I was the first to implant the iolAMD (London Eye Hospital Pharma), in a small hospital study of three eyes of two patients, to determine the safety and efficacy of the lens system in the treatment of macular disease. The technology, which incorporates two injectable, hydrophobic acrylic IOLs, is intended in particular for the surgical treatment of dry age-related macular degeneration (AMD). The lens behaves much like a telescope, providing patients with 1.3X magnification of the original image.
MINIMALLY INVASIVE PROCEDURE
AMD is the most common cause of irreversible severe visual loss in the developed world in individuals more than 60 years old. Several concepts have been discussed in recent years to overcome the problem of an aging macula in patients with age-related cataracts; however, many previous attempts to solve this problem involved a more complex surgical procedure accompanied by high risks and large incisions of up to 12 mm (see Other Concepts Intended to Prevent Vision Loss Related to Macular Disease).
The iolAMD technology is different: This patient- and surgeon-friendly approach is minimally invasive, as it entails implantation of the dual-lens system through a 3-mm, sutureless incision. The procedure takes approximately 5 to 10 minutes, compared with 45 to 60 minutes for other AMD lens implant surgeries.
based on a galilean telescope
The lens, which received the CE Mark in 2014, is based on a Galilean telescope using two lenses of differing powers in minus and plus. Both lenses are manufactured from a hydrophobic acrylic material and can be injected with a standard soft-tip cartridge and injector system. The first is a capsular-bag–positioned IOL that has a high minus power, a 4-mm optic, and an overall length of 11.2 mm. The second is a sulcus-positioned IOL with a high plus power, a 5-mm hyper-aspheric optic, and an overall diameter of 11.75 to 12 mm.
HOW IT WORKS
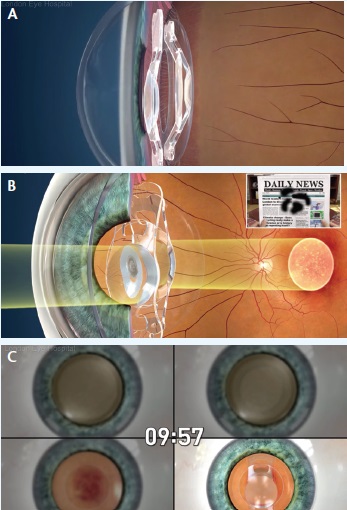
The iolAMD is composed of two hydrophobic acrylic IOLs, one of which is implanted in the capsular bag and the other in the sulcus (A). Together, the two lenses act like a Galilean telescope and provide magnification of 1.3X the original image (B). The lenses are implanted after cataract extraction in a procedure that takes approximately 10 minutes (C).
Other Concepts Intended to Prevent Vision Loss Related to Macular Disease
Implantable Magnification Technologies
IOL VIP
IMT
Retina Prosthesis
Argus II Retinal Prosthesis System
1. Orzalesi N, Pierrottet CO, Zenoni S, et al. The IOL-Vip System: a double intraocular lens implant for visual rehabilitation of patients with macular disease. Ophthalmology. 2007;114(5):860-865.
After implantation, the lenses behave like a telescope, providing magnification of the original image by approximately 1.3X (see How It Works). Because the sulcus-positioned lens optic is slightly decentered, it creates a prismatic effect, which diverts the magnified image to a healthy part of the retina, adjacent to the macula. The healthy retina then takes over the role of the macula, thereby providing improved vision.
PERSONAL EXPERIENCE
The two patients in my prospective, nonrandomized clinical trial had visually significant cataracts and intermediate dry AMD with drusen. Exclusion criteria were pathologies that might be aggravated by the IOL or make it ineffective, other pathologies of the retina or cornea, glaucoma, pseudoexfoliation, zonular loss, or a history of uveitis. Three eyes were treated; all underwent standard small-incision phacoemulsification using topical anesthesia. Lens orientation was determined with the iolAMD Simulator, which is included with the initial lens kit. Preoperatively, the simulator predicted postoperative near visual acuity of 20/200 to 20/100, and this was achieved 1 day after cataract surgery in all eyes.
In follow-up to 3 months, distance and near visual acuities improved. In fact, one eye reached near and distance BCVAs of 20/32 and 20/25, respectively. All distance BCVA results were within 20/40 to 20/25 after 3 months. The prismatic IOL effect did not lead to diplopia in the patient implanted bilaterally.
AT A GLANCE
• The iolAMD, composed of two hydrophobic acrylic IOLs,
behaves much like a telescope, providing macular disease
patients with 1.3X magnification of the original image.
• Early results suggest that the iolAMD is a promising
technology to improve near and distance visual acuities in
eyes with intermediate dry AMD.
CONCLUSION
Early results suggest that the iolAMD is a promising technology to improve near and distance visual acuities in eyes with intermediate dry AMD. Further studies with more patients and long-term follow-up are currently under design, and the regulatory process to bring this technology to more markets is in motion. The IOL may also be useful in patients with other macular diseases, including those with diabetic maculopathy, macular hole, and myopic degeneration. n
Fritz H. Hengerer, MD, PhD
- Deputy Director and Senior Head Physician, Department of Ophthalmology, Johann Wolfgang Goethe University, Frankfurt, Germany
- Fritz.Hengerer@kgu.de
- Financial disclosure: None

