centration of the lens is crucial. With a nontoric IOL, it may be possible to rotate the lens within the mildly oval capsular bag so that haptic alignment allows more natural centration.
Diffractive trifocal IOLs like the AT LISA tri are particularly sensitive to residual astigmatism and will most commonly be used as a toric model in patients with 1.00 D of cylinder or more. This restricts the use of a rotational centering technique to let the capsule position the IOL naturally. In these cases, more effort needs to be made to remove OVD from behind the IOL and maximize capsular contact for permanent positioning. With a large CWC or angle kappa, positioning of a diffractive trifocal IOL becomes much more important. One compromise is to center the IOL between the Chang-Waring corneal light reflex and the pupil center.
Another consideration in terms of IOL decentration is implanting a trifocal IOL in a highly myopic eye with a large capsule. This is because the relatively small IOL can become more easily decentered (often inferiorly). The four-haptic design of the AT LISA tri, compared to C-loop haptic designs, has been shown to be more stable long term in such eyes.6,7
STUDY
We recently conducted a study looking at decentration, contrast sensitivity, and long-term visual acuity in 51 eyes implanted with the AT LISA tri. At 1-year postoperative, IOL centration remained within normal limits for all eyes; visual acuity at near, intermediate, and distance remained excellent; and subjective refraction was stable.
Decentration. At 1-year postoperative, the average decentration in all different directions was 0.25 ±0.21 mm. Most importantly, there was no statistically significant correlation between the decentration relative to the pupil or the corneal vertex compared to contrast sensitivity, UCVA and BCVA, or higher-order aberrations.
Contrast sensitivity. We also found no correlation between contrast sensitivity and decentration in mesopic and photopic conditions with and without glare stimuli (Figure 2). Our contrast sensitivity results in patients were equivalent to those of phakic patients a decade younger. There has been a lot of talk about the loss of contrast sensitivity with diffractive trifocal lenses, but our study clearly showed that, compared to a phakic person of a decade younger, patients with the AT LISA tri are actually doing very well. There is a tradeoff with any diffractive technology because light must be split, affecting contrast sensitivity. However, the advantages of this lens appear to make up for any downside in most people.
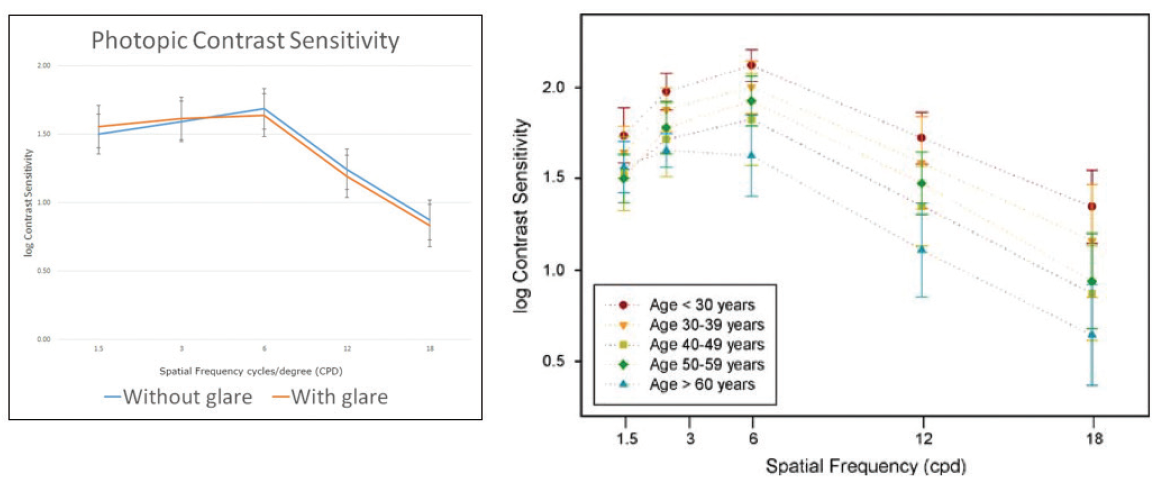
Figure 2. Contrast sensitivity with the AT LISA tri in mesopic and photopic conditions with and without glare stimuli.
Long-term visual data. Patients had excellent BCVA and UCVA at all distances. Notably, patients’ binocular distance UCVA was particularly good. And as you would expect, the binocular visual acuities were better than monocular due to binocular summation.
CONCLUSION
At normal physiological angle kappa or CWC values that would be expected in a normal population of cataract patients, the amount of IOL decentration does not appear to be detrimental to the outcome of implanting the AT LISA tri or AT LISA tri toric. Further, our study indicated that the AT LISA tri is a robust lens that can be beneficial in a wide variety of eyes.
Get in touch for more information!
1. Jung CK, Chung SK, Baek NH. Decentration and tilt: silicone multifocal versus acrylic soft intraocular lenses. J Cataract Refract Surg. 2000;26:582-585.
2. Wallin TR, Hinckley M, Nilson C, Olson RJ. A clinical comparison of single-piece and three-piece truncated hydrophobic acrylic intraocular lenses. Am J Ophthalmol. 2003;136:614-619.
3. Eppig T, Scholz K, Loüffler A, Messner A, Langenbucher A. Effect of decentration and tilt on the image quality of aspheric intraocular lens designs in a model eye. J Cataract Refract Surg. 2009;35:1091-1100.
4. Tandogan T, Son HS, Choi CY, Knorz MC, Auffarth GU, Khoramnia R. Laboratory evaluation of the influence of decentration and pupil size on the optical performance of a monofocal, bifocal, and trifocal intraocular lens. J Refract Surg. 2017;33(12):808-812.
5. Qi Y, Lin J, Leng L, et al. Role of angle κ in visual quality in patients with a trifocal diffractive intraocular lens. J Cataract Refract Surg. 2018;44(8):949-954.
6. Zhu X, He W, Zhang Y, Chen M, Du Y, Lu Y. Inferior decentration of multifocal intraocular lenses in myopic eyes. Am J Ophthalmol. 2018;188:1-8.
7. Meng J, He W, Rong X, Miao A, Lu Y, Zhu X. Decentration and tilt of plate-haptic multifocal intraocular lenses in myopic eyes. Eye and Vision. 2020;7:1-9.





