CASE PRESENTATION
A 73-year-old woman reports that, at the age of 5, she suffered an injury to her right eye with scissors, leading to a scleral rupture and subsequent repair. The patient says she had “pretty good” vision in that eye until a couple of years ago. Recently, she received radiation treatment for a squamous cell cancer on the right side of her nose, and she has noticed a significant decrease in her vision since treatment, which she completed 4 months ago. On examination, the patient’s BCVA measures -0.25 +1.00 X 120º = 20/50 OD and plano +0.75 X 003º = 20/25 OS. A slit-lamp examination of the patient’s right eye reveals a clear cornea, with irregular conjunctiva nasally in the area of previous scleral rupture and repair. Synechiae are present nasal to the cornea, and areas of iris atrophy are visible in the same location (Figure 1). A 2+ nuclear sclerotic cataract is present, denser centrally, and no distinct phacodonesis is evident, although there is a suggestion of possible lenticular posterior tilt. It is difficult to determine whether this is true lenticular tilt or if it is due to the appearance of the synechiae. Results of a dilated retinal examination and OCT of the retina are normal.
How would you approach cataract surgery in this case? Which IOL options would you offer?
—Case prepared by Audrey R. Talley Rostov, MD
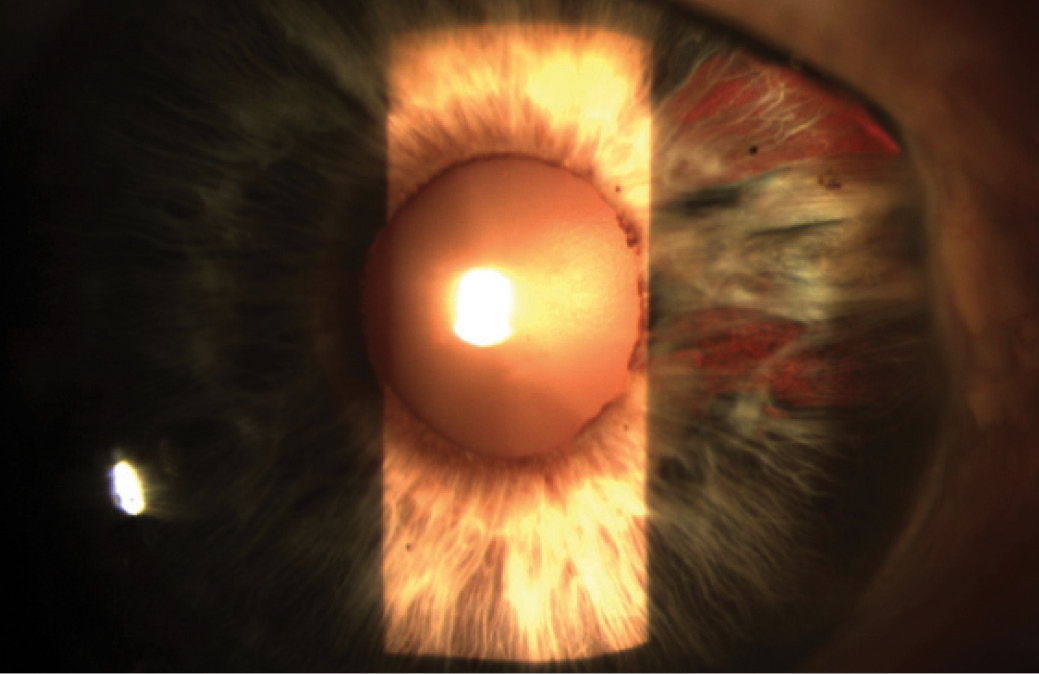
Figure 1. Conjunctival thickening nasally. Iris atrophy and nuclear sclerosis of the lens.

ZAINA AL-MOHTASEB, MD
The patient’s history of scleral rupture and the objective signs of iris transillumination defects, posterior synechiae, and potential lenticular tilt point to zonular loss in this case. Before proceeding with surgery, I would discuss with her the increased risk of complications and the potential need for a future surgery. Additionally, given that the patient’s initial trauma occurred when she was 5 years old, which is still in the amblyogenic age range, I would explain that her true visual potential after cataract surgery is unknown. The fact that her vision was better before the cataract developed means that she likely has good visual potential. Because of her history, I would also carefully evaluate the ocular surface and explain the risks of radiation.
In preparation for potential surgical complications, I would use trypan blue and have capsular tension rings and segments (CTRs and CTSs) in the room, given the possibility of zonular loss. I would perform an intracameral injection of epinephrine to aid in pupillary dilation and use the OVD cannula to break up the posterior synechiae, while being careful not to tear the anterior capsule. If the pupil still did not dilate well, I would use iris hooks.
I would be cautious while performing the capsulorrhexis and look for zonular loss (abnormal wrinkling of the capsule or movement of the lens). If I observed any, then I would stabilize the lens with capsular hooks while carefully proceeding with hydrodissection, phacoemulsification, and cortical cleanup. I would place a CTR at the necessary time. (If placed prior to cortex removal, then I would perform viscodissection first.) Depending on the amount of zonular loss, I would either insert a CTR or suture two CTSs in addition to passing Gore-Tex sutures (W.L. Gore & Associates; off-label use) through sclerotomies 2 mm posterior to the limbus.
Should an anterior and posterior tear occur, or if I were unable to save the bag because of significant zonulopathy, then I would proceed with double-needle transscleral fixation of a three-piece IOL using the Yamane technique.1 Other reasonable options are iris suturing, sclera suturing, and—based on the patient’s age—an anterior chamber IOL. My colleagues and I recently reported similar visual outcomes with all three methods.2
If the patient had complained of significant glare secondary to iris defects before developing a cataract, I would have considered repairing the iris with 9-0 polypropylene sutures. Postoperatively, I would carefully evaluate the retina because of her history of trauma.

MARJAN FARID, MD
Although the central nuclear sclerotic cataract is likely related to the patient’s age and history of radiation treatment, I would be concerned about sectoral zonular weakness from the pediatric trauma and ruptured globe repair. The sectoral iris trauma, possible lens tilt, and synechiae would raise my suspicion for zonulopathy.
Prior to starting the case, I would make sure to have on hand iris expanders, a CTR or CTS, capsular hooks, suture material, and an OVD with a heavy molecular weight. An iris expander or hooks might be required if the area of iris atrophy does not dilate well. I would use a heavy cohesive OVD because zonular loss in one segment can make capsulorrhexis formation more challenging, owing to a lack of tension on the capsule. I think a laser capsulotomy would be an excellent option in this case because the capsulotomy could be centered over the entire lens complex rather than the pupil.
If the entire lens-capsule complex has shifted, I would use capsular hooks to recenter the entire complex prior to phacoemulsification. If there is traumatic zonulopathy, I would recommend phaco chop with minimal manipulation or presoftening with a femtosecond laser to facilitate nuclear removal with minimal tension on the capsule. It might be necessary to place a CTR or CTS to center the capsule prior to lens implantation.
My preference would be a three-piece IOL because the haptics offer spring-like capsular support and are versatile for suturing to the iris or fixating to the sclera, if needed. It may prove necessary to repair the area of iris atrophy to minimize postoperative glare.

RUSSELL SWAN, MD
Given the recent decline in vision and radiation therapy, I would perform preoperative visual field testing to rule out an optic neuropathy. I would also emphasize to the patient the risk of surgery, given her history of trauma and potential zonular and capsular instability.
If she chose to proceed, I would approach the case in a routine manner with topical and intracameral anesthesia and oral sedation with an MKO Melt (midazolam, ketamine HCl, ondansetron, lemon; Imprimis Pharmaceuticals). Through a temporal incision, using a dispersive OVD, I would perform nasal synechialysis. I would use a Malyugin Ring (MicroSurgical Technology) for inadequate pupillary dilation.
I would use the Zepto Capsulotomy System (Mynosys Cellular Devices) because of the importance of an intact, well-sized capsulorrhexis in this case. Given the uncertainty of the peripheral capsule, I would avoid hydrodissection, perform hydrodelineation, and then debulk and disassemble the central nucleus. Next, I would viscodissect the capsule from the lens. Bimanual irrigation and aspiration would remove the remaining cortex.
I would be prepared to place a CTR or CTS if and when I encountered zonular instability, but I would check the status of the capsular bag prior to inserting the IOL. I would have on hand triamcinolone acetonide (Triesence; Alcon), equipment for a bimanual vitrectomy, and—in case scleral fixation proved necessary—an EC-3 PAL IOL (Aaren Scientific).
I would offer this patient a monocular aspheric IOL or a toric IOL if the astigmatism is regular and there are no intraoperative zonular issues. I would not recommend a multifocal or extended depth of focus IOL in this case because of the risk of late-onset radiation-induced optic neuropathy or retinopathy.

SHACHAR TAUBER, MD
When contemplating treatment options for what appears to be a visually significant cataract, it is important to consider the unique risk factors in this patient’s past medical history, which include a distortion of the ocular anatomy from a pediatric injury, the potential coexistence of amblyopia due to this injury, and the possibility of radiation-induced keratitis sicca. No information is provided regarding her other eye, so I cannot determine what role radiation has played in her ocular issues.
My approach would be to determine what “pretty good vision” was through a search of old records, if possible, to see if there was preexisting amblyopia. During the preoperative examination, I would undertake a thorough evaluation for dry eye disease and initiate appropriate therapy, if indicated. Confirmation of corneal astigmatism, whether regular or irregular, should be made with topography. By comparing measurements using anterior segment OCT, ultrasound biomicroscopy, and biometry for both eyes, I would determine the anterior chamber depth and angle and lens characteristics for the patient’s right eye. If the measurements differed between eyes, I would consider the possibility of phacodonesis.
Preparation for surgery would include a plan for adequate anesthesia. My inclination would be a sub-Tenon approach for this potentially complex surgery. Management of a small pupil would involve pharmacologic and/or mechanical dilation with hooks or rings. If adequate dilation can be achieved, I think that a femtosecond laser could be advantageous in creating a large capsulotomy of 5.5 mm centered on the pupil. Here, the benefits of having the laser in the OR become evident because I could place a ring or hooks and break up possible synechiae prior to using the laser for the capsulotomy and lens fragmentation. I would perform standard phacoemulsification and irrigation/aspiration of cortical material.
I would have CTRs and CTSs available. If the zonules and bag allow its proper placement, a standard one-piece lens would be my preference, but I would not hesitate to place a three-piece IOL with intrascleral fixation if required. The key would be to avoid the area of prior scleral laceration when extruding the haptics.
1. Yamane S, Sato S, Maruyama-Inoue M, Kadonosono K. Flanged intrascleral intraocular lens fixation with double-needle technique. Ophthalmology. 2017;124(8):1136-1142.
2. Brunin G, Sajjad A, Kim EJ, et al. Secondary intraocular lens implantation: complication rates, visual acuity, and refractive outcomes. J Cataract Refract Surg. 2017;43(3):369-376.



