“The fundamental things apply, as time goes by.”
— Herman Hupfeld, “As Time Goes By,” 1931
The human crystalline lens changes throughout life. It increases in size and mass over time, the optical properties of the lens change, the gradient refractive index changes, accommodation decreases, and presbyopia sets in. Ultimately, cataract develops, and the crystalline lens is removed and replaced in cataract/IOL surgery.
AT A GLANCE
• The crystalline lens grows throughout life.
• Over time, the optical properties of the lens change, the gradient refractive index changes, accommodation decreases, presbyopia sets in, and cataract develops.
• Humans are outliving the life expectancy of the crystalline lens.
• Efforts are under way to reverse presbyopia and cataract by undoing age-related protein damage at the molecular level.
Hundreds of years ago, when the average lifespan was much shorter than it is today, age-related conditions such as presbyopia and cataracts did not pose complex challenges for most people. Now, however, due to increasing life expectancy, most people outlive their lenses. Thus, it has become necessary for us to better comprehend the changes that occur in the crystalline lens over time in order to address their effects on patients’ quality of vision. This article highlights five fundamental concepts in our understanding of the lens as time goes by.
1. THE LENS IS ECTODERMAL IN ORIGIN
The crystalline lens is ectodermal in origin. The anterior cells of the lens vesicle form the epithelial cells, and the posterior cells of the lens vesicle form the lens fiber cells. These cells, which originate in the embryonic lens, form the embryonic nucleus that still exists in the adult lens. The adult lens contains the embryonic nucleus at its center, surrounded by the fetal nucleus, the juvenile nucleus, and the adult nucleus. Among the oldest cells and oldest proteins in the body are those contained inside that embryonic nucleus. The nucleus is surrounded by the cortex, which contains some of the youngest cells in the lens.1,2
2. THE CRYSTALLINS MUST HAVE A LONG LIFESPAN
In most tissues, proteins are constantly being degraded, synthesized, and replaced. This turnover ensures a constant supply of new, functional proteins. Nonfunctional, damaged, or toxic molecules can be destroyed and removed. However, this process cannot happen in the lens because the lens is constrained within the capsule and has no vascular supply. The crystallin proteins of the lens are among the oldest, longest-lived proteins in the body.3
The crystallins constitute about 90% of the proteins within the lens, and they are normally water-soluble. The three main crystallin types found in the eye are the alpha-, beta-, and gamma-crystallins. Crystallins are heat-shock proteins that are molecular chaperones; alpha-crystallins bind to the beta-crystallins after the beta-crystallins degrade in an effort to prevent their aggregation.
High concentrations of tightly packed crystallins allow the lens to be transparent, with a refractive index that is greater than the surrounding media and a gradient refractive index. The crystallins must pack tightly and regularly on a scale similar to that of the wavelengths of light. Additionally, they must be soluble, they must not crystallize at high concentrations, and they must have a long lifespan, as there is no mechanism for turnover, repair, or breakdown inside the lens.
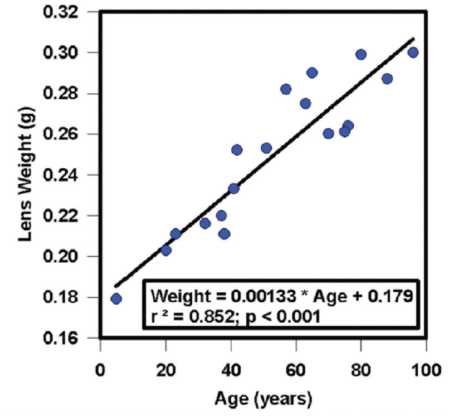
Figure 1. Growth of the human lens throughout life.
3. THE LENS CHANGES GREATLY OVER TIME
Many changes occur within the lens. The lens grows throughout life, accumulating weight as a function of age, as shown in Figure 1 as a linear increase in wet weight between 5 and 97 years.4 With that aging and growth comes an increase in axial thickness of the human lens in vivo but no increase in lens equatorial diameter.5 Similarly, the lenses of adult rhesus monkeys show an increase in axial thickness but no increase in equatorial diameter with increasing age.6
There is tremendous variation in shape and size of individual human lenses.4 Despite this variability, there are some systematic changes that occur over time. The focal length of the lens changes as a function of age, and there is a systematic increase in the focal length of isolated lenses (Figure 2). There is also a systematic change in the spherical aberration of the lens. Young isolated lenses have negative spherical aberration, but, at about age 40 years, that negative spherical aberration starts to become positive spherical aberration, and this trend continues to progress through the remainder of life (Figure 3).4
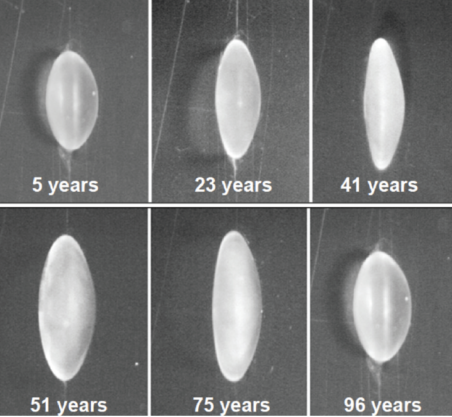
Figure 2. Age-related variations in human lenses.
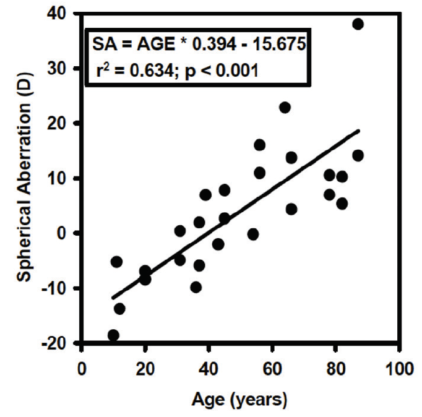
Figure 3. Age-related changes in spherical aberration.
As previously mentioned, the lens has a gradient refractive index. In a 7-year-old lens, this gradient has a parabolic curve, whereas in an 82-year-old lens, the gradient plateaus centrally.7
Crystallins are normally water-soluble proteins. With age, they undergo posttranslational modifications. These include racemization, deamidation, and oxidation of amino acid subgroups. This results in degradation and cleavage of the backbones of those proteins, altering their 3-D structure. This process allows intramolecular disulfide bonding of those proteins, exposes their hydrophobic core, and leads to aggregation, insolubility, and, ultimately, cataract.8
Amino acids are chiral molecules. That is, they exist in left- and right-handed mirror image forms called enantiomers or optical isomers; they can be L-isomers or D-isomers. Racemization is the process whereby an enantiomerically pure form is converted into a mixed form. Racemization of serine, an amino acid that is part of the crystallin structure, has been demonstrated as a function of age in the nucleus of human lenses. Racemization is a bad process for amino acids to undergo; the result is a loss of soluble alpha-crystallins from the human lens nucleus with increasing age.
Increased thickness, stiffness. There are many components of proteins in the lens that change, and they change in many ways. These include racemization, proteolysis, deamidation, glycation, disulfide bonding ultimately resulting in truncation, and protein aggregation, which is the formation of nonnative, high-molecular-weight structures.3 When a young lens undergoes accommodation, it is the nucleus—not the cortex—that undergoes an increase in thickness.
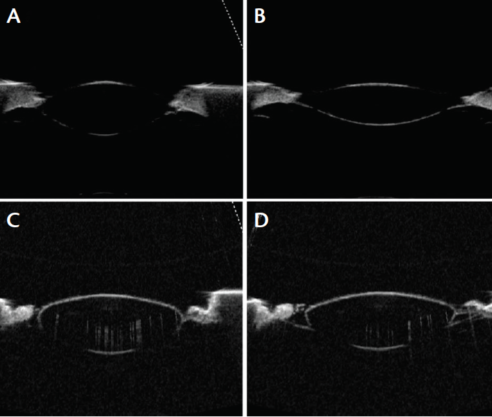
Figure 4. A 6-year-old monkey lens unstretched (A) and stretched (B) compared with a 46-year-old human lens unstretched (C) and stretched (D).
When young monkey lenses are subjected to mechanical stretching, the lens fiber cells are highly elastic, allowing the lens to undergo considerable changes in shape. A young monkey lens is soft and pliable, whereas a 46-year-old human lens essentially fails to undergo any change in shape with mechanical stretching (Figure 4). This is due to the progressive loss of accommodation, which, ultimately, by age 50, effectively results in complete loss of accommodation.
We have demonstrated this with isolated human lenses, mechanically stretching them to produce accommodative optical changes. Young lenses stretched in this way undergo 12.00 to 16.00 D of accommodative change, but the same amount of stretching applied to 60-year-old lenses fails to produce any accommodative optical change.4 That is because these lenses have become stiff and unable to accommodate. Mechanical compression of intact human lenses shows that they undergo an exponential increase in stiffness.9 In sectioned lenses, the young nucleus is softer than the cortex. However, with increasing age, there is an exponential increase in the stiffness of both the nucleus and the cortex, with an overall much greater increase in the nucleus.
Presbyopia. The complete loss of accommodation occurs at around age 50; however, the progressive increase in stiffness of the lens continues well beyond the time point at which accommodation is completely lost.4,9 Rhesus monkeys also lose their accommodative ability with increasing age, and the same kind of exponential increase in lens stiffness as seen in humans can be observed.
Accelerated aging can also be duplicated in porcine lenses by exposing them to elevated temperatures for a prolonged period of time. Eight hours of exposure at 50°C has been shown to yield a progressive increase in stiffness of porcine lenses.10 Exposure to elevated temperature also results in the same progressive decrease in soluble alpha-crystallin levels in those lenses as in human lenses. Unsurprisingly, if one plots the loss of alpha-crystallins from solution with the progressive decline in accommodative amplitude, the two curves are virtually superimposable.11
Population surveys have shown that the age of onset of presbyopia is inversely related to temperature. In climates with warmer temperatures, people tend to develop presbyopia at an earlier age.12 The crystallin proteins in the lens undergo a similar process: The elevated temperatures that the lens is exposed to for a protracted period cause the same type of denaturation.
Cataract. As noted above, the lens contains long-lived crystallin proteins with no opportunity for turnover, breakdown, or repair. This results in aggregation of high-molecular-weight proteins and disruption of the organized packing of the crystallins.3 This process—which is the same that causes presbyopia—ultimately results in disruption of the cellular structure of the lens and localized regional changes in protein and cell density, thereby causing light scatter and cataract. The crystallin structure in the lens fiber cells results in a transparent lens, but, as damage occurs and those crystallins become unfolded and disorganized and precipitate, opacities from cataract develops.
4. RESEARCH EFFORTS ARE UNDER WAY
Can this aging of the lens be delayed or reversed? The primary cause of both presbyopia and cataract is the posttranslational modifications of the lens crystallins. Research is under way to try to reverse this process.
A topical formulation of lipoic acid choline ester 1.5% (EV06 [formerly Dioptin]; Encore Vision) has shown promise in a phase 1/2 clinical trial.13 Participants taking the drug twice daily for 3 months showed improvements in distance-corrected near visual acuity, and the drug was well tolerated. EV06 is an oxidizing agent designed to break disulfide bonds in an effort to reduce lens stiffness in the crystalline lens and restore accommodation.
Lanosterol, a tetracyclic triterpenoid, has also been shown to reverse crystallin aggregation and reduce cataract severity in animal studies.14 If solutions containing high concentrations of alpha-crystallins are exposed to elevated temperature, those crystallins will aggregate into visible structures. When that solution is exposed to lanosterol, it completely dissolves those aggregated compounds of crystallins in vitro. Further, investigators have shown lanosterol treatment may reduce cataract severity and increase transparency in dissected rabbit cataractous lenses in vitro and cataract severity in vivo in dogs.14
5. THE HUMAN LIFESPAN REMAINS A CHALLENGE
The problem with both presbyopia and cataract is that humans are outliving the life expectancy of the lens. In the early 1900s, when life expectancy was around age 50, most people were not experiencing the effects of cataracts and presbyopia to the extent that we are today.15 We are outliving the natural lifespan of our lenses, and the lifelong exposure of lens crystallins to elevated body temperature results in aggregation and precipitation of those crystallins. Aggregation of lens crystallins results in increased stiffness, leading to presbyopia and cataract.
CONCLUSION
Efforts are under way to reverse presbyopia and cataract by undoing age-related protein damage at the molecular level. Further study hopefully will elucidate which methods show the greatest potential to increase the longevity of the lens.
1. Cvekl A, Ashery-Padan R. The cellular and molecular mechanisms of vertebrate lens development. Development. 2014;141(23):4432-4447.
2. Kuszak JR, Al-Ghoul KJ, Costello MJ. Pathology of age-related human cataracts. In: Duane’s Clinical Ophthalmology. Duane T, ed. Philadelphia: Lippincott Williams & Wilkins; 2006.
3. Toyama BH, Hetzer MW. Protein homeostasis: live long, won’t prosper. Nat Rev Mol Cell Biol. 2013;14(1):55-61.
4. Glasser A, Campbell MC. Biometric, optical and physical changes in the isolated human crystalline lens with age in relation to presbyopia. Vision Res. 1999;39(11):1991-2015.
5. Strenk SA, Semmlow JL, Strenk LM, Munoz P, Gronlund-Jacob J, DeMarco JK. Age-related changes in human ciliary muscle and lens: a magnetic resonance imaging study. Invest Ophthalmol Vis Sci. 1999;40(6):1162-1169.
6. Wendt M, Croft MA, McDonald J, Kaufman PL, Glasser A. Lens diameter and thickness as a function of age and pharmacologically stimulated accommodation in rhesus monkeys. Exp Eye Res. 2008;86(5):746-752.
7. Jones CE, Atchison DA, Meder R, Pope JM. Refractive index distribution and optical properties of the isolated human lens measured using magnetic resonance imaging (MRI). Vision Res. 2005;45(18):2352-2366.
8. Hanson SR, Hasan A, Smith DL, Smith JB. The major in vivo modifications of the human water-insoluble lens crystallins are disulfide bonds, deamidation, methionine oxidation and backbone cleavage. Exp Eye Res. 2000;71(2):195-207.
9. Heys KR, Cram SL, Truscott RJ. Massive increase in the stiffness of the human lens nucleus with age: the basis for presbyopia? Mol Vis. 2004;10:956-963.
10. Heys KR, Friedrich MG, Truscott RJ. Presbyopia and heat: changes associated with aging of the human lens suggest a functional role for the small heat shock protein, alpha-crystallin, in maintaining lens flexibility. Aging Cell. 2007;6(6):807-815.
11. Glasser A, Campbell MC. Presbyopia and the optical changes in the human crystalline lens with age. Vision Res. 1998;38(2):209-229.
12. Miranda MN. The geographic factor in the onset of presbyopia. Trans Am Ophthalmol Soc. 1979;77:603-621.
13. Encore Vision announces successful phase I‐II study of topical EV06 for the treatment of presbyopia [press release]. Encore Vision. May 5, 2016. http://encorevisioninc.com/2016/05/05/encore-vision-announces-successful-phase-i%E2%80%90ii-study-topical-ev06-treatment-presbyopia/. Accessed September 29, 2016.
14. Zhao L, Chen XJ, Zhue J, et al. Lanosterol reverses protein aggregation in cataracts. Nature. 2015;523:607-611.
15. Life tables for the United States Social Security Area 1900-2100. Social Security Administration. https://www.ssa.gov/oact/NOTES/as120/LifeTables_Body.html. Accessed September 29, 2016.





