Cataract surgery in the setting of anticipated or unexpected zonular instability generally results in a complex situation for the anterior segment surgeon. A particular skill set is required to overcome the challenges associated with the diagnosis and management of this not-so-uncommon entity.
Zonular weakness is known to be associated with several systemic conditions, but it is especially important to rule out conditions that could potentially result in death, such as Marfan syndrome and homocystinuria. Additionally, it is important to note the number of clock hours affected and whether the zonular deficiency is focal or generalized, as this will determine the type and number of devices required for zonular support.
Presence of vitreous prolapse should also be ascertained during the initial exam, and anterior vitrectomy should be planned if it is detected. This article details five fundamental strategies for managing zonular deficiency and highlights our preferred techniques for these challenging cases.
Fundamental 1: ANTERIOR VITRECTOMY
In the presence of known or unexpected vitreous prolapse, not uncommon in zonular deficiency cases, we recommend using a pars plana approach for anterior vitrectomy, as this avoids pulling any additional vitreous into the anterior chamber while vitrectomy is performed (Figure 1). We prefer to access the eye using a 23-gauge trocar, which provides additional separation between the instrument and the vitreous base, theoretically reducing the likelihood of retinal tears.
FUNDAMENTAL NO. 1: FIGURES
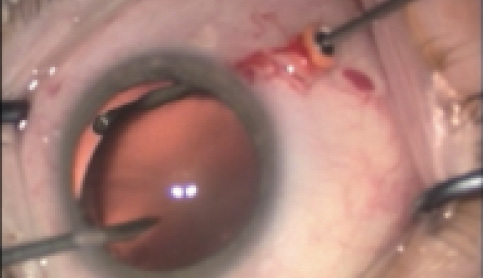
Figure 1. Pars plana anterior vitrectomy using a 23-gauge port.
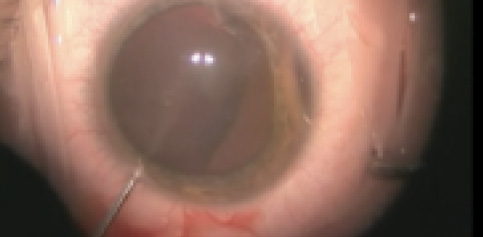
Figure 2. Triamcinolone injection through a paracentesis using a 27-gauge cannula.
Triamcinolone staining is helpful to aid in visualization of vitreous prolapse; therefore, we routinely inject it into the anterior chamber when the presence of vitreous is suspected (Figure 2).1 Because triamcinolone also stains viscoelastic materials, we prefer to inject it into a fluid-filled anterior chamber before an ophthalmic viscosurgical device (OVD) is used in order to prevent any confusion during anterior vitrectomy.
Fundamental 2: VITAL DYES
Trypan blue dye can be injected under fluid, but in these cases we recommend injecting it under an OVD, using a cannula as a paintbrush (Figure 3). This reduces the likelihood of staining the vitreous cavity through a zonular gap, compromising the red reflex; it also provides greater control over the anterior chamber and avoids additional vitreous prolapse.
FUNDAMENTAL NO. 2: FIGURES
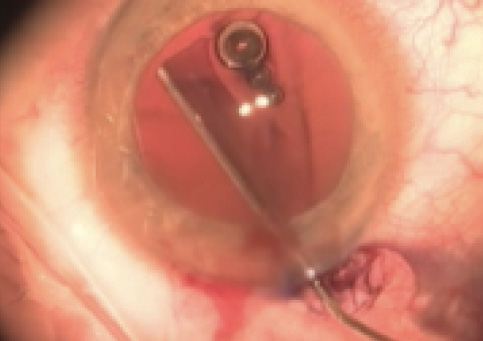
Figure 3. Trypan blue staining under OVD using a 27-gauge cannula.
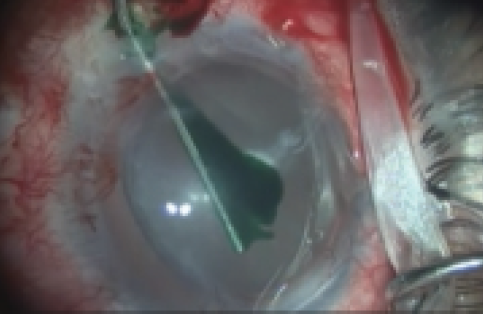
Figure 4. Indocyanine green staining using a 27-gauge cannula.
Trypan blue not only aids in the visualization of the anterior capsule, but it also affects its biomechanics, decreasing capsular elasticity.2 This, in turn, helps compensate for the lack of zonular countertraction during creation of the capsulorrhexis.
The decrease in capsular elasticity also means that the capsule will tear under less stretching. This is why, in capsules that are already fragile, such as in a congenital aniridic eye, we recommend avoiding trypan blue and using indocyanine green dye instead. Indocyanine green provides similar staining properties but will not change the elasticity of the capsule (Figure 4).3-4
Fundamental 3: CAPSULORRHEXIS
Initiating the capsulorrhexis in an eye with zonular weakness can be challenging due to the lack of countertraction and the elastic properties of the anterior capsule. Surgeons may notice a pincushion effect while trying to create the initial tear (Figure 5).5 To deal with this issue, we use the crossed-swords technique, in which we enter the anterior chamber using two opposing 30-gauge needles and create a fold on the anterior capsular surface (Figure 6).6 When both needles meet at the center of the capsule, one of them eventually pierces the capsular fold. We can then initiate the capsular tear to start the rhexis or, at least, to allow insertion of an iris or capsular hook through the tear to provide temporary countertraction, allowing us to complete the capsulorrhexis.
In these types of cases, we recommend the use of microforceps with a rhexis ruler, as this allows the surgeon to measure both the size of the desired rhexis and of the actual rhexis as it is being created. Aiming slightly smaller than the usual 5 mm will leave enough peripheral rim to support fixation devices.
FUNDAMENTAL NO. 3: FIGURES
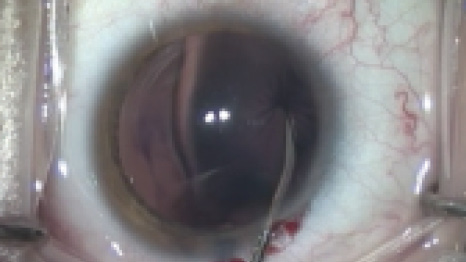
Figure 5. Pincushion effect using a cystotome over the anterior capsule in the setting of zonular deficiency, while attempting the initial capsulorrhexis tear.
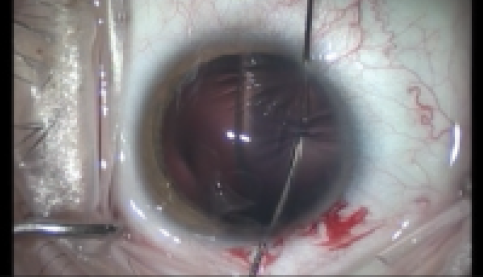
Figure 6. Crossed-swords technique using two opposing 30-gauge needles.
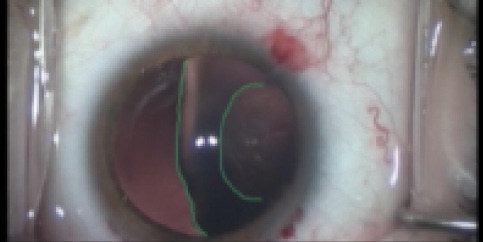
Figure 7. Linear capsulorrhexis edge parallel to flat edge of a subluxed and misshaped lens.
Despite the availability of femtosecond laser technology to create the capsulorrhexis, we prefer a manual approach in eyes with zonular deficiency, as it allows us to customize the shape of the capsulorrhexis based on the shape of the crystalline lens. In eyes with congenital or longstanding zonular deficiency, the lens edge will be flat in areas of severe zonular weakness, and this shape must be replicated as a parallel line when that area of the capsulorrhexis is created (Figure 7). In this way, after a capsular tension ring (CTR) is inserted, the lens edge will acquire the round shape of the ring, and this will also round out the capsulorrhexis margin, resulting in a circular rhexis. Failure to recognize this phenomenon can lead to an insufficient peripheral rim to support certain types of fixation devices inside the capsular bag.
Further, a manual capsulorrhexis has the biomechanical advantage of having a continuous margin with no stress points, as compared with a femtosecond laser-assisted capsulorrhexis, which has multiple stress points at each of the laser spots used to create it.7 Finite element modeling analysis has shown that this characteristic could be detrimental to the integrity of the laser-assisted capsulorrhexis,8 especially in a setting in which devices that apply pressure to the capsulorrhexis margin may be used.
Fundamental 4: INTRAOPERATIVE CAPSULAR BAG SUPPORT
Phacoemulsification in the setting of zonular weakness can be challenging, and temporary capsular support may be required to successfully remove the lens. We prefer to use capsular retractors, inserted through three small paracenteses (Figure 8); these devices provide several advantages over standard iris hooks, as they apply the tension forces to the equator of the bag instead of directly on the capsulorrhexis margin, potentially avoiding anterior capsular tears. They can also be used to provide countertraction to complete the capsulorrhexis, if that has not yet been done.
FUNDAMENTAL NO. 4: FIGURES
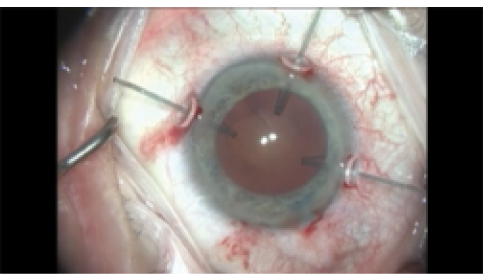
Figure 8. Three capsular retractors stabilizing the capsular bag.
Fundamental 5: LONG-TERM ZONULAR SUPPORT
We recommend using a CTR in all eyes with potential zonular weakness, including all cases of pseudoexfoliation syndrome, even if the zonules appear to be stable. Having a CTR inside a bag that has a considerable risk of future subluxation will considerably decrease the complexity of a repositioning procedure if one becomes necessary.
FUNDAMENTAL NO. 5: FIGURES
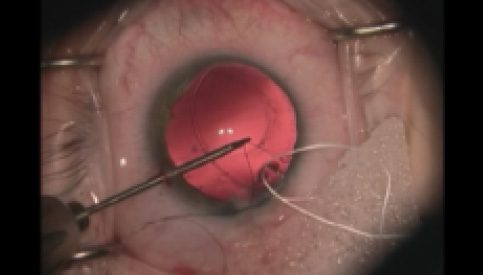
Figure 9. Modified Cionni CTR with a ePTFE suture through the fixation eyelet and 10-0 nylon suture through the leading eyelet. Countertraction is being applied to the nylon suture though a paracentesis.
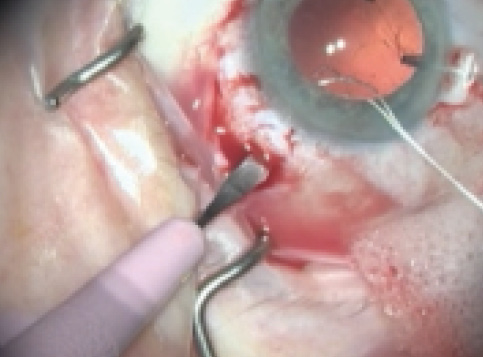
Figure 10. A sclerotomy is created using a 15° blade in the area of greatest zonular weakness, through which one end of an ePTFE suture will be retrieved.
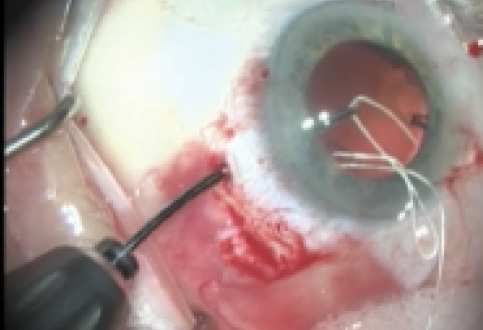
Figure 11. Needleless ePTFE suture is retrieved through a sclerotomy using 25-gauge microforceps.
Whenever the zonular deficiency compromises more than 3 clock hours, or if it is felt that a standard CTR will not provide enough bag stability, we strongly recommend scleral fixation of the capsular bag/IOL complex. Our go-to devices are the Cionni modified CTR9 and the Ahmed capsular tension segment (both from Morcher).10
Although this is an off-label use, we favor expanded polytetrafluoroethylene (ePTFE; Gore-Tex) over polypropylene sutures for long-term scleral fixation, considering the known limited lifespan of 10-0 polypropylene.11 Similarly, 9-0 polypropylene has started to show the same problems, as illustrated by recent cases managed in our clinic 10 years after the initial surgery, leading us to believe that the suture lifespan is related more to material type than to thickness.
In terms of surgical technique, we like to prethread the fixation eyelet of the modified CTR outside the eye using a needleless ePTFE suture, and the leading eyelet using 10-0 nylon, to provide countertraction if necessary (Figure 9). This countertraction can be applied either through the main incision or through the opposing paracentesis, depending on the vector of force required, and it can be quite useful if the ring gets trapped in a fold of the loose and unstable capsular bag.
AT A GLANCE
• Zonular weakness is not uncommon, and a particular skill set is required to overcome the challenges associated with its diagnosis and management.
• The number of clock hours affected and whether the zonular deficiency is focal or generalized determines the type and quantity of devices required for zonular support.
• When a surgeon encounters a complex zonular deficiency case, he or she must have a plan B to rely on if things do not go as planned and the integrity or functionality of the capsular bag cannot be maintained.
Once the CTR is fully inserted into the bag, we create two sclerotomies, 2 mm apart, with a 15° blade, at the sulcus level (Figure 10). These allow us, using 23- or 25-gauge microforceps, to retrieve each end of the ePTFE suture and secure them to the wall of the eye (Figure 11).12 The same technique is applied if a capsular tension segment is also required, 180° apart, especially in cases of diffuse zonular weakness. All ePTFE knots are buried inside the scleral wall and covered by conjunctiva, resulting in a stable capsular bag fixated to the wall of the eye.
Plan B for Managing Zonular Deficiency
If things do not go as planned in complex cases of zonular deficiency, and the integrity or functionality of the capsular bag cannot be maintained with steps outlined in our accompanying article, it is important for surgeons to have a backup plan. Our preferred technique includes the use of a PMMA posterior chamber IOL with fixation eyelets on its haptics with expanded polytetrafluoroethylene (ePTFE) sutures for scleral fixation.1
In this approach, a 7-mm scleral tunnel is created, along with two pairs of sclerotomies 180° apart, with a 15° blade. Using the same concept applied for the Cionni ring, we insert the needleless end of an ePTFE suture into the anterior chamber, and that suture is retrieved through one of the proximal sclerotomies using microforceps. We do the same with the other end of the needleless suture through the second proximal paracentesis, which creates a loop of ePTFE suture outside the eye (Figure 1). That loop is folded, preparing a girth hitch or cow hitch, through which we slide the proximal haptic of the PMMA IOL, trapping the fixation eyelet into the hitch; this provides two-point fixation for the proximal haptic (Figure 2). Next, we rotate the IOL 180° and repeat the process using the distal pair of sclerotomies and trapping the fixation eyelet on the distal haptic, adding an extra two-point fixation to the IOL. We then proceed to insert the IOL into the posterior chamber through the pupil, while gently pulling the suture slack. Knots are tied in a 3-1-1 fashion and rotated counterclockwise on both distal and proximal sides, burying them into the scleral wall.
This technique enables true four-point fixation, avoids both tilt and torque, and yields long-term stability. It also provides the option to fine-tune centration by rotating the ePTFE sutures to displace a trapped haptic, resulting in centration of the optic into the desired position based on the Purkinje reflexes.
1. Snyder ME, Perez MA. Tiltless and centration adjustable scleral-sutured posterior chamber intraocular lens. J Cataract Refract Surg. 2014;40(10):1579-1583.
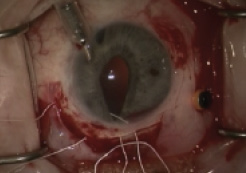
Figure 1. After both ePTFE suture ends have been retrieved through a set of sclerotomies, a girth hitch is prepared for haptic fixation.

Figure 2. Haptic eyelet trapped into an ePTFE girth hitch.
Did You Know?
Homocystinuria is an inherited disorder that affects the metabolism of the amino acid methionine. It must be ruled out in any zonular deficiency case before entering the operating room, due to major anesthetic considerations, including development of thromboembolism, need to avoid nitrous oxide, and hypoglycemia.
Asghar A, Ali FM. Anaesthetic management of a young patient with Homocystinuria. J Coll Physicians Surg Pak. 2012;22(11):720-722.
When one encounters complex zonular deficiency, one must have a plan B to rely on if things do not go as planned and the integrity or functionality of the capsular bag cannot be maintained. Techniques available for such an event include iris fixation or use of an iris-claw IOL or a scleral-fixated IOL. For our preferred approach, see Plan B for Managing Zonular Deficiency.
CONCLUSION
Several alternatives are available for the management of complex zonular deficiency. It is important for the cataract surgeon to become familiar with his or her surgical technique of choice in order to prevent complications and to successfully manage them when they occur. n
1. Burk SE, Da Mata AP, Snyder ME, Schneider S, Osher RH, Cionni RJ. Visualizing vitreous using Kenalog suspension. J Cataract Refract Surg. 2003;29:645-651.
2. Dick HB, Aliyeva SE, Hengerer F. Effect of trypan blue on the elasticity of the human anterior lens capsule. J Cataract Refract Surg. 2008;34:1367-1373.
3. Schneider S, Osher RH, Burk SE, Lutz TB, Montione R. Thinning of the anterior capsule associated with congenital aniridia. J Cataract Refract Surg. 2003;29:523-525.
4. Snyder ME. Staining the capsule in congenital aniridic eyes. J Cataract Refract Surg. 2008;34:1367-1373.
5. Snyder ME, Perez MA. Complications of capsulorrhexis. In: Buratto L, Brint S, Romano MR, eds. Cataract Surgery Complications. Thorofare, NJ: SLACK; 2013.
6. Snyder ME, Lindsell LB. Crossed-swords, capsule-pinch technique for capsulotomy in pediatric and/or loose lens cataract extraction. J Cataract Refract Surg. 2010;36:197-199.
7. Abell RG, Davies PE, Phelan D, Goemann K, McPherson ZE, Vote BJ. Anterior capsulotomy integrity after femtosecond laser-assisted cataract surgery. Ophthalmology. 2014;121:17-24.
8. Krag S, Thim K, Corydon L, Kyster B. Biomechanical aspects of the anterior capsulotomy. J Cataract Refract Surg. 1994;20:410-417.
9. Cionni RJ, Osher RH. Management of profound zonular dialysis or weakness with a new endocapsular ring designed for scleral fixation. J Cataract Refract Surg. 1998;24:1299-1306.
10. Ahmed IK, Kranemann C, Crandall AS. Capsular hemi-ring: next step in effective management of profound zonular dialysis. Film presented at: ASCRS Film Festival; April 2003; San Francisco, California.
11. Price MO, Price FW Jr, Werner L, Berlie C, Mamalis N. Late dislocation of scleral-sutured posterior chamber intraocular lenses. J Cataract Refract Surg. 2005;31:1320-1326.
12. Hoffman RS, Snyder ME, Devgan U, Allen QB, Yeoh R, Braga-Mele R; ASCRS Cataract Clinical Committee; Challenging/Complicated Cataract Surgery Subcommittee. Management of the subluxated crystalline lens. J Cataract Refract Surg. 2013;39:1904-1915.
Mauricio A. Perez, MD
- Cornea, Refractive Surgery and External Diseases Fellow, Toronto Western Hospital, University of Toronto, Canada
- mperez@gmx.us
- Financial disclosure: None
Michael E. Snyder, MD
- Member of the Board of Directors, Cincinnati Eye Institute, University of Cincinnati, Ohio
- msnyder@cincinnatieye.com
- Financial disclosure: Consultant (Alcon, Haag-Streit)




