



PICKING THE RIGHT PLAY
An effort to find the best method of refractive correction after PKP and PRK.
By Rory M. Murphy; Kirk Stephenson, MB, BCh, BAO; Tim Fulcher, MB, MMed Sc, EBOD, FRCOphth; and Ian J. Dooley, MB, BAO, BCh, FRCSI, MRCOphth, MSc(Hons)
A 52-year-old woman presented to our institution for refractive correction of postoperative myopic astigmatism in her left eye. She had undergone successful PKP for progressive keratoconus in 1976 at age 18, followed by PRK for correction of residual astigmatism in 1998.
Her right eye, which demonstrated significant apical scarring with poor vision and had been treated with PRK, is not discussed further in this case report. In her left eye, preoperative UDVA and CDVA were 20/200 and 20/30, respectively. Her refraction was -7.00 -6.00 X 40º, yielding a spherical equivalent (SEQ) of -10.00 D. Intolerance to contact lenses and peripheral distortion with glasses motivated the patient to seek alternative methods for refractive correction.
Preoperative Orbscan (Bausch + Lomb) imaging revealed considerable eccentric inferonasal steepening without stromal thinning (Figure 1). The anterior chamber depth was 4.22 mm, measured from the epithelium to the crystalline lens.
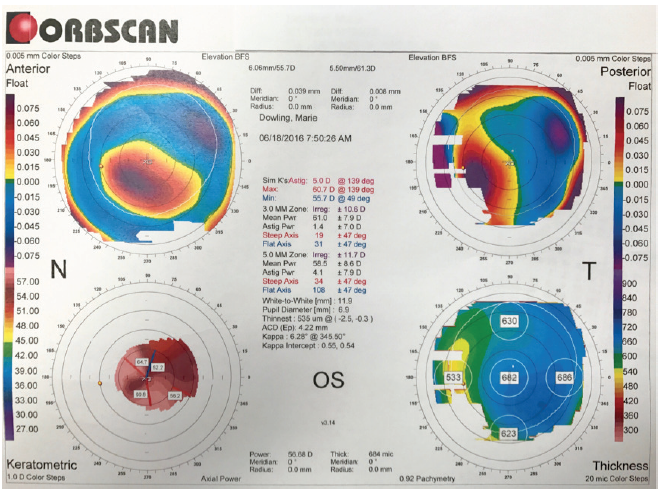
Courtesy of Rory M. Murphy
Figure 1. Preoperative Orbscan imaging demonstrating eccentric inferonasal corneal steepening anteriorly and posteriorly without corneal thinning.
We proceeded with uncomplicated insertion of an Artisan iris-fixated toric phakic IOL (Ophtec) with a spherical power of -8.50 D and a cylindrical power of -6.50 D, fixated at 40°, in the patient’s left eye. Postoperative UDVA at 12 months was 20/30-2, and CDVA was 20/30. SEQ at 12 months was -0.25 D, with refraction of +0.75 -2.00 X 125º.
We used the Alpins method for vector analysis, via the VECTrAK astigmatic vector calculator software (ASSORT).1,2 Treatment-induced astigmatism was 4.75 D, and surgically induced astigmatism (SIA) was 6.72 D. The difference vector was 1.99 X 125°, with an angle of error of -1°. The correction index was 1.41, with an index of success of 0.42 and coefficient of adjustment of 0.71.
DISCUSSION
This case highlights the complex decision-making process in post-PKP eyes. Although PKP achieves a high degree of success in managing a number of corneal pathologies, a high percentage of eyes remain ametropic, and up to 30% develop postoperative astigmatism greater than 5.00 D.3-5
Regular astigmatism and minimal anisometropia can be managed successfully with glasses. Contact lenses are more appropriate for irregular astigmatism and higher degrees of ametropia; however, their long-term use often results in eventual intolerance or surface complications, as in this case. Assuming tectonic and refractive stability, one has an array of possible options for surgical correction of residual astigmatism, which can largely be divided into corneal surface procedures and intraocular surgery.
Corneal surface procedures. Corneal surface procedures include incisional surgeries (transverse or arcuate keratotomies), PRK, and LASIK. Recently, the advent of femtosecond laser has enhanced the accuracy and predictability of refractive outcomes compared with manual or mechanical arcuate keratotomy, although results are more favorable in the correction of regular astigmatism.6,7 Irregular astigmatism can be effectively managed with either PRK or LASIK, although their range of correction is less than that with intraocular options.
Intraocular surgery. Intraocular treatment options include phakic IOLs or refractive lens exchange, both of which offer rapid visual recovery and a wide range of refractive correction. Although phakic IOLs can provide young patients with continuation of accommodation for UNVA and UDVA, for our patient discussed here at 52 years of age, presbyopia has already manifested, and the relative merits of these procedures for this purpose are lessened.
Phakic IOLs may be placed in the posterior chamber (eg, Visian Toric ICL; STAAR Surgical) or the anterior chamber (eg, angle- or iris-fixated), as in our case. Iris-fixated IOLs have been in use for more than 10 years and have demonstrated satisfactory long-term safety, particularly with respect to endothelial cell count (ECC).8 This is particularly relevant after PKP, when there is typically a low ECC—although some studies have shown faster ECC loss in the presence of a phakic IOL in eyes with previous PRK.9,10 Precise pupil centration and high rotational stability are attainable, although enclavation requires more surgical sills than insertion of a posterior chamber toric IOL.
Preoperative considerations for an iris-fixated lens include ECC (>2,100 cells/mm2), iris configuration (flat), scotopic pupil diameter (>6 mm), and anterior chamber depth (≥2.6 mm). All of the relevant contraindications to these criteria can be overcome with a posterior chamber phakic IOL.11-14
Tahzib et al reported results in 36 eyes with phakic IOL insertion after PKP.15 They observed a reduction in refractive cylinder from -7.06 ±2.01 D (range, -3.00 to -11.00 D) preoperatively to -1.69 ±1.15 D at 1 year.15 Mean SEQ was reduced from -3.19 ±4.31 D (range, +5.50 to -14.25 D) preoperatively to -1.03 ±1.20 D (range, +1.00 to -5.25 D) at 1 year. Our refractive outcomes compare favorably. One of the patients in the Tahzib et al study experienced endothelial decompensation over the 3 years of follow-up; this highlights the importance of setting realistic expectations in preoperative counseling and in the informed consent process.15
CONCLUSION
In carefully selected cases, implanting an Artisan toric phakic IOL can provide good visual outcomes in post-PRK eyes with high compound myopic astigmatism. The procedure is reversible, avoids inherent complications of surface ablations, and maintains the structural integrity of the cornea.
Up to 11% of corneas treated with PKP for keratoconus are diagnosed with progressive ectasia 20 to 25 years postoperatively, with a mean latency of 10 years.16 This is particularly relevant in a young patient with previous PKP for keratoconus, who should live long enough for there to be risk of recurrent keratoconus in the donor graft, making implantation of a toric phakic IOL an attractive option.
Our patient achieved good refractive and visual outcomes despite largely irregular preoperative astigmatism, and the graft continues to function in good health.
1. Alpins NA. New method of targeting vectors to treat astigmatism. J Cataract Refract Surg. 1997;23(1):65-75.
2. Alpins N. Astigmatism analysis by the Alpins method. J Cataract Refract Surg. 2001;27(1):31-49.
3. Olson RJ, Pingree M, Ridges R, Lundergan ML, Alldredge C, Clinch TE. Penetrating keratoplasty for keratoconus: a long-term review of results and complications. J Cataract Refract Surg. 2000;26(7):987-991.
4. Troutman RC, Lawless MA. Penetrating keratoplasty for keratoconus. Cornea. 1987;6(4):298-305.
5. Javadi MA, Motlagh BF, Jafarinasab MR, et al. Outcomes of penetrating keratoplasty in keratoconus. Cornea. 2005;24(8):941-946.
6. Kumar NL, Kaiserman I, Shehadeh-Mashor R, Sansanayudh W, Ritenour R, Rootman DS. IntraLase-enabled astigmatic keratotomy for post-keratoplasty astigmatism: on-axis vector analysis. Ophthalmology. 2010;117(6):1228-1235.e1221.
7. Viswanathan D, Kumar NL. Bilateral femtosecond laser-enabled intrastromal astigmatic keratotomy to correct high post-penetrating keratoplasty astigmatism. J Cataract Refract Surg. 2013;39(12):1916-1920.
8. Tahzib NG, Nuijts RM, Wu WY, Budo CJ. Long-term study of Artisan phakic intraocular lens implantation for the correction of moderate to high myopia: ten-year follow-up results. Ophthalmology. 2007;114(6):1133-1142.
9. Moshirfar M, Barsam CA, Parker JW. Implantation of an Artisan phakic intraocular lens for the correction of high myopia after penetrating keratoplasty. J Cataract Refract Surg. 2004;30(7):1578-1581.
10. Al-Dreihi MG, Louka BI, Anbari AA. Artisan iris-fixated toric phakic intraocular lens for the correction of high astigmatism after deep anterior lamellar keratoplasty. Digit J Ophthalmol. 2013;19(2):39-41.
11. Iovieno A, Guglielmetti S, Capuano V, Allan BD, Maurino V. Correction of postkeratoplasty ametropia in keratoconus patients using a toric implantable collamer lens. Eur J Ophthalmol. 2013;23(3):361-367.
12. Alfonso JF, Lisa C, Abdelhamid A, Montés-Micó R, Poo-López A, Ferrer-Blasco T. Posterior chamber phakic intraocular lenses after penetrating keratoplasty. J Cataract Refract Surg. 2009;35(7):1166-1173.
13. Aerts AA, Jonker SM, Wielders LH, et al. Phakic intraocular lens: two-year results and comparison of endothelial cell loss with iris-fixated intraocular lenses. J Cataract Refract Surg. 2015;41(10):2258-2265.
14. Doors M, Cals DW, Berendschot TT, et al. Influence of anterior chamber morphometrics on endothelial cell changes after phakic intraocular lens implantation. J Cataract Refract Surg. 2008;34(12):2110-2118.
15. Tahzib NG, Cheng YY, Nuijts RM. Three-year follow-up analysis of Artisan toric lens implantation for correction of postkeratoplasty ametropia in phakic and pseudophakic eyes. Ophthalmology. 2006;113(6):976-984.
16. Raecker ME, Erie JC, Patel SV, McLaren JW, Hodge DO, Bourne WM. Long-term keratometric changes after penetrating keratoplasty for keratoconus and Fuchs endothelial dystrophy. Am J Ophthalmol. 2009;147(2):227-233.

WEIGHING THE OPTIONS
Learning points from a hard-to-navigate case.
By Sumitra Khandelwal, MD
A 61-year-old man presented with blurry vision after cataract extraction with toric IOL implantation in his left eye 2 years earlier. The patient reported that his vision was blurry after surgery and did not improve, so his surgeon performed an Nd:YAG capsulotomy in that eye 3 months after surgery. Following the laser treatment, his vision was better with glasses but worse without.
The patient’s preoperative topography was not available, but postoperative topography was obtained. These topographic maps showed a steep cornea with symmetric bowtie astigmatism: 46.14/47.00 D @ 157º on the Galilei (Ziemer; Figure 2) and 45.83/48.15 D @ 159º on the Atlas (Carl Zeiss Meditec; Figure 3) topographers.
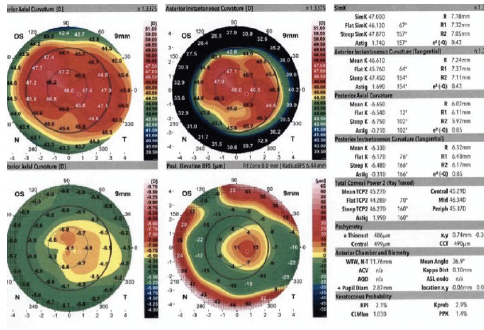
Courtesy of Sumitra Khandelwal, MD
Figure 2. Postoperative topography of the patient’s left eye.
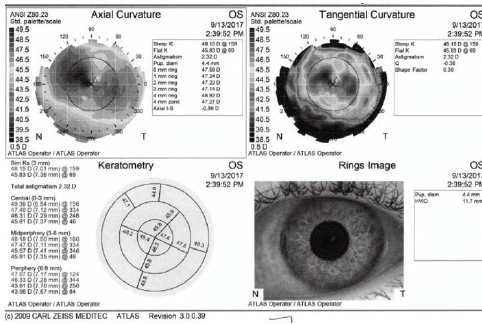
Courtesy of Sumitra Khandelwal, MD
Figure 3. Postoperative topography of the patient’s left eye.
The patient’s lens card indicated that he had a 3.50 D toric IOL (STAAR Surgical) placed at the time of surgery. Examination at the slit lamp revealed a silicone one-piece plate-haptic toric IOL oriented at 105° with a large posterior capsular opening. The patient’s refraction was -5.00 +3.75 X 175º with 20/20 CDVA.
DISCUSSION
This case presents multiple learning points. First, the STAAR Surgical toric IOL is a one-piece silicone plate-haptic lens that was the first approved in the United States and has excellent optics. Due to its silicone material, however, this IOL has a tendency to rotate. Additionally, its plate haptic may cause more instability in the bag compared with lenses with rigid haptics. It is unknown what the IOL position was after the surgery, but it is possible that the lens rotated after the Nd:YAG capsulotomy, as the patient noted worse uncorrected vision following the laser treatment. The patient desired distance vision if possible.
We entered the patient’s data into the Berdahl-Hardten calculator (astigmatismfix.com), and the recommendation was to rotate the lens 128° clockwise to minimize the existing astigmatism (Figure 4). However, even with this rotation, the anticipated residual refraction would yield a -3.00 D SEQ. Additionally, the plate haptics were fused, so rotation of the lens would have been difficult with such a large posterior capsular opening and peripheral anterior posterior capsular fusing.
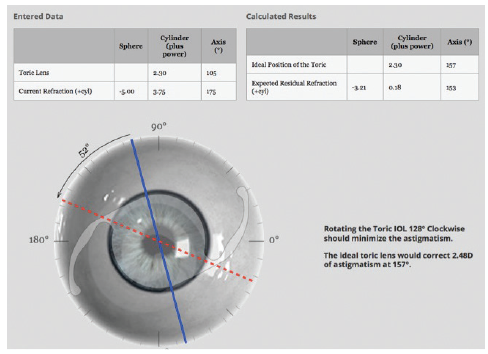
Courtesy of Sumitra Khandelwal, MD
Figure 4. The recommendation provided by the Berdahl- Hardten calculator was to rotate the lens 128° clockwise to minimize the astigmatism.
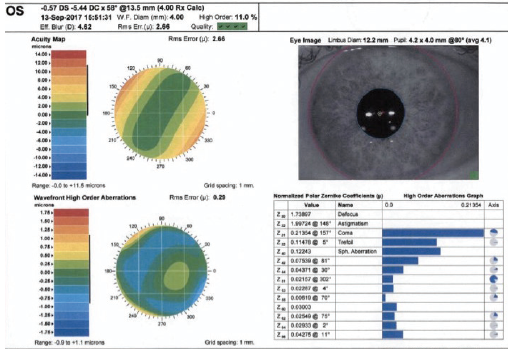
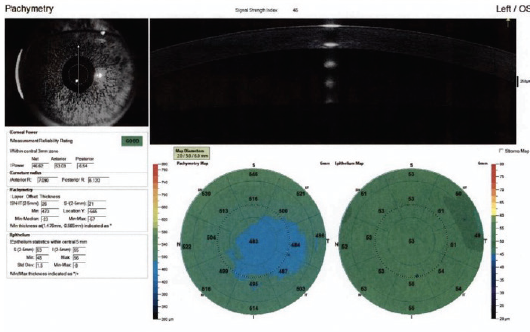
Another option was surface ablation, although the amount of cylinder correction was higher than usual. The correction was even higher on the wavefront map that was captured (Figure 5). Additionally, topography and OCT epithelial thickness mapping revealed thin corneas (Figure 6).
Courtesy of Sumitra Khandelwal, MD
Figure 5. The amount of cylinder to be corrected was higher than usual on the wavefront map.
Courtesy of Sumitra Khandelwal, MD
Figure 6. Topography and OCT epithelial thickness mapping revealed thin corneas.
We elected to remove the silicone lens and place a monofocal three-piece IOL in the sulcus with a slightly myopic refractive target to avoid mixed astigmatism. The patient subsequently underwent surface ablation (conventional treatment) for a target refraction of -3.75 +1.75 X 165º. The patient did well after surgery, with a refraction of -0.25 +0.50 X 170º at 3-month follow-up and anticipated improvement in the coming months.



POST-LASIK ECTASIA
Visual function and spectacle independence can be restored using a variety of surgical approaches.
By Daniel Elies, MD; Mercè Morral, MD; and José L. Güell, MD, PhD
Corneal ectasia after LASIK is a progressive steepening and thinning of the cornea associated with increasing astigmatism. This condition results from the loss of biomechanical integrity of the cornea and can cause a reduction in UCVA and BCVA.1,2 With improved understanding of the risk factors and the limits of excimer laser ablation, the current incidence of post-LASIK ectasia is estimated to be between 0.04% and 0.20%.
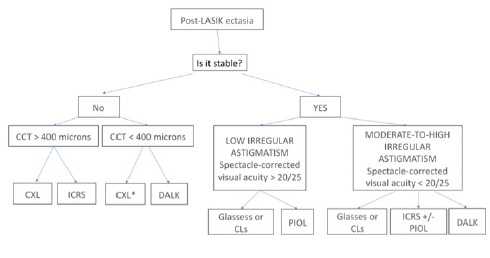
Management of post-LASIK ectasia and its associated astigmatism is often challenging. It is dependent on the patient’s CDVA with glasses and with rigid contact lenses, the degree of corneal irregularity induced by surgery, and the stability of the patient’s corneal ectasia. Figure 7 summarizes our stepwise approach for treating post-LASIK ectasia.
Images courtesy of Daniel Elies, MD
Figure 7. Treatment algorithm for ectasia after corneal refractive ablative procedures. CCT, central corneal thickness; ICRS, intrastromal corneal ring segment; CLs, contact lenses; PIOL, phakic IOL; DALK, deep anterior lamellar keratectomy.
The following case report demonstrates full visual rehabilitation in a patient with post-LASIK ectasia, resulting in complete spectacle independence.
CASE REPORT
In February 2008, a 23-year-old ophthalmic surgeon underwent bilateral myopic LASIK for low myopia (-2.50 D OD, -2.75 D OS). Three years later, he developed post-LASIK ectasia in both eyes and was treated elsewhere with intrastromal corneal ring segment (ICRS) implantation in his right eye in July 2012 and CXL in his left eye in October 2013.
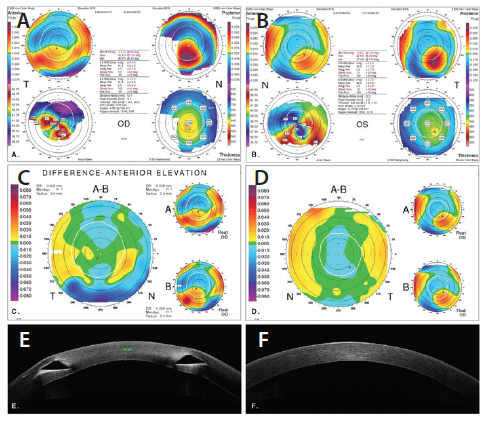
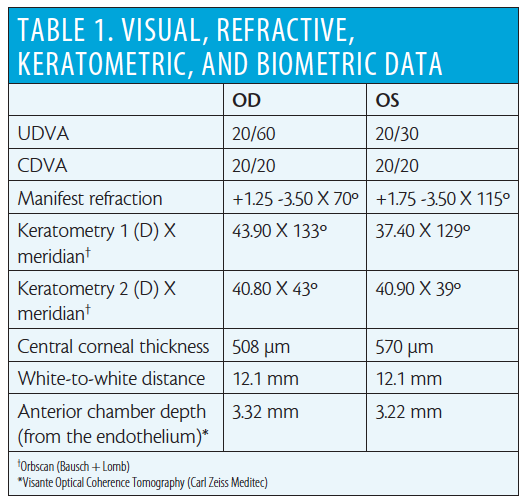
Upon presentation to our clinic in October 2015, the patient reported that his manifest refraction had been stable for several years, he had become contact lens–intolerant, and he desired to be independent of glasses once again. Corneal topography showed corneal ectasia in both eyes, and this remained stable at a second visit 6 months later (Figure 8). The LASIK flap interface was not clearly visible on anterior segment OCT, so residual stromal thickness (RST) and flap thickness could not be unequivocally determined. Table 1 summarizes the visual, refractive, keratometric, and biometric data for both eyes. Corneal ECCs were within normal limits in both eyes.
Images courtesy of Daniel Elies, MD
Figure 8. Orbscan corneal topography showing corneal ectasia after LASIK in the patient’s right eye (A) and left eye (B). Corneal anterior elevation difference maps between two measurements 6 months apart show stability of the corneal ectasia in both eyes (C, D). Anterior segment OCT shows an inferior ICRS OD and clear cornea OS (E, F).
Because the patient had CDVA of 20/20 OU and stable refraction and keratometry, we considered post-LASIK ectasia progression to be extremely unlikely. Therefore, we planned to perform bilateral implantation of the Visian Toric ICL. The IOL powers chosen were -3.00 +4.00 X 163º OD and -1.50 +4.00 X 24º OS. Under topical anesthesia, a 2.8-mm temporal incision was performed, and an Evo Visian ICL was implanted in each eye uneventfully. No peripheral iridotomy is required with the myopic Evo ICL. One week after surgery, the patient’s UCVA was 20/20 OU, and it has remained unchanged since then.
DISCUSSION
Post-LASIK ectasia is a serious complication that can occur even in the absence of known risk factors. It may arise from a variety of causes, including weak corneas before surgery, an RST too low to maintain structural integrity, trauma, and predisposition for keratoconus development.3 Patients who develop ectasia without any of these risk factors tend to be younger.
Because we did not have access to the patient’s preoperative topography or intraoperative data, we could not determine whether any preexisting corneal abnormality was the cause for his corneal ectasia. Additionally, because we could not definitively determine his flap thickness and RST with anterior segment OCT, we do not know whether undesirably thick flaps left insufficient RST.
Studies have demonstrated improved accuracy, uniformity, and predictability with flaps created with the femtosecond laser compared with the mechanical microkeratome. Therefore, the probability of creating a thicker flap than planned, resulting in a thinner RST and a higher risk of ectasia, is much lower with the femtosecond laser. It seems advisable to use a femtosecond laser for flap creation, especially in patients with moderate to high myopia and thin corneas.
Thorough preoperative assessment is key to minimize the risk of ectasia. All patients should be counseled about the potential risk of ectasia, even in the absence of known risk factors. Surgeons should also bear in mind that some patients may be predisposed to the development of keratoconus.
CONCLUSION
Several surgical strategies may be required to visually rehabilitate patients with post-LASIK ectasia. CXL is the only technique that has proven effective to stop the progression of ectasia, and the role of ICRSs remains controversial.
Once the patient’s ectasia is stabilized, hybrid or scleral contact lenses can help to achieve the best potential visual acuity and visual quality. Surgical options may be considered when patients become contact lens–intolerant or when CDVA is 20/20 with good quality of vision, as in the case of our patient described here.
1. Saad A, Binder MS, Gatinel D. Evaluation of the percentage tissue altered as a risk factor for developing post-laser in situ keratomileusis ectasia. J Cataract Refract Surg. 2017;43:946-951.
2. Seiler T, Koufala K, Richter G. Iatrogenic keratectasia after laser in situ keratomileusis. J Refract Surg. 1998;14:312-317.
3. Condon PI. 2005 ESCRS Ridley Medal Lecture: Will keratectasia be a major complication for LASIK in the long term? J Cataract Refract Surg. 2006;32:2124-2132.
4. Farjo AA, Sugar A, Schallhorn SC, et al. Femtosecond lasers for LASIK flap creation. A report by the American Academy of Ophthalmology. Ophthalmology. 2013;120:e5-e20.
5. Güell JL, Morral M, Malecaze F, et al. Collagen crosslinking and toric iris-claw phakic intraocular lens for myopic astigmatism in progressive mild to moderate keratoconus. J Cataract Refract Surg. 2012;38(3):475-484.


TOPO-GUIDED REFRACTIVE SURGERY IN COMA-DOMINATED CORNEAL OPTICS
Significant discrepancy between subjective astigmatism and objectively measured ocular astigmatism can occur.
By Aleksandar Stojanovic, MD, PhD; and Wen Zhou, MD
It is common for coma-like aberrations to be dominant in the corneal optics of orthogonally asymmetric corneas, as in keratoconus1,2 and corneal ectasia caused by LASIK.3 However, these aberrations may also occur with other types of corneal pathology.4-6
In the presence of significant higher-order aberrations (HOAs), manifest refraction cannot represent the true ocular spherocylindrical error due to the contribution of HOAs that may refract as astigmatism or sphere. The influence of coma-like HOAs on subjective astigmatism as measured by manifest refraction has been described.7,8 Further, it has been suggested that vertical coma refracts as against-the-rule astigmatism, whereas horizontal coma refracts as with-the-rule astigmatism.9
When topography-guided custom refractive surgery is used, corneal HOAs are treated by corneal surface regularization. If, in that situation, subjective astigmatism—which contains a component from coma refracting as astigmatism—is chosen as the astigmatism treatment endpoint, treatment of the coma itself and its astigmatic effect within subjective astigmatism will both occur. This results in double treatment of the astigmatism caused by coma. Therefore, we recommend that subjective astigmatism be disregarded, and total corneal astigmatism (TCA; derived from both the anterior and the posterior corneal astigmatism) should be treated along with corneal HOAs.
This case report describes a clinical case managed using this strategy.
CASE REPORT
A 58-year-old man presented to our clinic complaining of decreased corrected visual acuity and visual disturbances in the form of multiple images. Irregular astigmatism secondary to map-dot-fingerprint corneal dystrophy was diagnosed in his left eye.
The patient’s UDVA and CDVA OS were 20/63 and 20/50, respectively. Manifest refraction was +0.50 -3.00 X 17º. Scheimpflug topography and tomography (Precisio; iVIS Technology) showed an asymmetric anterior elevation map within the pupillary area, with increased superotemporal elevation and inferonasal depression (Figure 9). The axial map demonstrated an orthogonally asymmetric pattern (Figure 9).
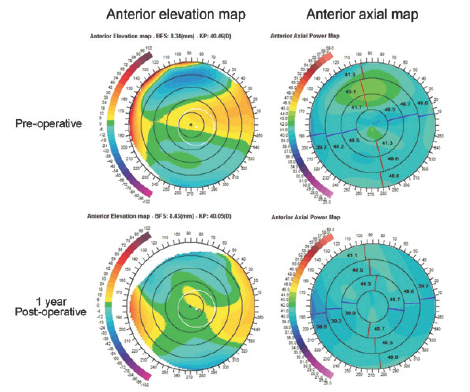
Courtesy of Aleksandar Stojanovic, MD
Figure 9. Scheimpflug topography and tomography showed asymmetric anterior elevation within the pupillary area, with increased superotemporal elevation and inferonasal depression. The axial map demonstrated an orthogonally asymmetric pattern.
Uneven epithelial thickness was revealed by both slit-lamp examination and anterior segment OCT with the RTVue-100 (OptoVue; Figure 10). Increased ocular HOAs, as measured with the OPD-Scan II (Nidek), are shown in Table 2. We measured and calculated the patient’s TCA and lenticular astigmatism and estimated the contribution of coma-like HOAs to subjective astigmatism by methods we introduced in a previous study.9
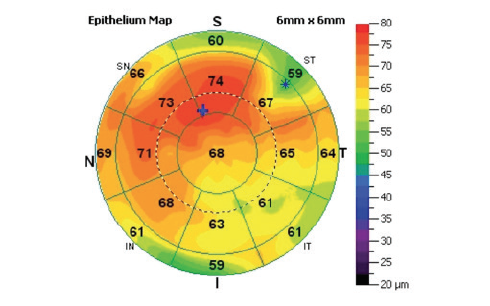
Courtesy of Aleksandar Stojanovic, MD
Figure 10. Uneven epithelial thickness was revealed on both slit-lamp examination and anterior segment OCT.
Anterior corneal elevation maps obtained with the Precisio device were used as the basis for custom ablation design with Corneal Interactive Programmed Topographic Ablation software (iVIS Technology). The presence of so-called discrepancy astigmatism, representing the discrepancy between the subjective astigmatism and the sum of the TCA and lenticular astigmatism (Table 2), assumed that the coma-like HOAs influenced the subjective astigmatism.
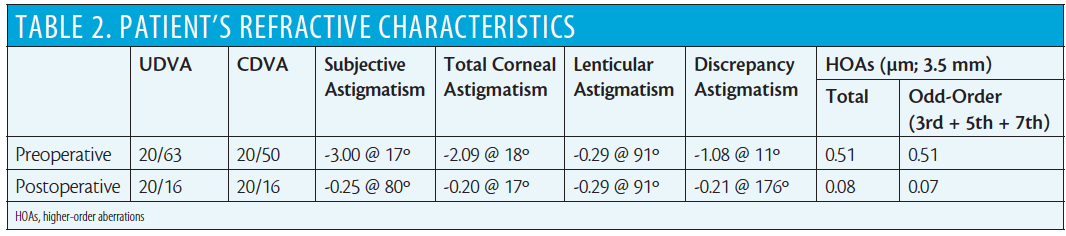
We planned to treat both TCA and the anterior corneal surface irregularities (the source of anterior corneal HOAs).
At 1-year postoperative, the patient’s UDVA had improved to 20/16. Manifest refraction was plano -0.25 X 80º. Significant topographic regularization (Figure 1) and reduction in HOAs (Table 2) were achieved. The patient was satisfied with his postoperative vision and reported overall good quality of vision with no disturbances.
DISCUSSION
Significant discrepancy between subjective astigmatism and objectively measured ocular astigmatism is known to occur in conjunction with corneal pathologies that cause coma-dominated optics.7-9 In visual assessment at the phoropter, the subjectively refracted cylinder power and axis will be a vector sum of two components: one caused by the real astigmatism (ie, ocular astigmatism), which is mainly derived from the cornea and the lens, and the other caused by coma subjectively refracting as cylinder.
In the case described here, the patient’s TCA and estimated lenticular astigmatism were 2.09 @ 18º and 0.29 @ 91º, respectively, resulting in objective ocular astigmatism of 1.86 @ 21º. However, subjective astigmatism at the corneal plane was 2.93 @ 17º, indicating a discrepancy of 1.08 D vector difference. Regarding the asymmetric anterior corneal topography and coma-dominant corneal optics, we estimated that the coma was causing the discrepancy.
For such a case, when corneal HOAs and TCA are treated by topography-guided refractive surgery, all sources of subjective astigmatism except for lenticular astigmatism—ie, anterior and posterior corneal astigmatism and coma refracting as astigmatism—are addressed. If we had chosen to treat subjective astigmatism along with the corneal HOAs, then the corneal coma itself, as a part of the corneal HOAs and its effect on subjective astigmatism, would both have been treated. That would have amounted to a double treatment of the subjective astigmatism component caused by coma: that is, removal of the cause and, at the same time, the effect of its treatment. As a result, a new, unexpected astigmatism would have been induced.
In the current case, the strategy of regularizing the corneal surface by topography-guided ablation and treatment of TCA was used, resulting in minimal postoperative TCA.
The main shortcoming of the strategy chosen is the potential for error in combining the data acquired by two separate instruments (Precisio and OPD Scan), using three different types of technologies (Scheimpflug and Placido-disc–based topographies and OPD-based wavefront aberrometry) for calculation of the lenticular astigmatism. In addition to the registration error that may occur between two separate examinations, one must take into consideration the potential error due to compatibility issues. Ideally, a single instrument, using one type of technology, should be used for the measurement of lenticular astigmatism.
A correct estimation of a patient’s lenticular astigmatism may have a significant impact, as the current approach is based on the treatment of TCA instead of the subjective astigmatism, and the lenticular astigmatism is chosen to be left untreated. However, the astigmatic power of the lens, which constitutes a dynamic live tissue, has been precisely measured only in vitro,10 and, to our knowledge, solid clinical research in this respect is lacking.
Surgeons should be mindful of the influence of coma-like HOAs on subjective astigmatism. In cases like this one, we suggest using a topography-guided custom ablation, choosing to correct the TCA independent of the subjective astigmatism along with corneal HOAs. This is our preferred treatment strategy for dealing with coma-like aberrations dominating the corneal optics, especially if the lenticular astigmatism is estimated to be insignificant.
1. Maeda N, Fujikado T, Kuroda T, et al. Wavefront aberrations measured with Hartmann-Shack sensor in patients with keratoconus. Ophthalmology. 2002;109(11):1996-2003.
2. Saad A, Gatinel D. Evaluation of total and corneal wavefront high order aberrations for the detection of forme fruste keratoconus. Invest Ophthalmol Vis Sci. 2012;53(6):2978-2992.
3. Pinero DP, Alio JL, Barraquer RI, Uceda-Montanes A, Murta J. Clinical characterization of corneal ectasia after myopic laser in situ keratomileusis based on anterior corneal aberrations and internal astigmatism. J Cataract Refract Surg. 2011;37(7):1291-1299.
4. McCormick GJ, Porter J, Cox IG, MacRae S. Higher-order aberrations in eyes with irregular corneas after laser refractive surgery. Ophthalmology. 2005;112(10):1699-1709.
5. Pesudovs K, Figueiredo FC. Corneal first surface wavefront aberrations before and after pterygium surgery. J Refract Surg. 2006;22(9):921-925.
6. Leung TW, Lam AK, Kee CS. Ocular aberrations and corneal shape in adults with and without astigmatism. Optom Vis Sci. 2015;92(5):604-614.
7. Wei RH, Lim L, Chan WK, Tan DT. Higher order ocular aberrations in eyes with myopia in a Chinese population. J Refract Surg. 2006;22(7):695-702.
8. Alpins NA. Treatment of irregular astigmatism. J Cataract Refract Surg. 1998;24(5):634-646.
9. Zhou W, Stojanovic A, Utheim TP. Assessment of refractive astigmatism and simulated therapeutic refractive surgery strategies in coma-like-aberrations-dominant corneal optics. Eye Vis. 2016;3:13.
10. Augusteyn RC. On the growth and internal structure of the human lens. Exp Eye Res. 2010;90(6):643-654.



