




Periorbital wrinkles are lines or creases that form in the skin in the periorbital regions of the face. The formation of these wrinkles is a common and multifactorial phenomenon influenced by natural aging, photoaging (including UV light exposure), and other factors.
Treatment for improving the skin’s surface and thickness includes a group of procedures known as skin resurfacing, but these techniques have limitations, particularly for the treatment of the periorbital region where the skin is thin and delicate. Other treatment modalities such as lasers or intense pulsed light (IPL) emit radiation, which presents a hazard to eye safety.
Uses of Tixel
The Tixel fractional system (Novoxel) is a medical device designed to rejuvenate facial skin, including the delicate skin around the eyes, without emitting radiation. Published clinical study results show Tixel’s ability to rejuvenate facial skin1 and treat rosacea, active acne, hemangioma, hypertrophic scars, and more. Drug delivery using Tixel has also been extensively researched.2 The interim results from clinical studies investigating Tixel for the treatment of dry eye disease (DED) presented at the 2020 ASCRS Virtual Annual Meeting compare favorably with results reported for IPL. Patients treated with Tixel reported a decrease in DED symptoms, and investigators observed an objective improvement in tear film osmolarity and noninvasive tear breakup time after treatment (Table 1).3 Our clinical experience treating more than 100 patients with Tixel confirms these findings.
How It Works
Tixel employs a thermomechanical technology designed to provide clinical effects comparable to those of a fractional pulsed laser. The device is powered by the supply of contact-transferred heat from a high-temperature (385–400 °C) metal element called a tip, which consists of an array of miniature pyramids (Figures 1 and 2). The tip creates a superficial thermal effect in the tissue (usually 200–300 μm deep) within a few milliseconds of contact, generating a matrix of coagulation sites on the skin. The dermal heating and coagulative effects that occur during a Tixel treatment session provide dermal remodeling and collagen restructuring that promote wrinkle reduction and general wrinkle appearance improvement.

Figure 1. The periorbital tip of the Tixel device.

Figure 2. The facial tip of the Tixel device.
Courtesy of Novoxel
Tixel for DED
A protocol for treating DED with the Tixel system has been developed and clinically tested on 72 patients in a prospective study approved by an ethics committee and conducted by Drs. Hanneken and Shah. Tixel and IPL for the treatment of DED are compared in Table 1.
Tixel treatment is performed on the superior and inferior eyelids above the meibomian glands adjacent to the rim. Each treatment consists of 40 pulses on both eyes (10 pulses per eyelid in two parallel rows of five pulses each on the lid) covering a total skin area of 12 cm2 on both eyes. The applied energy is 0.012 J/cm2 (compared with about 12 J/cm2 with IPL).
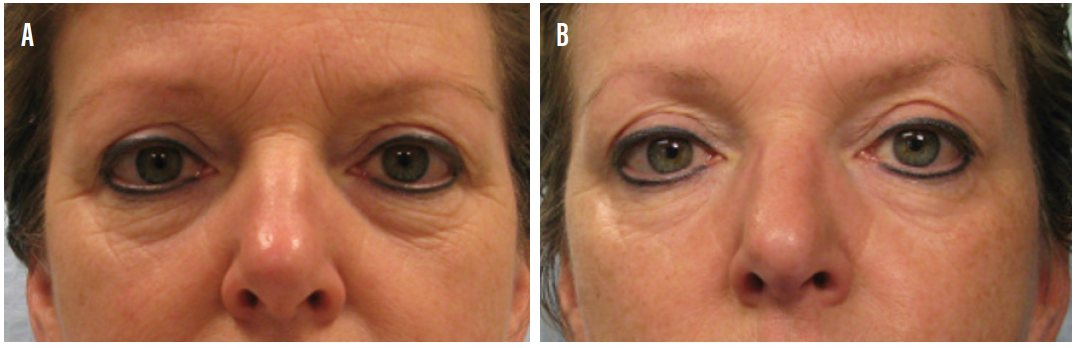
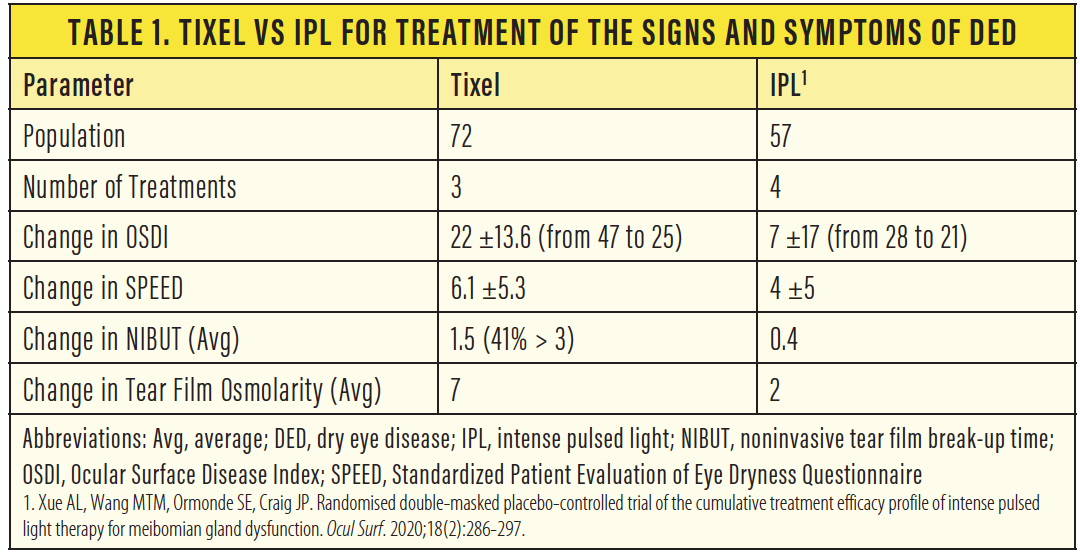
Figure 3. A patient before treatment (A) and the same patient after four treatment sessions in 4-week intervals (B).
Courtesy of Xavier Martens, MD
Treatment takes about 2 minutes for both eyes and consists of three treatments spaced 2 weeks apart. Eye protection is not required because the device does not emit radiation. Analgesic cream can be applied if the patient desires, but it is not necessary in most cases. No side effects have been reported other than mild erythema and edema, which disappeared within 2 days.
1. Elman M, Fournier N, Barnéon G, Bernstein EF, Lask G. Fractional treatment of aging skin with Tixel, a clinical and histological evaluation. J Cosmet Laser Ther. 2016;18(1):31-73.
2. Shavit R, Dierickx C. A new method for percutaneous drug delivery by thermo-mechanical fractional injury. Lasers Surg Med. 2020;52(1):61-69.
3. Shah S, Hanneken, L, Dutta D. Tixel, a completely novel way of treating dry eye. Paper presented at: The 2020 ASCRS Virtual Annual Meeting; May 16, 2020.
Iris Color Change
By Marguerite B. McDonald, MD, FACS

Stro–ma Medical has perfected a way to change eye color safely and permanently using the Stro–ma Laser System (Stro–ma Medical; Figure 4). It may sound to some like a frivolous thing to do, but people all over the world have spent countless millions of dollars on colored contact lenses and even artificial iris implants, which are well known to be highly risky.

Figure 4. A patient’s eyes before receiving treatment with the Strōma Laser System (A). The same patient after receiving five treatments to the left eye and one to the right eye (B).
Courtesy of Stro-ma Medical
How it Works
The Stro–ma Laser System can make dark eyes lighter, transforming them to various shades of blue, green, and gray. The laser uses a frequency-doubled Nd:YAG laser (532-nm wavelength) to raise and lower the temperature of the anterior iris pigment 15 times over 30 seconds, which stimulates pigment on the anterior surface of the iris and triggers a cytokine and macrophage response. The macrophage response takes up to 3 weeks to carry away the pigment via the vascular system, exposing the stromal fiber.
The iris color change is done in stages. When the patient walks out after the first treatment, his or her iris looks the same. It takes up to 3 weeks for the color change to become apparent.
Safety and Efficacy
In a pilot study of this technique and technology, a one-eighth segment of one eye of each patient’s iris was treated with the Stro–ma Laser System. This study was completed in September 2019.
The investigators looked at all clinical outcome measures including dry eye, visual acuity, color, contrast sensitivity, retinal health, IOP, OCT images, the crystalline lens, corneal health, endothelial cell counts. No adverse events were reported, and they were able to establish the parameters needed to move forward (unpublished data).
The laser system has a high-speed tracking feature that ensures that each shot is delivered with great accuracy. There have been no reports of the laser missing the targeted area of treatment.
US trials of the laser have not yet started, but full iris trials outside the United States have shown promising safety and efficacy data. Thirty patients were assigned to three groups, 10 patients in each group (unpublished data). Researchers also administered a variety of psychometric tests to patients postoperatively, asking questions such as, “Do you think your eyes are more attractive?” No patient has reported thinking that his or her eyes are less attractive postoperatively.
Intense Pulsed Light
By Laura M. Periman, MD

IPL is not a laser. It is a powerful broad beam light, well above the UV spectrum. This mode of cosmetic skin treatment has been around for more than 20 years and has a long list of US FDA-approved indications, including rosacea and benign vascular lesions. Although many manufacturers outside of ophthalmology produce IPL machines, the M22 Optima IPL (Lumenis) has built-in treatment algorithms and safety features that make it easy for ophthalmologists to incorporate into their clinical practices.
When clinicians begin to look closely at dry eye disease, they will notice the high prevalence of ocular and facial rosacea in patients with dry eye. The M22 Optima IPL is my go-to treatment modality for patients with rosacea who have Fitzpatrick skin types I-IV. In my experience, IPL results in excellent improvement of a variety of dry eye disease symptoms, including clinical findings with fluorescein staining of the ocular surface, tear breakup time, meibomian gland expressibility, osmolarity readings (TearLab Osmolarity System), and inflammation (InflammaDry, Quidel). IPL has also been found effective in the treatment of acute and chronic chalazia (unpublished data)1-3 as well as in the control of Demodex.4
But the part that patients really love is that they look better, too. IPL is the only thing in our advanced dry eye toolkit that also gives patients an aesthetic benefit. This contributes to the very high patient satisfaction ratings (9.2 out of 10) with this treatment modality among our patients.
If you limit IPL to just that, however, you’ll miss out on other aesthetic uses of IPL. In my practice, for example, we also use it for treating cherry angiomas around the eyelids without surgery, for which it is incredibly effective (Figure 4), improving the appearance of nasal angle telangiectasis (common in rosacea), and, occasionally, permanent removal of unwanted facial hair on the upper lip or chin.
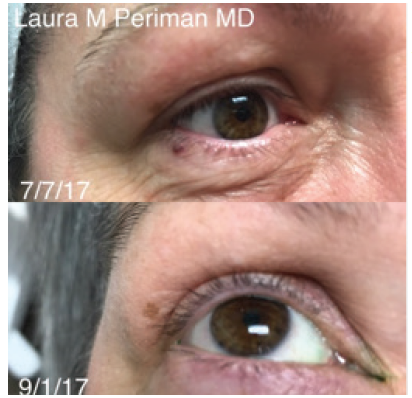
Figure 5. A 55-year-old patient with rosacea-associated ocular surface disease and incidental angioma (top). The angioma was effectively treated (bottom) in step 4 (aesthetic clean up) of the Periman Protocol.
Courtesy of Laura M. Periman, MD
How it Works
In addition to its photocoagulation, photoimmunomodulation, photosanitization, and photobiomodulation properties, IPL also has photorejuvenation properties that can be leveraged across a variety of areas in ophthalmic practice.5
In Practice
In my advanced dry eye practice, when I speak with a patient who has ocular or facial rosacea, I explain, “IPL is an effective technology for controlling this aspect of your dry eye problem.” I also share some clinical photos and explain to the patient how IPL works to normalize tear osmolarity and decrease inflammation (manuscript in process).
I like to joke with patients that IPL has a side effect that they need to know about: It will make your skin look better. We don’t lead with that in my practice because aesthetics is not our main focus. We just mention it as a happy side effect. Many patients have told us that with the complexion-improving effects of IPL, they need much less of the dry eye–exacerbating cosmetics, foundations, and powders than they did prior to treatment.
If you’re going to use IPL mainly for its dry eye benefits, it’s important to use the The M22 Optima IPL, not only for its excellent safety features and efficacious performance but also because most of the peer-reviewed literature on IPL is based on this specific platform.
Additionally, I have found that taking the time to treat the eyelids with laser-grade corneal shields in place enhances the ocular rosacea and meibomian gland dysfunction treatment results.
Ideal Patient
There are two main patient pools for IPL, and they often overlap. Most ophthalmic practices treat patients with dry eye. If a dry eye patient presents with telangiectasis of the lid margins, significant meibomian gland dysfunction, or with evidence of ocular rosacea or facial rosacea, you should begin a discussion about IPL and the dry eye benefits associated with it. On the other hand, if you want to focus on the aesthetic aspects of IPL, the ideal patient is someone who has a skin issue such as rosacea or sun damage and is self-conscious about it. It depends on what’s important to the patient.
A Workhorse
I have found IPL to be the workhorse of my advanced dry eye practice. Patients are thrilled with the improvements in their dry eyes and their appearance. We have made some instructional videos on YouTube and will be adding more content to DryEyeMaster.com. I encourage anyone interested in this technology to explore it further.
1. Paper presented at the: 37th Congress of the ESCRS; September 14-18, 2019; Paris.
2. Paper presented at the: ASCRS/ASOA 2019; May 3-7, 2019; San Diego.
3. Paper presented at: Women in Ophthalmology 2019 Summer Symposium; August 22-25, 2019; Coeur d’Alene, Idaho.
4. Fishman HA, Periman LM, Shah AA.Real-time video microscopy of in vitro Demodex death by intense pulsed light. Photobiomodul Photomed Laser Surg. doi: 10.1089/photob.2019.4737. [published online ahead of print January 27, 2020]
5. Dell SJ. Intense pulsed light for evaporative dry eye disease. Clin Ophthalmol. 2017;11:1167-1173.
Injectable Dermal Fillers
Anaïs L. Carniciu, MD

Dermal fillers are injectable agents that treat the volume loss and associated signs of facial aging that occur with maturity. They can also be used to volumize skin irregularities such as depressed scars.
FILLER MATERIALS
Currently, dermal fillers are made from four main materials. The most commonly used material is hyaluronic acid (HA). Other available materials include calcium hydroxylapatite (CaHA), poly-L-lactic acid (PLLA), and polymethylmethacrylate (PMMA) with bovine collagen. See Table 2 for detailed information about each of these materials.
Of note, PMMA-based dermal filler carries the risk of an allergic reaction to the bovine collagen. With proper preprocedure allergy skin testing, this material may be considered only for patients seeking very long-term volume augmentation.
FILLER TRAINING
For ophthalmologists interested in additional aesthetics training after residency, it’s best to make use of resources from the AAO. The AAO offers programs at its annual meeting that can help surgeons learn the nuances of filler techniques and materials through hands-on training. Ophthalmologists with an especially strong interest in facial aesthetics can pursue additional subspecialty training in oculofacial plastic surgery as part of an American Society of Ophthalmic Plastic and Reconstructive Surgery fellowship, which is the route I took.
It’s advisable to become an expert with HA filler injections prior to incorporating any of the other materials in your clinical practice. HA is the safest of the filler materials available because it can be reversed with the injection of hyaluronidase. Without hyaluronidase injection, HA fillers dissolve on their own over 1 to 2 years.
Prior to adding filler injections to your clinical practice, I highly recommend that you have expertise with the injection of a cosmetic neurotoxin such as Botox (onabotulinumtoxinA, Allergan). Because cosmetic neurotoxin injection carries a much lower risk profile than dermal filler injection, the former is a safer clinical approach for developing injector expertise.
COMPLICATIONS AND PRECAUTIONS
A thorough, expert-level understanding of oculofacial anatomy is necessary for any aesthetic provider offering dermal fillers. Potential side effects of filler injections include under- and overtreatment, bruising, edema, allergic reactions, infection, and granuloma formation. Because injectable dermal fillers are also associated with rare but serious complications, including skin necrosis, blindness, and stroke, surgeons should always have a filler crash cart. These are materials that should always be available in the office, fully stocked and readily accessible, just as you’d have an endotracheal tube available in an intensive care unit. The crash cart should contain aspirin, nitroglycerin paste, and hyaluronidase to reverse an HA filler, if needed.
CONCLUSION
Dermal fillers are excellent nonsurgical treatments to offer your patients. Patient satisfaction is typically quite high, especially among patients with mild early facial aging changes who aren’t yet ready to go to an oculoplastic surgeon for a more invasive surgery in the OR.


