
Patients with cataracts frequently have coexisting ocular conditions such as glaucoma. This condition can develop spontaneously in eyes with pseudoexfoliation or with uveitis or occur after ocular trauma, ocular surgery, or angle closure. Whatever the underlying cause, glaucoma is a progressive disease that necessitates lifelong observation of the patient.
Cataract surgery generally reduces IOP by 1 to 2 mm Hg in patients with glaucoma,1,2 and the procedure can improve the interpretability of perimetry. The timing of the surgery, however, must be based not only on the cataract grade and the patient’s visual acuity but also on glaucoma severity, the number of glaucoma medications the patient is administering, the target IOP, and whether the IOP is well controlled. After cataract surgery, it is important for functional and structural baselines and the target IOP to be reestablished.
Cataract surgeons may wish to obtain a glaucoma specialist’s opinion during surgical planning and postoperative follow-up, and it may be necessary for cataract and glaucoma surgeons to work together in some clinical situations. This article describes four clinical situations in which a referral to a glaucoma specialist may be warranted.
1. CATARACT AND MILD TO MODERATE GLAUCOMA
Thanks to the expanding options for MIGS and growing interest in interventional glaucoma (see What Is Interventional Glaucoma?), glaucoma surgery may be recommended early in the disease course to prevent progression and decrease the medication burden. Current MIGS procedures increase aqueous outflow via subconjunctival, trabecular meshwork, or suprachoroidal pathways, and these procedures can be combined with cataract surgery.3
What Is Interventional Glaucoma?
By Iqbal Ike K. Ahmed, MD, FRCSC
Philosopher Thomas Kuhn maintained that scientific research and thought are defined by paradigms. According to Kuhn, scientists typically accept an existing paradigm and then try to extend its scope over time. Eventually, however, their efforts may expose the paradigm’s inadequacies or contradict it entirely. An accumulation of these difficulties triggers a crisis, which, in turn, sparks an intellectual revolution or a paradigm shift. This is a model of change that we may be experiencing in glaucoma.
From issues with treatment noncompliance to the complex, chronic nature of the disease itself, glaucoma has long presented challenges to those who strive to manage it and those who live with it. These challenges have, in turn, served as an impetus for innovation and have contributed to the formation of a new school of thought: interventional glaucoma (IG). The IG paradigm is founded on the objective to approach glaucoma in a proactive—not reactive—way and to intervene in a disease-centric and patient-centric manner. The IG specialist considers not only the disease’s impact on a patient’s quality of life but the treatment’s impact as well. Although MIGS was developed with this in mind, IG does not refer solely to one type of intervention.
Those who can view glaucoma from the patient’s perspective and factor quality of life into their treatment decision-making will be able to prioritize an IG mindset. As innovation continues and this patient-centric outlook grows, we can all approach glaucoma in a way that addresses safety, efficacy, and quality of life. Although the path is long and there is much to accomplish, the desire to truly improve the lives of our patients with glaucoma is the driving force we need to push this movement forward.
MIGS procedures that target the trabecular meshwork are indicated for the treatment of mild to moderate glaucoma in conjunction with cataract surgery. Familiarity with gonioscopic anatomy and comfort working with a surgical goniolens are necessary. Cataract surgeons who lack experience with these relatively safe procedures may wish to refer patients to a glaucoma specialist. It can also be beneficial for anterior segment and glaucoma surgeons to discuss the patient’s target IOP after cataract surgery.
2. CATARACT AND ADVANCED GLAUCOMA
Cataracts can make the assessment of advanced structural and functional glaucomatous damage difficult. Cataracts can also decrease quality of vision and quality of life in patients who have lost peripheral vision. As mentioned earlier, cataract surgery alone provides only a modest reduction in IOP. A combined procedure is therefore often a better choice for patients with cataracts and advanced glaucoma, particularly if IOP is uncontrolled (Figure 1).
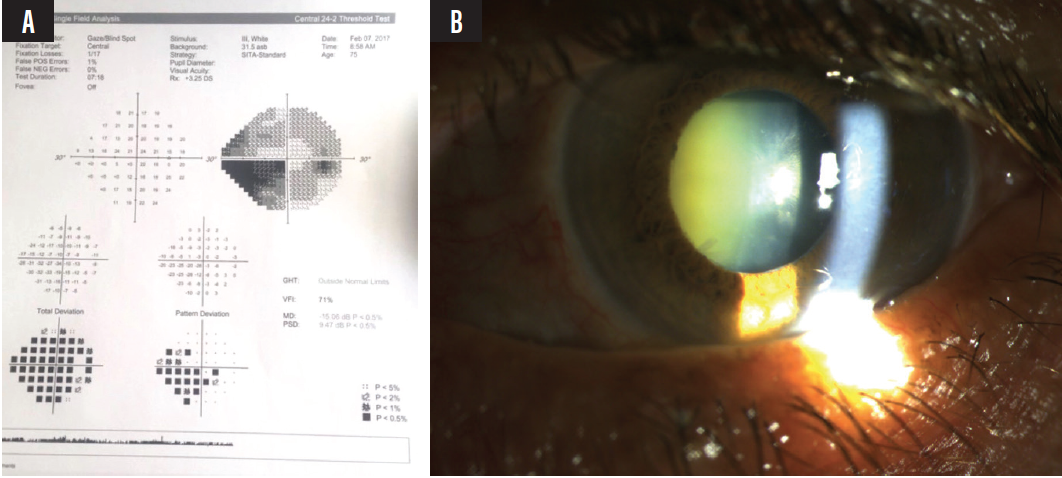
Figure 1. Visual field of a patient with cataracts (A), pseudoexfoliation glaucoma, and advanced visual field defects (B). Note the difference between the total and pattern deviation maps (A) supporting the presence of cataracts.
Combining cataract surgery and MIGS offers the advantage of relative safety. Because many of these patients need a low target IOP, however, a more invasive glaucoma procedure such as trabeculectomy or the implantation of a glaucoma drainage device may be required. The choice of surgical procedures and their timing can be discussed with a glaucoma specialist.
Alternatively, a patient can be referred if filtering surgery is planned or if cataract surgery will be complex and the patient has secondary glaucoma such as neovascular glaucoma and poor vision potential (Figure 2). Another reason to refer patients with advanced glaucoma is that they are at increased risk of an IOP spike after cataract surgery.
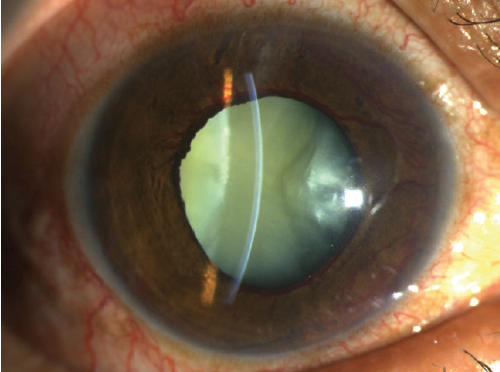
Figure 2. Cataract in a patient with neovascular glaucoma.
3. EARLY POSTOPERATIVE IOP SPIKE
The early detection of IOP peaks after cataract surgery in patients with glaucoma may be the most important part of postoperative management. Patients with advanced disease require close monitoring of their IOP and visual fields after the cataract procedure because of the risk of central vision loss secondary to IOP spikes.
Because patients sometimes decide to cease administering glaucoma medications after cataract surgery, it is important to counsel them that they should continue their preoperative drug regimen to maintain IOP control during the postoperative period. Another cause of increased IOP after cataract surgery is a response to topical steroids.4 In patients with advanced glaucoma, it may be prudent to prescribe an NSAID in lieu of a steroid after cataract surgery to prevent an increase in IOP. Anterior segment surgeons may wish to refer patients with advanced glaucoma to a glaucoma specialist after cataract surgery to facilitate the detection of an IOP spike and disease progression.
4. PREVIOUS FILTERING SURGERY
Patients who have undergone filtering surgery often benefit from a referral to a glaucoma specialist, both for assistance with planning cataract surgery and for postoperative care, because these patients are at increased risk of a failed filter after the cataract procedure.5 Cortical cleanup must be meticulous to minimize postoperative inflammation, which can cause the bleb to fail. Moreover, postoperative inflammation may require more frequent or extended dosing of topical steroids, which increases the risk of an IOP spike and glaucomatous progression.
An overhanging or avascular bleb can make cataract surgery more technically difficult and increases the risk of postoperative endophthalmitis. A glaucoma specialist can perform bleb revision or excision before the cataract procedure (Figure 3) and provide postoperative monitoring of these patients for increases in IOP.
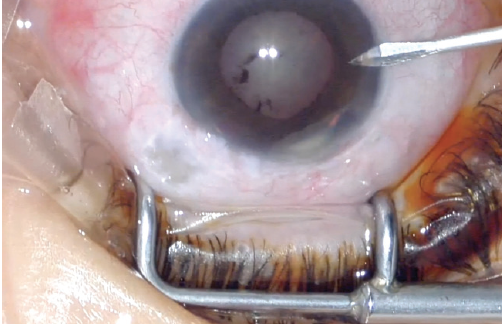
Figure 3. A patient with advanced glaucoma, an avascular bleb, and a history of recurrent bleb leaks. The bleb was excised, and tube shunt surgery was planned to address the risk of an increase in IOP after cataract surgery.
CONCLUSION
Cataract surgeons with limited experience performing MIGS procedures should look to partner with—and refer to when necessary—a glaucoma specialist who is well-versed in this type of surgery. In many clinical situations, the postoperative outcomes are most favorable when cataract and glaucoma surgeons work together to provide care.
1. Shingleton BJ, Pasternack JJ, Hung JW, O’Donoghue MW. Three and five year changes in intraocular pressures after clear corneal phacoemulsification in open angle glaucoma patients, glaucoma suspects, and normal patients. J Glaucoma. 2006;15(6):494-498.
2. Rao A. Diurnal curve after phacoemulsification in patients with pseudoexfoliation syndrome and cataract. Semin Ophthalmol. 2012;27(1-2):1-5.
3. Rabin RL, Rabin AR, Zhang AD, Burney EN, Rhee DJ. Co-management of cataract and glaucoma in the era of minimally invasive glaucoma surgery. Curr Opin Ophthalmol. 2018;29(1):88-95.
4. Chang DF, Tan JJ, Tripodis Y. Risk factors for steroid response among cataract patients. J Cataract Refract Surg. 2011;37(4):675-681.
5. Husain R, Liang S, Foster PJ, et al. Cataract surgery after trabeculectomy: the effect on trabeculectomy function. Arch Ophthalmol. 2012;130(2):165-170.



