
The list of IOLs that are currently available seems endless, and each comes with risks and benefits. The responsibility falls to cataract surgeons to discuss IOL options with patients and recommend a lens with a reasonable postoperative target vision.
IOL selection is always an important step, and it depends on the patient’s ocular condition and anatomy, past history, and personal needs.1 For patients who have or who are at risk of developing a retinal disease or retinal detachment, certain types of IOLs should be avoided or used with caution.
MATERIAL
Polymethylmethacrylate has largely been replaced by foldable materials such as silicone and acrylic because they allow an IOL to be inserted through a small incision. The choice of material is a major consideration when patients have a retinal condition or an increased likelihood of requiring retinal surgery.
Silicone IOLs can make vitreoretinal surgery difficult by limiting the visibility of the retina. These lenses can become fogged during fluid-air exchange, a common procedure in retinal detachment repair. Silicone oil may adhere to the lens and decrease the patient’s quality of vision. Removing the droplets is technically difficult because of the strong hydrophobic interaction between the oil and the lens. Droplets can remain attached to the posterior surface of a silicone IOL even after their removal is attempted.2,3 These complications are more common with silicone and polymethylmethacrylate IOLs than with other materials. Hydrophobic acrylic is the most compatible with silicone oil.
Hydrophilic acrylic IOLs carry a risk of opacification that may necessitate lens exchange. Filling the anterior chamber or vitreous cavity with a gas bubble during a retinal procedure may promote a calcification process, leading to opacification of the IOL. Hydrophilic acrylic IOLs can also develop localized calcification on the anterior surface and subsurface of the optic, generally limited to the area of the capsulorhexis opening.4
Material type is also a risk factor for the development of posterior capsular opacification (PCO), as are IOL design, optic diameter, and edge configuration. The occurrence of PCO, a condition that impedes the view of the retina, varies among IOLs. It can be treated easily with an Nd:YAG laser or intraoperative capsulectomy, but both procedures may damage or dislocate the IOL and can cause cystoid macular edema and retinal detachment. An open posterior capsule may also change the approach a retina surgeon takes to planning the surgery. PCO is more common with silicone (Figure) than hydrophobic acrylic IOLs.
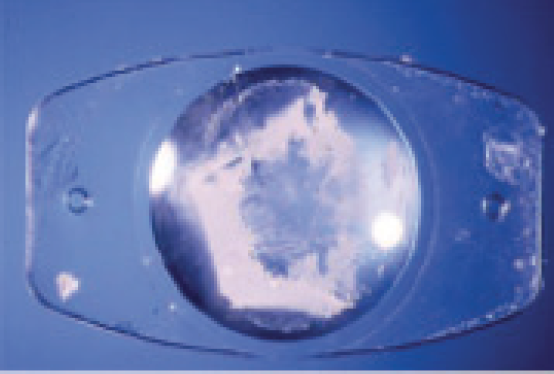
Figure. Explanted plate-haptic silicone lens exhibiting calcification of the posterior optic surface.
Courtesy of Liliana Werner, MD, PhD
Another consideration is whether or not to use an IOL with blue-blocking material in retina patients. All IOLs decrease the amount of UV light that passes into the eye. The rationale for blue‐light–blocking IOLs is to prevent the development or progression of age‐related macular degeneration (AMD), but it remains unclear whether these IOLs preserve macular health. A significant proportion of patients with AMD are phakic adults over 60 years of age.5
DESIGN
Following is some guidance on the use of certain IOL designs in retina patients.
Multifocal IOLs. Lenses with a multifocal design should be used with caution in patients with AMD or other diseases requiring vitreoretinal surgery as well as in patients who are at risk of developing these diseases. These patients are more often dissatisfied with their postoperative quality of vision compared to routine patients because of a reduction in contrast sensitivity, especially under mesopic conditions and with increased metamorphopsia in cases of macular edema.
It is worth noting that few studies have found that patients with cataracts and retinal diseases such as macular degeneration and diabetic retinopathy benefit from the multifocality of an IOL or report a satisfactory improvement in vision.6,7
Further, pars plana vitrectomy through a multifocal IOL presents several challenges. The concentric optical zones of these IOLs can interfere with the surgeon’s view and make surgical instruments appear to jump as they cross the rings. They may also make it more difficult for the surgeon to see fine details on the retinal surface during epiretinal membrane peeling.8,9 Moreover, subtle displacement of an IOL may occur during a fluid-air exchange, which—particularly with a multifocal lens—can decrease quality of vision for the patient. Retinal surgery may also induce postoperative refractive changes such as myopia and corneal astigmatism.10
Phakic IOLs. An iris-fixated phakic IOL may limit pupillary dilation and thereby hinder a fundus examination. The decrease in pupillary diameter may be transient with a posterior chamber phakic IOL.11,12
Accommodating IOLs. Accommodating lenses with a dual-optic design, small optic, silicone material, or flexible plate-haptic configuration may cause problems with stability and visualization in retina patients.
Wavefront technology and aspheric IOLs. Recently, manufacturers have used wavefront technology to improve the visual quality delivered by IOLs. Aspheric IOLs offer the advantages of neutralizing corneal spherical aberration and allowing a better view of the peripheral retina without distortion. Further clinical study of these technologies is warranted.
STRUCTURE
Smaller optic diameters may limit the surgeon’s view of the vitreous and the retina and interfere with laser treatment. A posterior square edge, forced against the capsule, creates a barrier to lens epithelial cell migration behind the optic and prevents PCO.
The rate of dislocation, both spontaneous and after laser capsulotomy, is high with plate-haptic IOLs.13-15 Significant differences between one- and three-piece IOLs have not been found with regard to tilt and decentration after implantation in an intact capsular bag.16
CONCLUSION
When choosing an IOL for a patient who has retinal disease or who is at risk of requiring retinal surgery, cataract surgeons must select lenses that will maximize visual potential. It is just as important for these patients to understand the goal of cataract surgery as it is for the surgeon to carefully manage their expectations.
With that in mind, the following preoperative steps are key:
- Explain to patients that postoperative visual improvement will be suboptimal and inform them of the potential of disease progression after surgery;
- Discuss visual expectations, particularly if a premium IOL is selected; and
- Carefully examine the macula and peripheral retina in addition to the anterior segment.
To date, there are no published guidelines on IOL selection for retina patients. The choice of IOL for patients who have retinal conditions is based on the lens’ compatibility with silicone oil, opacification rate, optic diameter, and stability and on the view of the retina that it provides.
1. Örnek K. Cataract surgery in retina patients. IntechOpen. February 7, 2013. Accessed November 2, 2020. https://www.intechopen.com/books/cataract-surgery/cataract-surgery-in-retina-patients
2. Stappler T, Williams R, Wong D. F4H5: a novel substance for the removal of silicone oil from intraocular lenses. Br J Ophthalmol. 2010;94:364-367.
3. Charles S. Vitrectomy techniques for complex retinal detachments. Taiwan J Ophthalmol. 2012;2:81-84.
4. Park DI, Ha SW, Park SB, Lew H. Hydrophilic acrylic intraocular lens optic opacification in a diabetic patient. Jpn J Ophthalmol. 2011;55:595-599.
5. Mainster MA, Turner PL. Blue-blocking IOLs decrease photoreception without providing significant photoprotection. Surv Ophthalmol. 2010;55:272-289.
6. Kamath GG, Prasad S, Danson A, Phillips RP. Visual outcome with the Array multifocal intraocular lens in patients with concurrent eye disease. J Cataract Refract Surg. 2000;26:576-581.
7. Gayton JL, Mackool RJ, Ernest PH, Seabolt RA, Dumont S. Implantation of multifocal intraocular lenses using a magnification strategy in cataractous eyes with age-related macular degeneration. J Cataract Refract Surg. 2012;38:415-418.
8. Ahmad BU, Shah GK, Hardten DR. Presbyopia-correcting intraocular lenses and corneal refractive procedures: a review for retinal surgeons. Retina. 2014;34:1046-1054.
9. Hadayer A, Jusufbegovic D, Schaal S. Retinal detachment repair through multifocal intraocular lens – overcoming visualization challenge of the peripheral retina. Int J Ophthalmol. 2017;10:1008-1010.
10. Randleman JB, Hewitt SM, Stulting RD. Refractive changes after posterior segment surgery. Ophthalmol Clin North Am. 2004;17:521-526.
11. Lemarinel B, Racine L, Rohart C, Hoang-Xuan T, Gatinel D. Long-term changes in pupil size after implantation of an Artisan phakic intraocular lens for correction of high myopia. J Fr Ophtalmol. 2007;30:11-16.
12. Zhu Y, Yue Y, Zhu H, Chen J, Zhou J. Influence of refractive error on pupil diameters in highly myopic eyes with implantable collamer lenses. Eur J Ophthalmol. 2019; 1120672119863716.
13. Apple DJ, Solomon KD, Tetz MR, et al. Posterior capsule opacification. Surv Ophthalmol. 1992;37:73-116.
14. Mackool RJ. Decentration of plate-haptic lenses. J Cataract Refract Surg. 1996;22:396.
15. Akerele T, Minasian M, Little B, Jagger J. Posterior dislocation of STAAR plate haptic silicone lenses following Nd:YAG capsulotomy. Eye. 1999;13:700-702.
16. Mutlu FM, Erdurman C, Sobaci G, Bayraktar MZ. Comparison of tilt and decentration of 1-piece and 3-piece hydrophobic acrylic intraocular lenses. J Cataract Refract Surg. 2005;31:343-347.


