For centuries, photographers and astronomers have applied the principle of small-aperture optics to their crafts. By the 1940s, ophthalmologists and optometrists started to take advantage of the concept in eye care. Today, small-aperture technology can improve the vision of presbyopic patients with and without cataracts using advanced approaches that extend depth of focus and reduce lower- and higher-order aberrations.
Channeling light through a small aperture blocks stray and unfocused rays, thereby sharpening vision and extending the depth of field. A small aperture can also minimize the impact of corneal aberrations.
SMALL-APERTURE STRATEGIES
Several small-aperture strategies have been proposed to treat presbyopia. They include topical miotic drops, inlays, and implants designed for the sulcus and capsular bag. These strategies are in various stages of use and development, and they are associated with a variety of outcomes.
Constricting drops. Several companies are exploring pharmacologic agents for pupillary constriction as a means of addressing presbyopia in emmetropic patients. The goal is to overcome the unwanted side effects of past strategies using miotic drops such as headache, nausea, and ciliary spasm.1,2 Although a noninvasive topical approach is attractive, daily administration could be unappealing to or difficult for patients. Any viable presbyopic drop formulation would have to be well-tolerated and long-lasting and have a rapid onset of action.
Several drops are currently being studied. Liquid Vision PRX-100 (Presbyopia Therapies) is a combination of aceclidine and tropicamide.3 Allergan has two drop candidates, AGN-199201 and AGN-190584, both of which are miotic agents.4 PresbiDrops/CSF-1 (Orasis Pharmaceuticals) contains an undisclosed parasympathomimetic to stimulate miosis and accommodation and an NSAID to prolong the effect.5
Inlays. The Kamra small-aperture intrastromal corneal inlay (CorneaGen) is intended for presbyopes who have a healthy crystalline lens. This removable device is a less-invasive option compared with IOL implantation. The inlay is implanted into a deep stromal pocket created by a femtosecond laser. The inlay’s safety and efficacy are documented, including in the recent publication of 3-year data from US clinical trials.6 Another large 4-year study involving nearly 3,000 patients found that fewer than 1% of Kamra inlays were removed due to haze or a refractive shift.7 Pearls for success include deep implantation, attention to ocular surface health, and a slightly myopic target in the inlay eye. Other inlays have been available (Raindrop, ReVision Optics, no longer available; and Flexivue Microlens, Presbia) in the past, but they have been removed from the market or are no longer available.
Sulcus implants. The XtraFocus IOL (Morcher) is a black, foldable hydrophobic acrylic implant intended for use in pseudophakic patients. The device is implanted in the ciliary sulcus in an add-on configuration with any monofocal IOL, including a toric lens. The implant is designed primarily to improve near visual acuity and manage irregular astigmatism in eyes with complex corneal aberrations, such as irregular astigmatism and high amounts of higher-order aberrations.8-11 (For more information on the XtraFocus, see “Pinhole Implant for Management of Irregular Astigmatism.”)
Pinhole Implant for Management of Irregular Astigmatism
An ancient principle can be useful in challenging cases.
By Claudio C. Trindade, MD, PHD

Small-aperture implants have recently drawn increased attention. However, the idea of using a small aperture in an IOL is not new. The first attempt to incorporate a pinhole aperture into an intraocular implant dates back to 1964, when renowned British ophthalmologist D. Peter Choyce, FRCS, FRCOphth, introduced the Mark V implant (Rayner), an anterior chamber PMMA IOL with an embedded pinhole mask.
Since that time, the concept of using a pinhole aperture to provide increased depth of field has been employed in intrastromal corneal implants and IOLs. In 2014, a proof-of-concept study of the XtraFocus Pinhole Implant (Morcher) was published.1 A fourth version of the XtraFocus Pinhole Implant received the CE Mark in 2016. Since then, the device has been commercially available in Europe, Australia, New Zealand, and some countries in Latin America and the Middle East.
DEVICE DETAILS
The device is made of a foldable black hydrophobic acrylic material. The optic has an occlusive portion with a diameter of 6 mm that contains a 1.3-mm clear central opening with no refractive power. The implant was designed to be placed in the ciliary sulcus of a pseudophakic eye in a piggyback fashion (Figure 1).
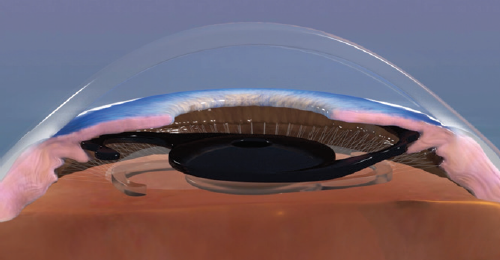
Figure 1. The implant was designed to be placed in the ciliary sulcus of a pseudophakic eye in a piggyback fashion.
The primary indication for the XtraFocus is for the treatment of irregular corneal astigmatism, for example in eyes after radial keratotomy, penetrating keratoplasty, and keratoconus (Figure 2), or in eyes with post-LASIK ectasia. Depth of focus extension, to provide near vision enhancement, is also a good indication, with the added benefit of reversibility.
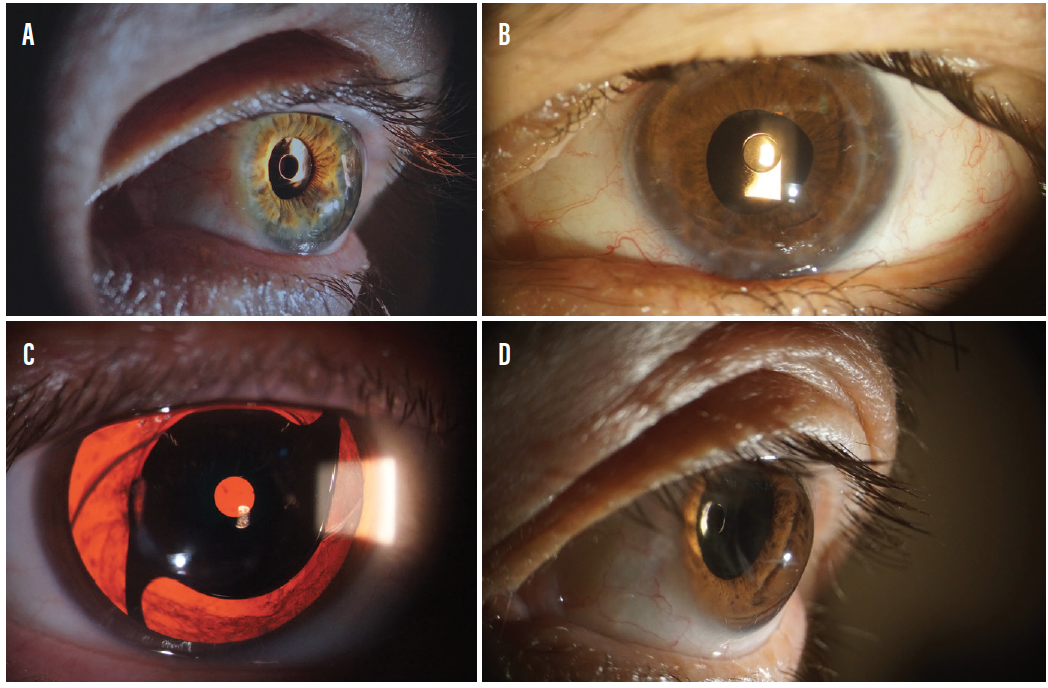
Figure 2. The XtraFocus in eyes after radial keratotomy (A), penetrating keratoplasty (B), and keratoconus (C, D).
The implant can be used with any other IOL, including toric versions. This is especially important in postpenetrating keratoplasty eyes and in patients with keratoconus. Although the pinhole effect can neutralize some regular astigmatism, in patients with a high magnitude of astigmatism the combination of the XtraFocus with a high-cylinder toric IOL can have a strong synergistic effect. Confirmation of topographic stability is mandatory in this scenario.
PATIENT SELECTION
Ideal patients are those with significant corneal aberrations with a transparent central cornea and a large pupillary diameter. A good example would be a cataract patient with keratoconus, mean keratometry of 58.00 D, central corneal thickness of approximately 380 µm, and a 4-mm pupil in mesopic conditions. Visual acuity in such a patient can achieve unexpectedly high levels, such as 20/30 UCVA.
To demonstrate postoperative acuity to the patient preoperatively, placing pinhole occlusion on top of the best refraction can provide a fair simulation. However, a favorable pinhole acuity test does not guarantee success. Corneal opacities (corneal dystrophies, scars, severe haze) represent a relative contraindication for this treatment.
Another important point to determine is the ideal refractive target. For an eye with irregular corneal astigmatism, a refractive target of -2.00 D is advisable. This provides solid uncorrected near visual acuity, while the extended depth of focus from the pinhole provides satisfactory uncorrected distance vision that can be further improved with glasses.
OTHER USES, PLUSES, AND MINUSES
The XtraFocus can also be used for the treatment of iris defects (Figure 3). Reduction of glare, ghost images, and light sensitivity can be expected with this approach.
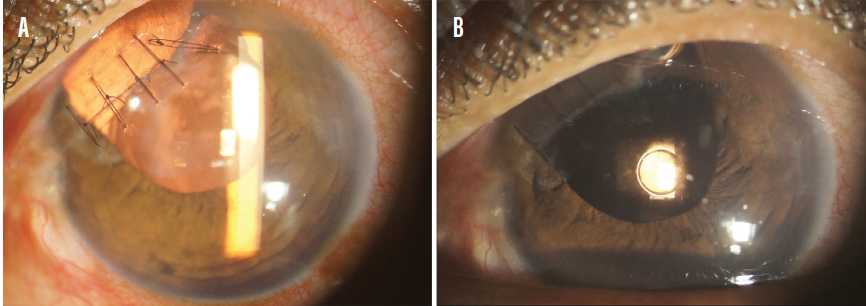
Figure 3. A patient who experienced penetrating ocular injury with iris loss before (A) and after (B) the XtraFocus was implanted.
Another interesting application for this implant is for the treatment of dysphotopsia after multifocal IOL implantation (Figure 4). The piggyback design provides a versatile solution when an IOL exchange might present significant risks, such as when a previous posterior capsulotomy is present.
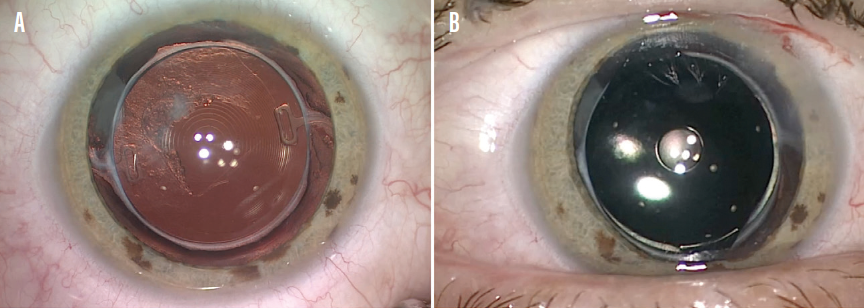
Figure 4. In an eye with dysphotopsia after multifocal IOL implantation (A), the XtraFocus implant can mitigate symptoms and preserve an extended range of functional vision (B).
The downside of pinhole implants is their inherent reduction of light entry. In some patients, this may translate into a sensation of dark vision under low-light conditions. This perception is usually well tolerated, and it is often outweighed by the remarkable improvement in visual acuity that good candidates experience. However, this is an important point to be discussed with patients preoperatively.
The black acrylic material has the unique feature of being transparent to infrared light, therefore allowing examination of structures located behind the implant with the use of infrared-based equipment (Figure 5). The occlusive body has a very slim profile (180 µm around the pinhole), with a concave-convex shape to avoid contact with the primary IOL. Because of the reduced thickness, the device can be implanted through a 2-mm corneal incision. It can also be safely implanted in the bag together with the primary IOL.

Figure 5. The black acrylic material is transparent to infrared light, allowing examination of structures located behind the implant using infrared-based equipment.
CONCLUSION
Although follow-up data are limited, the XtraFocus seems to be a promising new type of intraocular implant. Future studies will provide additional information.
1. Trindade CLC, Trindade BLC. Novel pinhole intraocular implant for the treatment of irregular corneal astigmatism and severe light sensitivity after penetrating keratoplasty. J Cataract Refract Surg Online Case Rep. 2015;3(1):4-7.
Suggested Reading:
• Trindade BLC, Trindade FC, Trindade CLC. Intraocular pinhole implantation for irregular astigmatism after planned and unplanned posterior capsule opening during cataract surgery. J Cataract Refract Surg. 2019;45(3):372-377.
• Trindade BLC, Trindade FC, Trindade CLC, Santhiago MR. Phacoemulsification with intraocular pinhole implantation associated with Descemet membrane endothelial keratoplasty to treat failed full-thickness graft with dense cataract. J Cataract Refract Surg. 2018;44(10):1280-1283.
• Trindade CC, Trindade BC, Trindade FC, Werner L, Osher R, Santhiago MR. New pinhole sulcus implant for the correction of irregular corneal astigmatism. J Cataract Refract Surg. 2017;43(10):1297-1306.
• Tsaousis KT, Werner L, Trindade CLC, Guan J, Li J, Reiter N. Assessment of a novel pinhole supplementary implant for sulcus fixation in pseudophakic cadaver eyes. Eye (Lond). 2018;32(3):637-645.
Capsular bag implants. The IC-8 IOL (AcuFocus) combines an aspheric monofocal IOL with an embedded opaque mini-ring to simultaneously address refractive error and presbyopia.
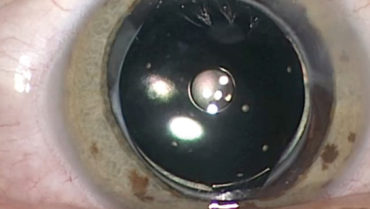
Implanted in the capsular bag at the time of cataract surgery (Figure), the one-piece hydrophobic acrylic IOL has modified C-loop haptics and an overall diameter of 12.5 mm. The biconvex aspheric optic is 6 mm in diameter with a square posterior edge for 360º. The embedded annular mask has an outer diameter of 3.23 mm and a central aperture diameter of 1.36 mm.
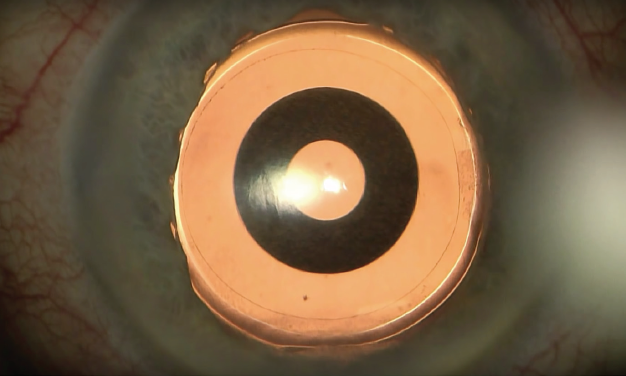
Figure. Intraoperative view of the IC-8 IOL after laser cataract surgery.
The IC-8 IOL has the CE Mark. Results from a multicenter postmarket European trial of 105 patients were recently published.12 Six months after implantation, monocular uncorrected distance visual acuity (UDVA) was 0.87 logMAR (20/23 Snellen), uncorrected intermediate acuity (UIVA) was 0.83 logMAR (20/24), and uncorrected near acuity (UNVA) was 0.66 logMAR (20/30).
With respect to binocular vision, 99%, 95%, and 79% of patients achieved 20/32 or better UDVA, UIVA, and UNVA, respectively.12 The majority of patients (95.9%) reported that they would have the procedure again. Most (84.8%) reported using spectacles occasionally to never. Binocular contrast sensitivity matched the monocular contrast sensitivity achieved in the contralateral eye with a monofocal IOL.12
Patients in this study and others have tolerated as much as 1.00 D of residual refractive error, suggesting that this small-aperture IOL may be more forgiving than monofocal and multifocal IOLs.12 Notably, astigmatic patients in a prospective clinical trial who each received an IC-8 IOL and no additional astigmatic management were able to tolerate up to 1.50 D of refractive astigmatism.13
COMPLEMENTARY TOOLS
The small-aperture approaches discussed in this article are complementary: Each addresses a unique patient group. For example, drops could be used as an interim solution for emmetropic presbyopes, but they will not address refractive error or cataracts. Drops could also be instilled to demonstrate small-aperture vision to patients considering a permanent implant.
Inlays can be used in patients with healthy corneas. Many surgeons routinely combine the Kamra inlay with laser vision correction in patients with sufficient corneal thickness. Patients who may not be good candidates for a Kamra inlay are those with uncontrolled moderate to severe dry eye. Ocular surface quality should be managed proactively and aggressively in order to achieve the best possible results.
Sulcus-based implants are reserved almost exclusively for pseudophakic patients whose corneas are highly aberrated. This technology can also benefit pseudophakic patients who need a wider range of vision than is provided by their currently implanted IOL.
A capsular bag implant such as the IC-8 IOL is suitable for a broad range of patients, including those with irregular corneas.14,15 The implant can be used in patients with naturally occurring corneal higher-order aberrations of 0.6 µm or more or after corneal refractive surgery and keratoplasty.
Patients with diseased corneas, keratoconus, or ocular trauma are also potential candidates for this technology. The majority of patients do well with this lens, but some patients with larger mesopic pupils (≥6 mm) may experience photic phenomena due to light coming around the outside edge of the opaque ring. As a result, it is important to evaluate pupil size when considering the IC-8 lens.
CONCLUSION
Small-aperture optics offer a dynamic, physiologic solution to the problem of presbyopia. The strategies described in this article can be effective for a range of accommodation loss and for pseudophakia. As a whole, the category of small-aperture optics will continue to expand—an indication of the needs of our patients who live more active and longer lives.
1. Karanfil FC, Turgut B. Update on presbyopia-correcting drops. European Ophthalmic Review. 2017;11(2):99-102.
2. Renna A, Alió JL, Vejarano LF. Pharmacological treatments of presbyopia: a review of modern perspectives. Eye Vis (Lond). 2017;4:3.
3. Lipner M. A unique drop. EyeWorld. https://www.eyeworld.org/article-a-unique-drop. Accessed May 21, 2019.
4. Pending presbyopia treatments edge closer to disrupting the marketspace. https://www.healio.com/ophthalmology/cornea-external-disease/news/print/ocular-surgery-news/%7Ba40b0eb9-cddf-4908-b9bb-9911bef60bf1%7D/pending-presbyopia-treatments-edge-closer-to-disrupting-the-marketspace. Accessed May 21, 2019.
5. Lindstrom RL. Update on the pharmacologic treatment of presbyopia. Paper presented at: Hawaiian Eye 2019; January 19-25, 2019; Waikoloa, Hawaii. https://www.healio.com/ophthalmology/cornea-external-disease/news/online/%7B18a90ab7-fae7-4e56-9566-c58958647d5e%7D/upcoming-pharmaceutical-treatments-for-presbyopia-may-prevent-delay-surgery. Accessed May 21, 2019
6. Vukich JA, Durrie DS, Pepose JS, et al. Evaluation of the small-aperture intracorneal inlay: three year results from the cohort of the US Food and Drug Administration clinical trial. J Cataract Refract Surg. 2018;44(5):541-556.
7. Ito M. Effectiveness and safety of combining LASIK and corneal inlay implantation: 4-year follow-up. Poster presented at: the American Academy of Ophthalmology Refractive Surgery Subspecialty Day; October 27-30, 2018; Chicago.
8. Trindade BLC, Trindade FC, Trindade CLC. Intraocular pinhole implantation for irregular astigmatism after planned and unplanned posterior capsule opening during cataract surgery. J Cataract Refract Surg. 2019;45(3):372-377.
9. Trindade BLC, Trindade FC, Trindade CLC, Santhiago MR. Phacoemulsification with intraocular pinhole implantation associated with Descemet membrane endothelial keratoplasty to treat failed full-thickness graft with dense cataract. J Cataract Refract Surg. 2018;44(10):1280-1283.
10. Trindade CC, Trindade BC, Trindade FC, et al. New pinhole sulcus implant for the correction of irregular corneal astigmatism. J Cataract Refract Surg. 2017;43(10):1297-1306.
11. Tsaousis KT, Werner L, Trindade CLC, et al. Assessment of a novel pinhole supplementary implant for sulcus fixation in pseudophakic cadaver eyes. Eye (Lond). 2018;32(3):637-645.
12. Dick HB, Piovella M, Vukich J, et al. Prospective multicenter trial of a small-aperture intraocular lens in cataract surgery. J Cataract Refract Surg. 2017;43:956-968.
13. Ang RE. Small-aperture intraocular lens tolerance to induced astigmatism. Clin Ophthalmol. 2018;12:1659-1664.
14. Agarwal S, Thornell EM. Cataract surgery with a small-aperture intraocular lens after previous corneal refractive surgery: visual outcomes and spectacle independence. J Cataract Refract Surg. 2018;44(9):1150-1154.
15. Barnett V, Barsam A, Than J, Srinivasan S. Small-aperture intraocular lens combined with secondary piggyback intraocular lens during cataract surgery after previous radial keratotomy. J Cataract Refract Surg. 2018;44(8):1042-1045.