Artis Symbiose
This complementary lens system provides mix-and-match IOLs by design.
I have, over the past 6 months, had the privilege of using a new trifocal lens system, the Artis Symbiose (Cristalens). The system consists of a complementary pair of lenses. One lens, the Plus, is designed for increased near vision and implanted in the nondominant eye. The other lens, the Mid, is designed to provide relatively more intermediate vision and implanted in the dominant eye. The lens system is different from other trifocal offerings in providing depth of focus by preserving continuous phase, with none of the phase inversion or loss of focus seen as troughs in the defocus curves of other diffractive lenses (Figure 1).
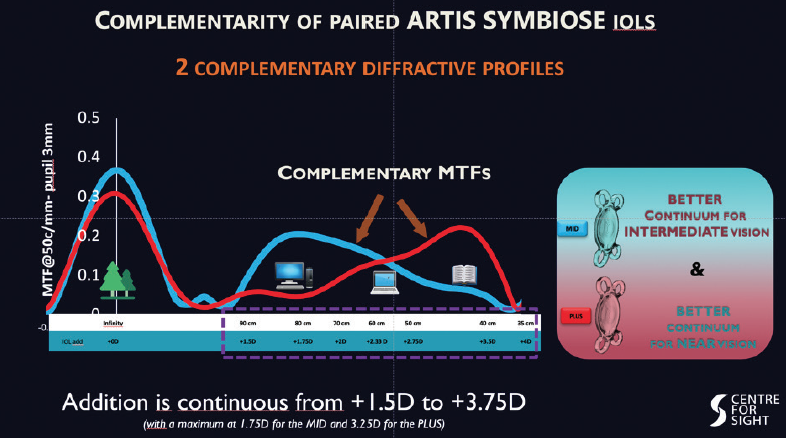
Figure 1. The Artis Symbiose Plus and Mid lens models have complementary diffractive profiles, providing a continuum of good vision.
Courtesy of Sheraz M. Daya, MD, FACP, FACS, FRCS(Ed), FRCOphth
At last data audit, we had performed 105 implantations of the Artis Symbiose, of which 50 were bilateral and five were unilateral Plus lenses in patients who were undergoing monocular cataract surgery or who had undergone surgery elsewhere in the other eye with a less-than-satisfactory result. At 6 weeks postoperative, 84% were within ±0.50 D of emmetropia, and 100% were within ±1.00 D. Binocular uncorrected distance visual acuity (UDVA) was 20/20 or better in 83% of eyes, uncorrected near visual acuity (UNVA) was 20/25 or better in 86% of eyes, and uncorrected intermediate visual acuity (UIVA) at 60 and 80 cm was 20/25 or better and 20/20 or better, respectively, both in 97% of eyes.
Patients were overall very satisfied, and only three spontaneously reported a difference in near vision between the two eyes. All patients had good intermediate vision right away with an overall wow response reminiscent of LASIK. Although halos were reported, no patient complained about this being debilitating or interfering with the ability to drive.
The defocus curves were interesting, in that there were no troughs with good vision at all distances from both Mid and Plus lenses with a binocular cumulative effect (Figure 2).
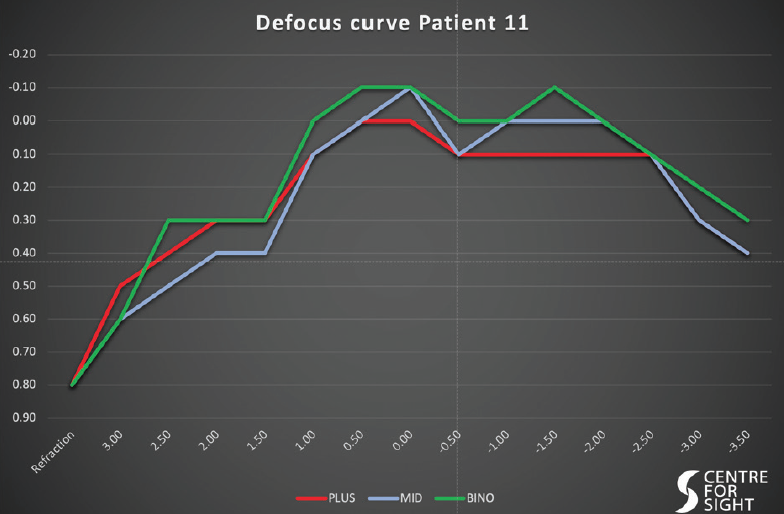
Figure 2. The defocus curves—in each eye and binocularly—of an individual implanted with the Artis Symbiose.
Courtesy of Sheraz M. Daya, MD, FACP, FACS, FRCS(Ed), FRCOphth
The lens system is a welcome addition to the armamentarium of trifocal lenses. Binocularly, the Artis Symbiose system provides good vision at all distances with what appears to be a reduced time for adaptation.
Tecnis Eyhance
Broader defocus curve enhances intermediate vision with this monofocal IOL.
I began using the Tecnis Eyhance (model ICB00, Johnson & Johnson Vision) monofocal lens with enhanced intermediate vision about 1 year ago. For the many patients under public health systems who cannot afford or do not have access to presbyopia-correcting IOLs, this lens makes it possible for me to turn ordinary cataract management into an extraordinary experience.
For patients, the Tecnis Eyhance adds what I like to think of as the lost dimension—intermediate vision. Everyday activities such as working on a laptop or tablet, playing cards, and cooking all take place primarily at intermediate focal lengths. But ophthalmologists don’t have appropriate ways of measuring intermediate vision. I am part of an ESCRS working group that recently published an editorial advocating better measures of functional vision, including intermediate function, as indicators for cataract surgery.1 I love that this lens can enhance intermediate vision without added dysphotopsia or any loss of distance visual acuity or clarity.
The Tecnis Eyhance offers a slightly broader defocus curve than a standard monofocal IOL that allows a wider sweet spot with enhanced intermediate vision. It does not rely on diffractive optics, rings, or zones. Rather, the broader defocus is achieved through an aspheric surface with continuous increase in power from the periphery to the center of the lens.
Use of this lens is an easy transition for any surgeon who is familiar with the Tecnis ZCB00 monofocal IOL because it has the same base geometry, material characteristics, edge design, haptics, and A-constant. There is no learning curve or additional chair time. Surgeons can be confident in the stable, well-established Tecnis platform that does a good job preventing posterior capsular opacification, balancing out corneal spherical aberration, and retaining its clarity after implantation.
CLINICAL EXPERIENCE
In a recently published prospective case series, my colleagues and I compared visual acuity at near (40 cm), intermediate (66 cm), and distance (4 m) at 6 months postoperative in patients bilaterally implanted with either the ZCB00 or Tecnis Eyhance.2 We also looked at quality-of-vision metrics.
Monocular and binocular UIVA was significantly better in the Eyhance group than the Tecnis group, while distance and near visual acuities were similar between the two groups (Figure 3). There were no significant differences in photopic contrast sensitivity, light scatter, modulation transfer function cutoff, Strehl ratio, or glare and halo perception between the groups.
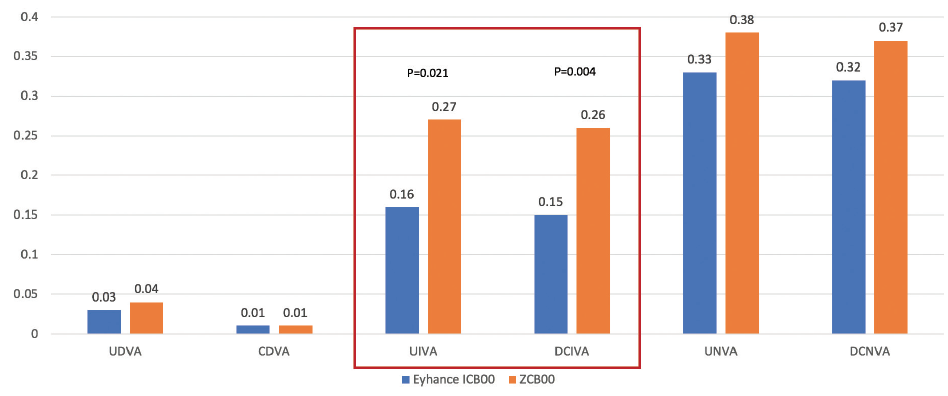
Figure 3. The Tecnis Eyhance monofocal IOL offered statistically significantly better UIVA and distance-corrected binocular intermediate visual acuity (DCIVA) compared to the Tecnis 1-piece ZCB00 monofocal. There were no significant differences in UDVA or corrected distance visual acuity (CDVA) or in UNVA or distance-corrected near visual acuity (DCNVA).
Courtesy of Johnson & Johnson Vision
NOT A MULTIFOCAL
The Eyhance IOL is a good option for risk-averse patients or those with pathology who might not be suitable candidates for a presbyopia-correcting IOL but would still like to maximize their functional vision. It is important to be clear with patients that the Eyhance is a monofocal, not a multifocal, lens.
This lens would not be the best choice for patients with a high demand for near vision who desire spectacle independence. In those patients, I direct them toward a multifocal, trifocal, or extended depth of focus (EDOF) IOL. It is also not the best choice for patients with significant astigmatism, as a toric version is not yet available. For all other monofocal candidates, the Tecnis Eyhance is now my preferred lens.
1. Ribeiro F, Cochener C, Kohnen T, et al; ESCRS Functional Vision Working Group. Definition and clinical relevance of the concept of functional vision in cataract surgery: ESCRS position statement on intermediate vision. J Cataract Refract Surg. 2020;46:S1-S3.
2. Mencucci R, Cennamo M, Venturi D, et al. Visual outcome, optical quality and patients’ satisfaction with a new monofocal intraocular lens, enhanced for intermediate vision: Preliminary results [published online ahead of print January 27, 2020]. J Cataract Refract Surg.
FineVision Triumf
Improved vision and contrast sensitivity are associated with the lens’ chromatic aberration-free optical design.
The FineVision Triumf (PhysIOL; Figure 4), is a trifocal IOL with EDOF optical technology. I was one of the first two surgeons worldwide to implant the Triumf IOL, and I am now participating in a prospective multicenter study of the lens.

Figure 4. The FineVision Triumf IOL.
Figures 4 and 5 courtesy of PhysIOL
The Triumf is made of PhysIOL’s proprietary glistening-free hydrophobic acrylic lens material. The overall diameter and the optic diameter of the Triumf are 11.4 and 6 mm, respectively, and the lens is UV- and blue light–filtering. This article reviews some of the early results and 6-month follow-up data on 78 eyes of 40 patients implanted with the lens.
The mean age of the patients was 65.6 years. Of the 40 patients, 18 were Asian and 22 were white. This is important because Asian eyes tend to be more myopic, and this study is providing valuable information on how the IOL behaves in both populations. We used a target refraction of plano to -0.25 D for two reasons.
First, we wanted the sphere to be consistently on target to calculate a universal A-constant. Second, we have found that a slight myopic spherical equivalent helps patients to achieve better near vision.
RESULTS
The mean monocular UDVA in these 78 eyes was 20/22, with 75% of patients achieving 20/20 or better and 79% achieving 20/25 or better. Mean monocular CDVA was 20/20, with 11% of eyes achieving 20/16 or better and 96% achieving 20/20 or better.
It is unusual to obtain results this good on a multifocal IOL platform. We attribute these results to the chromatic aberration characteristics of the Triumf, which I will discuss further later in this article.
Mean monocular UIVA at 6 months postoperative was 20/23, with 46% of patients achieving 20/20 or better and 89% achieving 20/25 or better. When patients were corrected for distance, the mean UIVA was approximately 20/22, with 64% of patients achieving 20/20 or better and 89% achieving 20/25 or better.
The mean monocular UNVA with the Triumf was 20/25. Further, 46% of patients achieved 20/20 or better and 68% achieved 20/25 or better. This is an excellent result, showing that near vision is not sacrificed with the Triumf, even as distance and intermediate acuities remain strong. When the influence of the slight myopia was corrected, about 75% of patients still achieved 20/25 DCNVA.
GOOD VISION AT ALL DISTANCES
From these results, we determined that mean visual acuity through all distances can be expected to be about 20/25.
The defocus curves, based on visual acuity outcomes from distance, intermediate, and near visual acuity tests, showed that the lens provides patients with a broad range of vision (monocular: -3.20 to +1.30 D [31 cm to infinity]; binocular: -3.80 to +1.50 D [26 cm to infinity]).
Patients enrolled in the study to date have been highly satisfied. All patients reported spectacle independence at distance and intermediate, and 95.7% reported spectacle independence at near. Only 4.3% of patients reported needing glasses for near vision tasks some of the time, and none reported needing them all or most of the time.
CONCLUSION
We attribute the improved vision and contrast sensitivity with the Triumf to its chromatic aberration-free optical design, which corrects longitudinal chromatic aberration. The major difference in the design of the Triumf, in comparison with the FineVision trifocal IOL, is the height of the steps in its dual bifocal elements. This difference in design controls chromatic aberration. This does not correct the natural chromatic aberration of the eye, but it does correct any chromatic aberration introduced by the diffractive optic of the IOL (Figure 5). The expected benefit is an improvement of visual quality and high contrast sensitivity.
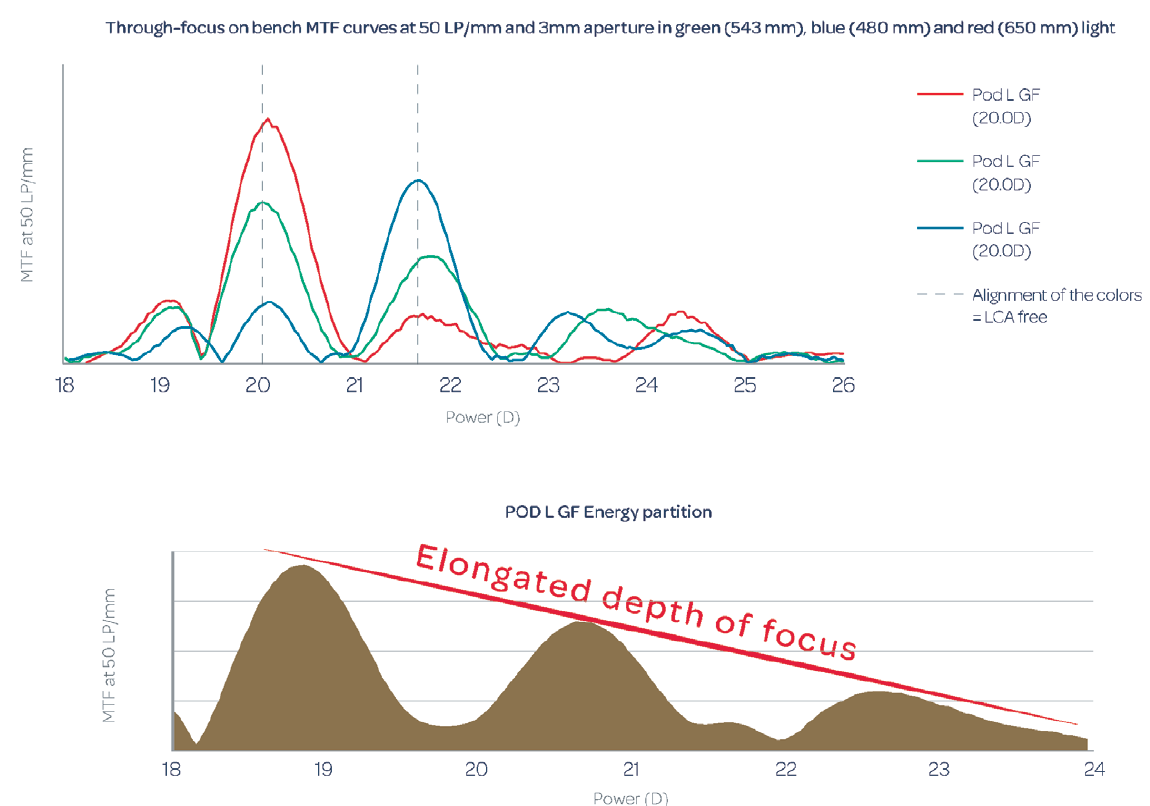
Figure 5. Through-focus on-bench modular transfer function curves at 50 lp/mm (data on file with PhysIOL).
IC-8
Small-aperture optics provide an effective physiologic presbyopia-correcting solution for a range of accommodation loss, regardless of cataract status.
The IC-8 IOL (AcuFocus; Figure 6 combines anaspheri monofocal IOL with an embedded opaquemini-rin to simultaneously address refractive error and presbyopia.

Figure 6. The IC-8 IOL.
Courtesy of AcuFocus
The one-piece hydrophobic acrylic IOL, which has modified C-loop haptics, an overall diameter of 12.5 mm, a 6-mm biconvex aspheric optic, and a square 360º posterior edge is implanted in the capsular bag at the time of cataract surgery. The embedded annular mask of the IOL has an outer diameter of 3.23 mm and a central aperture diameter of 1.36 mm.
Well tolerated, more forgiving than other IOLs
The IC-8 IOL has the CE Mark, and the company announced in June 2019 that enrollment for the pivotal US Investigational Device Exemption study had been completed. That 12-month prospective, multicenter, nonrandomized case control clinical trial is designed to evaluate the improvement in vision with the IC-8 IOL at all distances compared with traditional monofocal IOLs.1
I participated in a multicenter postmarket European trial of the IC-8 IOL implanted in 105 patients. In that study, 6 months after implantation, monocular UDVA was 0.87 logMAR (20/23 Snellen), UIVA was 0.83 logMAR (20/24), and UNVA was 0.66 logMAR (20/30).2
With respect to binocular vision, 99%, 95%, and 79% of patients enrolled in the trial had achieved 20/32 or better UDVA, UIVA, and UNVA, respectively.2
The majority of the 105 patients (95.9%) reported that they would select the IC-8 IOL again, and most (84.8%) reported using spectacles occasionally to never. Binocular contrast sensitivity matched the monocular contrast sensitivity achieved in the contralateral eye with a monofocal IOL.

Figure 7. Intraoperative view of the IC-8 IOL after laser cataract surgery.
Courtesy of H. Burkhard Dick, MD, PhD, FEBOS-CR
Patients in this study and others have tolerated as much as 1.00 D of residual refractive error, suggesting that the small-aperture concept of the IC-8 IOL may be more forgiving than monofocal and multifocal IOLs.2 Notably, astigmatic patients in a prospective clinical trial who received an IC-8 IOL and no additional astigmatic management were able to tolerate up to 1.50 D of refractive astigmatism.3
Suitable for a broad range of PATIENTS
A capsular bag implant such as the IC-8 IOL is suitable for a broad range of patients, including those with irregular corneas.4,5 The implant can be used in patients with naturally occurring corneal higher-order aberrations of 0.6 µm or more or after corneal refractive surgery and keratoplasty.
Patients with diseased corneas, keratoconus, or ocular trauma are also potential candidates for this technology. The majority of patients do well with this lens, but some patients with larger mesopic pupils (≥ 6 mm) may experience photic phenomena due to light coming around the outside edge of the opaque ring.
As a result, it is important to evaluate pupil size when considering the IC-8 lens.
1. AcuFocus completes study enrollment for U.S. IDE clinical trial of IC-8 lens. [press release]. Acufocus. June 14, 2019. http://www.acufocus.com/us/sites/default/files/MKU%20645%20Rev%20A%2C%20AcuFocus%20Completes%20Study%20Enrollment%20for%20U.S.%20IDE%20Clinical%20Trial%20of%20IC-8%20Lens.pdf. Accessed March 19, 2020.
2. Dick HB, Piovella M, Vukich J, et al. Prospective multicenter trial of a small-aperture intraocular lens in cataract surgery. J Cataract Refract Surg. 2017;43:956-968.
3. Ang RE. Small-aperture intraocular lens tolerance to induced astigmatism. Clin Ophthalmol. 2018;12:1659-1664.
4. Agarwal S, Thornell EM. Cataract surgery with a small-aperture intraocular lens after previous corneal refractive surgery: visual outcomes and spectacle independence. J Cataract Refract Surg. 2018;44(9):1150-1154.
5. Barnett V, Barsam A, Than J, Srinivasan S. Small-aperture intraocular lens combined with secondary piggyback intraocular lens during cataract surgery after previous radial keratotomy. J Cataract Refract Surg. 2018;44(8):1042-1045.
IsoPure 1.2.3
This EDOF monofocal IOL is less sensitive to defocus than a standard aspheric monofocal IOL.
The recently introduced IsoPure 1.2.3 (PhysIOL), based on isofocal technology, is an EDOF IOL that aims to overcome some of the limitations of existing EDOF lenses, including unwanted photopic phenomena.
The lens does this by using a unique anterior-posterior optical surface profile of increased negative spherical aberration that is fine-tuned for each diopter over the entire optic. Optical bench testing has shown that the optical system of the IsoPure 1.2.3 is less sensitive to defocus than a standard aspheric monofocal IOL and that it offers a continuous range of vision between distance and intermediate at any pupil size.
IN CLINICAL PRACTICE
I participated in the first multicenter prospective clinical study of the IsoPure lens. Thus far, 1-month outcomes are available for our study population of 19 patients (38 eyes).
The mean age of the 12 women and seven men in the study was 69.9 years. We aimed for a target of slight myopia in all eyes.
We found no statistically significant differences in postoperative refractive cylinder at 1 week and 1 month postoperative. In fact, the cylinder decreased slightly during the first month because the incision for lens implantation was created on the steepest corneal meridian.
As would be expected, monocular UDVA improved significantly from preoperative: 45% of eyes achieved 20/20 or better UDVA at 1 month postoperative. CDVA was 20/20 or better in 89% of eyes. Also at 1 month, monocular UIVA at 80 cm was 20/32 or better in 56% of eyes and 20/40 or better in 83% of eyes. DCIVA at 80 cm was 20/32 or better in 56% of eyes and 20/40 or better in 81%. DCIVA was also measured at 66 cm, at which distance 35% of eyes achieved 20/32 or better and 68% achieved 20/40 or better.
These data show that the IsoPure 1.2.3 can provide high-quality UCVA at both distance and intermediate lengths, and our results indicate that patients can see well from 77 cm to infinity.
Also at 1 month postoperative, the defocus curve (Figure 8) and contrast sensitivity were as expected. That is, the defocus range for visual acuity of at least 20/32 was -1.10 to 0.70 D, and contrast sensitivity for both photopic and mesopic was within the normal range for patients in this age group.
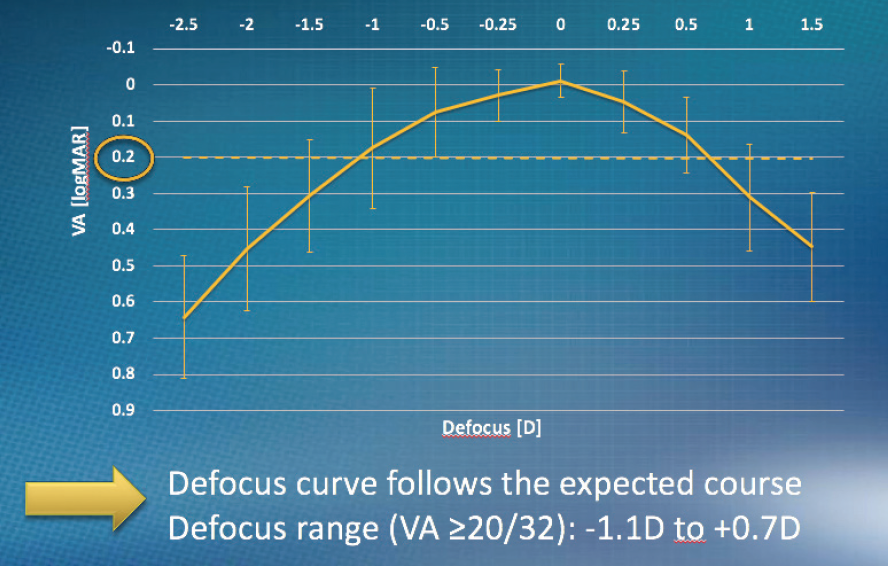
Figure 8. Defocus curve at 1 month postoperative.
Courtesy of PhysIOL
CONCLUSION
As with any new technology, longer follow-up in a larger number of eyes is warranted. For now, I look forward to using the IsoPure 1.2.3 in many more patients to provide them with increased depth of focus.
Lucidis T
Eyes with astigmatism can benefit from this lens’ extended depth of focus technology.
Spectacle independence after cataract surgery is probably one of the most frequently expressed patient desires that cataract surgeons hear preoperatively. Many solutions are available, including a wide range of IOL choices. EDOF IOLs are among the new categories of lenses we can offer patients for relief from presbyopia.
EDOF lenses have been found to be associated with fewer halos and less contrast sensitivity loss than traditional multifocal IOL technology.1 In the overall range of available IOL technologies, EDOF lenses appear to fit somewhere between monofocal and trifocal IOLs.
The Lucidis T IOL (Swiss Advanced Vision), launched during the 2019 ESCRS meeting, is a one-piece, foldable, multizonal refractive-aspheric bifocal toric EDOF IOL with closed C-loop haptics. It is designed for capsular bag implantation and made of a hydrophilic acrylic material with 26% water content.
The EDOF technology of the Lucidis T IOL uses a 1-mm central aspheric zone surrounded by a 5-mm refractive ring. The lens is available in a power range from +5.00 to +30.00 D and a cylinder power range from +1.00 to +4.50 D. According to the manufacturer, the main benefit of this particular design is to provide additional visual comfort at near and intermediate distances.2
CLINICAL EXPERIENCE
In our experience, the Lucidis T IOL technology seems to allow us to meet our promises to patients. That is, most of our patients who receive this lens report additional comfort at near and intermediate distances after cataract surgery. Further, with the Lucidis T IOL we have not encountered any centering problems, and no rotation of the IOL has been noted in our series to date.
We like to inject the implant using a manipulator in the other hand in order to provide stability, to maintain a constant IOP, and to give us the ability to manipulate the IOL at any time during unfolding (Figure 9). Rotation of the IOL into position is easy. Even though the lens has a closed C-loop haptic design, we recommend rotating it clockwise. Some surgeons have suggested first removing all dispersive OVD from about 20° before the target axis, but we think there is no need to do so with this lens.
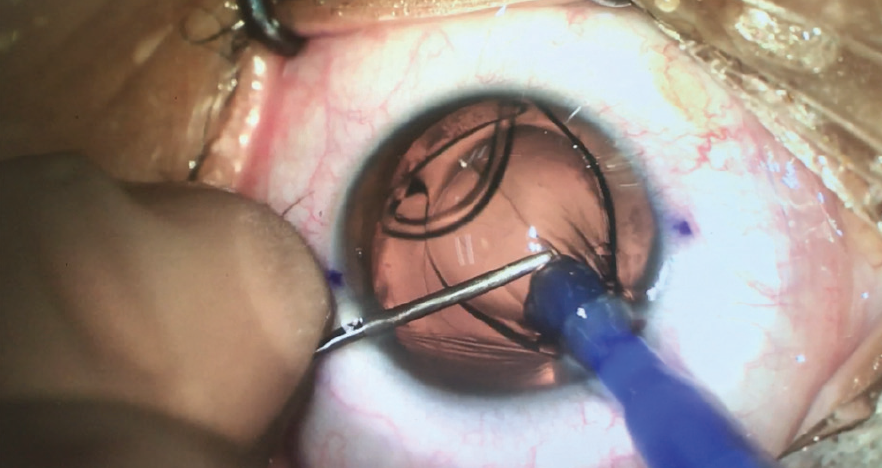
Figure 9. During Lucidis T IOL implantation, a manipulator is used in the nondominant hand.
Figures 9–11 courtesy of Elie Motulsky, MD, PhD, FEBO
Irrigation and aspiration can be done while the IOL is aligned in the target axis (Figure 10). When the I/A instrument is removed, no rotation is observed, and the IOL remains perfectly aligned to the target axis at the end of surgery (Figure 11).
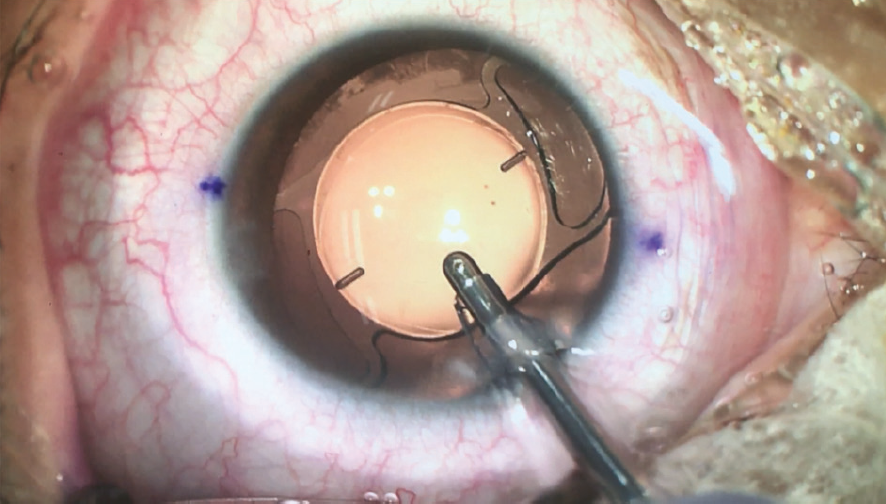
Figure 10. The Lucidis T IOL is aligned to target axis during irrigation and aspiration.
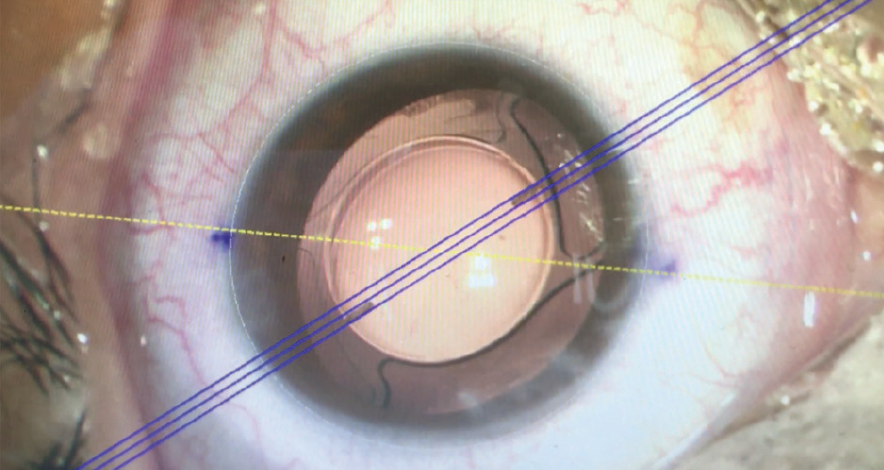
Figure 11. The IOL remains aligned to the target axis after the I/A instrument is removed at the end of the surgery.
CONCLUSION
The Lucidis T IOL seems to be a good choice for patients with astigmatism who want to benefit from EDOF technology after cataract surgery. More clinical studies are needed to show the benefits and drawbacks of this lens. Our first clinical impressions are quite enthusiastic. No patient has complained of halos, and most patients have good far vision, often with additional comfort in near and intermediate vision.
1. Breyer DRH, Kaymak H, Ax T, Kretz FTA, Auffarth GU, Hagen PR. Multifocal intraocular lenses and extended depth of focus intraocular lenses. Asia Pac J Ophthalmol (Phila). 2017;6(4):339-349.
2. Gillmann K, Mermoud A. Visual performance, subjective satisfaction and quality of life effect of a new refractive intraocular lens with central extended depth of focus. Klin Monbl Augenheilkd. 2019;236(4):384-390.
Nanex multiSert+
The injector system and the material of this IOL combine to allow insertion through a very small wound.
In 2019, I had the opportunity to evaluate the Nanex multiSert+ preloaded IOL and injection system (Hoya; Figure 12). One year previously, I had tested the Vivinex multiSert preloaded IOL and injector system (Hoya) and had been impressed by the performance of this device. The feeling of control, consistency, and ease of use with both systems were a level above any other system I had used before.
Figure 12. The Nanex multiSert+ preloaded IOL (right) and injection system (left).
Courtesy of Hoya
The design and working mechanism of the Nanex multiSert+ system is identical to that of the Vivinex multiSert. The difference is the lens material. Although the hydrophobic acrylic material of the Vivinex is newer, the material of the Nanex IOL is Hoya’s proven AF-1 hydrophobic acrylic, and the Nanex is the first AF-1 IOL to be treated with Hoya’s proprietary active oxygen processing treatment to reduce posterior capsular opacification. That treatment was previously available only on the Vivinex platform.
The properties of the AF-1 material allow the Nanex to be inserted through a cartridge tip smaller than was previously possible. I can successfully implant the Vivinex multiSert through an unenlarged 2.2-mm incision, and now the Nanex multiSert+ is just one degree easier to insert through an incision of that size. According to the manufacturer, the 1.62-mm injector tip of the multiSert+ is the world’s smallest nozzle size in an open-loop preloaded hydrophobic IOL system. I believe that the nozzle will allow surgeons to inject the lens through incisions as small as 1.8 mm.
AWAY FROM WOUND-ASSISTED TECHNIQUE
After years of wound-assisted IOL injection techniques, I have now converted to a fully through-the-wound technique. This avoids potential issues with a wound-assisted technique, including the specific skills required for successfully using such a technique, the possibility of a failed injection, and, worse, the possibility of a trapped IOL unfolded halfway through the incision that cannot move forward nor backward.
The theoretical advantage of wound-assisted implantation is the ability to use a smaller incision size, but some preloaded systems have large tips that, for me, were not compatible with a 2.2-mm incision. With both Hoya multiSert systems, I find it much easier to inject through a 2.2-mm incision. And I observe better wound integrity after injection. With the Nanex multiSert+ system I do not have to perform stromal hydration of the main incision, and it is consistently completely watertight.
EASE OF USE
The ease of use of both multiSert devices is astounding. Preparation by the operating assistant is straightforward, but it is the feeling for the surgeon during insertion in which this device surpasses others. Due to a dual mechanical design that moves the IOL forward, the surgeon experiences total direct tactile control with an effortless forward plunger movement. The sudden drop-off of force upon lens release that occurs with many injectors does not occur with these.
Additionally, the unique ability to switch between push and screw mode allows the surgeon to control implantation precisely. This is useful in challenging cases such as eyes with narrow pupils and zonular weakness. Because the inserter is fully through the wound, the surgeon can advance the plunger slowly without risk of the IOL unfolding inside the incision, making one feel much more in control and at ease than with other systems.
The versatility of the multiSert injectors is further enhanced by the incorporation of an insert shield designed to limit incision stretch and reduce induced corneal astigmatism. This shield also enhances control; when the injector is pushed against the eye, the position of the tip remains stable and cannot protrude too far into the eye or stretch the wound too much.
CONCLUSION
From my clinical experience, it is evident that the Nanex multiSert+ is a best-in-class preloaded IOL injector system. It allows, if desired, the use of incisions as small as 1.8 mm while maintaining the unprecedented control and safety of its predecessor, the Vivinex multiSert.
Precizon CTF
A lens with a multisegment optical design provides a continual range of focus.
One recent improvement in premium IOLs is the incorporation of a continuous range of optical powers. The intent behind this design is to provide a more natural visual experience than traditional multifocal IOLs and to minimize unwanted optical side effects.
I have early clinical experience with the Precizon Continuous Transitional Focus (Precizon CTF, Ophtec), a lens that uses a unique multiple-segment optical design to provide patients with clear vision at multiple distances.
SEGMENTAL DESIGN
The optic of the Precizon CTF contains multiple segments and sectors on its anterior surface, rather than the concentric rings seen on most multifocal IOLs. In my experience, this translates to excellent far and near vision and a smooth transition between the zones (Figure 13) for patients. Further, experience has shown that this lens has less diffusion of light transmission than is seen with diffractive designs.
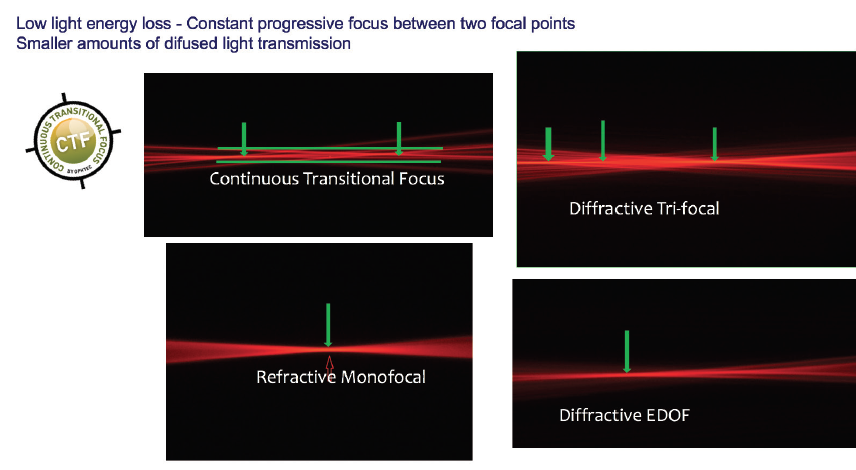
Figure 13. The Precizon CTF provides constant progressive focus between two focal points and smaller amounts of diffused light transmission.
Figures 13 and 14 courtesy of Ophtec
The Precizon CTF is available in two models (the 570 and the NVA), each with unique segment sizes, light distribution characteristics for far and near vision, and IOL aberrations. In the 570, light is distributed equally (50/50) for distance and near. In the NVA, the central distance optical zone is larger, so that the distribution of light is 60% for distance and 40% for near. This translates into a more natural distribution of light and takes into account intermediate vision. Normally, I prefer the 570 for elderly patients with less intermediate vision activity and the NVA in younger patients, who have an increased demand for intermediate vision correction.
The Precizon CTF’s optic is divided into three concentric sectors. The two peripheral sectors provide a 50/50 or 60/40 distribution of distance and near correction, depending on the IOL model, and the distribution changes among four segments in each sector (Figure 14). The central sector is dedicated to distance correction. In total, there are 11 distinct segments—five for distance vision and six for near—within the optical zone, creating a smooth and continuous transitional focus. This design facilitates a continuous defocus curve, sharpening images on the retina from infinity to intermediate focus and from intermediate to near.
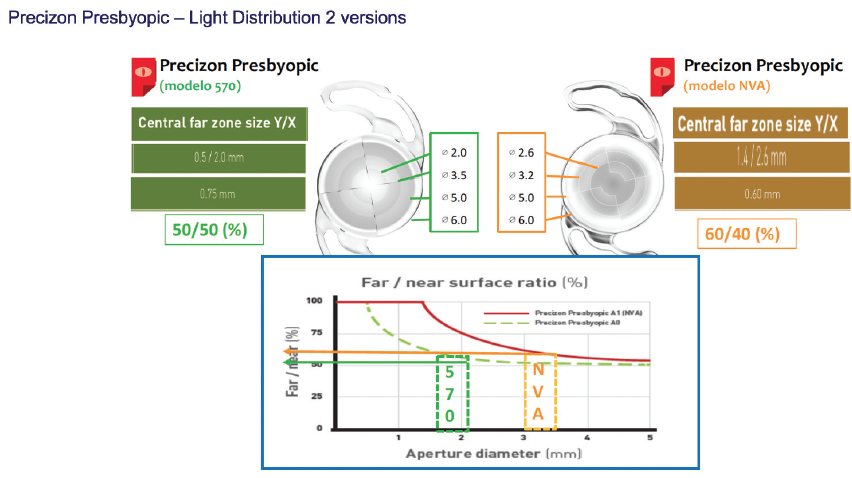
Figure 14. The Precizon CTF is available in two models.
CLINICAL RESULTS
A prospective European multicenter study of the Precizon enrolled 60 patients who were scheduled for either cataract surgery or refractive lens exchange.1 At 3 months postoperative, 80% of patients reported achieving spectacle independence. Patients also had good UCVA for near, intermediate, and distance vision. The majority of patients had no complaints of halos or glare at night; about 5% reported a variety of visual disturbances at night during the first 3 months of follow-up.
At my center, we enrolled 20 patients in the study. At 3 months postoperative, 100% of patients were within ±0.50 D of sphere target, and 87.5% were within ±0.50 D of their cylinder correction target. Further, 37.5% were within ±0.25 D of sphere and 37.5% were within ±0.25 D of cylinder correction targets.
CONCLUSION
The Precizon CTF IOL represents a new paradigm in lens-based presbyopia correction procedures. With its anterior surface segmental design, the lens provides excellent far and near vision with a smooth transition between zones, in essence creating a constant progressive focus between focal points. Because the Precizon CTF mimics the eye’s natural process of accommodation, patients should experience only a brief period of neural adaptation. As with any IOL, patient selection is crucial with the Precizon CTF.
1. Holzer M. Functional outcomes and patient satisfaction of the new Precizon presbyopic multifocal intraocular lens. Paper presented at: the European Society of Cataract and Refractive Surgeons Annual Meeting; October 7-11, 2017; Lisbon, Portugal.
RayOne Trifocal Toric
A new IOL that checks all the boxes.
I am always keen to add a new presbyopia-correcting IOL to my repertoire in the never-ending quest to improve patient outcomes and satisfaction. When considering a new lens, I look for several features:
- An established manufacturer;
- A lens material with a proven track record;
- A lens platform already demonstrated to be safe and effective;
- Excellent visual performance;
- An option to have the IOL preloaded;
- A microincision push injector for the IOL that supports a wound-assisted technique;
- Low incidence and severity of glare and halos;
- Low incidence of posterior capsular opacification; and
- For a toric IOL, a wide cylinder range and rotational stability.
I was therefore more than happy to participate in a multicenter evaluation of the RayOne Trifocal Toric IOL (Rayner; Figure 15) prior to its formal global launch at ESCRS Paris 2019. When I was presented with the specifications of the IOL, I was reassured to see that they fulfilled all my requirements.

Figure 15. A well-centered RayOne Trifocal Toric IOL seen with retroillumination.
Figures 15 and 16 courtesy of Amir Hamid, BMedSci, BMBS, FRCOphth, CERTLRS
This multicenter pilot study involved five sites and five surgeons in Germany, the United Kingdom, and Japan. Patients with bilateral visually significant cataract and no ocular comorbidity were enrolled after fully informed consent was obtained. After bilateral IOL implantation, the primary endpoints of the study were visual acuity, subjective refraction, cylinder reduction, and surgeon satisfaction at 3 months postoperative. The results of 20 eyes of 10 patients were available for analysis.1
RESULTS
Figure 16 summarizes the results. All patients were within ±0.50 D and 57% were within ±0.25 D of emmetropia. Regarding astigmatism correction, 100% of eyes had 0.75 D or less of astigmatism postoperatively, and 71% had 0.50 D or less.
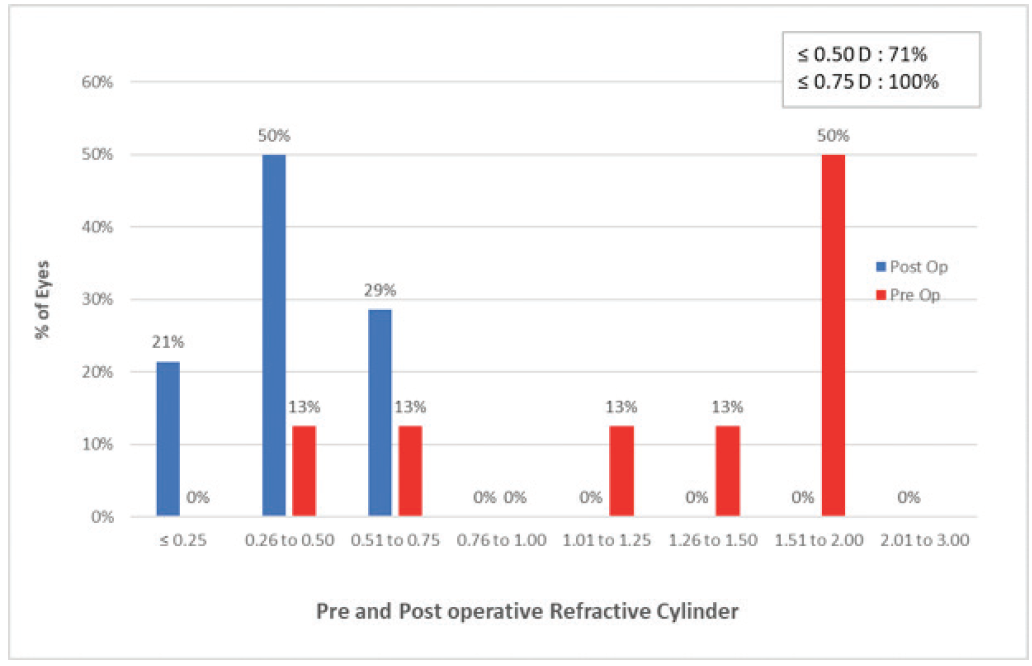
Figure 16. Pre- and postoperative refractive cylinder with the RayOne Trifocal in 20 eyes of 10 patients.
More important, 90% of patients achieved monocular and binocular uncorrected distance, intermediate, and near visual acuity of 0.1 logMAR or better (Table). And finally, all surgeons reported high levels of satisfaction with the lens and its injector system.
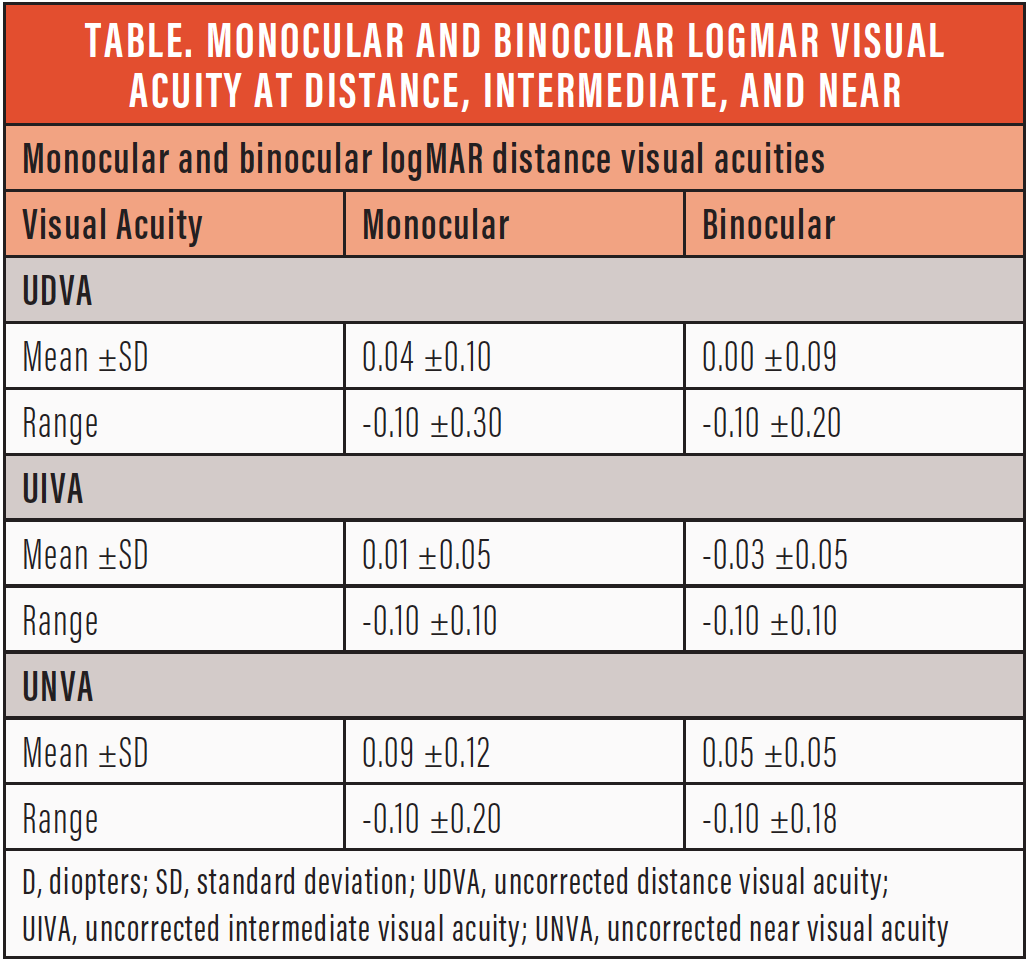
CONCLUSION
The RayOne Trifocal Toric IOL is a promising new entry in the presbyopia-correcting toric IOL category for surgeons. Initial results were excellent, with high levels of satisfaction among both patients and surgeons. Additionally, the RayPro digital platform (Rayner) for collecting patient-reported outcomes allows surgeons to continuously monitor long-term outcomes and satisfaction among our patients implanted with this and other presbyopia-correcting IOLs.
1. Barsam A, Butt A, Thomson P. First visual results after implantation of the RayOne trifocal intraocular lens. Poster presented at: the 24th ESCRS Winter Meeting; February 21-23, 2020; Marrakech, Morocco.
EDOF treatment with the Light Adjustable Lens
In clinical trials of the Light Adjustable Lens (RxSight), investigators reported excellent results with this first-of-its-kind IOL, including reduction in treated astigmatism, excellent UCVA, and a sharp reduction in residual spherical error.1
In the initial iteration of the procedure, the Light Adjustable Lens could be treated postoperatively with the Light Delivery Device (RxSight) to adjust sphere and cylinder. Now, European surgeons have gained access to an extended depth of focus (EDOF) treatment for the Light Adjustable Lens. This treatment modality is also already approved in Mexico, according to Market Scope.2
RxSight has demonstrated the EDOF adjustability in 18 patients who underwent bilateral implantation of the Light Adjustable Lens. All were given EDOF adjustments, and all achieved uncorrected distance and intermediate visual acuities of 20/20 or better and uncorrected near visual acuities of 20/25 or better.2
1. FDA approves first implanted lens that can be adjusted after cataract surgery to improve vision without eyeglasses in some patients [press release]. US Food and Drug Administration. November 22, 2017. https://www.fda.gov/news-events/press-announcements/fda-approves-first-implanted-lens-can-be-adjusted-after-cataract-surgery-improve-vision-without. Accessed March 19, 2020.
2. RxSight begins initial commercialization of Light Adjustable Lens in the US. Market Scope Ophthalmic Market Perspectives. September 26, 2019. https://www.rxsight.com/media/rjrbad2g/marketscope_2019_sep_26.pdf. Accessed March 19, 2020.
Tecnis Synergy
The EDOF addition improves near vision without sacrificing distance vision.
The Tecnis Synergy IOL (Johnson & Johnson Vision; Figure 17) was introduced in Europe last year, and since that time I have successfully implanted about 300 of them. Two of my senior optometrists, who have decades of experience and have been involved with more than 10,000 presbyopia-correcting IOL cases each, have chosen bilateral Tecnis Synergy lenses for their own eyes, which I think is a testament to the excellence of this lens.

Figure 17. The Tecnis Synergy IOL.
Figures 17 and 18 courtesy of Johnson & Johnson Vision
The Synergy combines diffractive multifocal optics with an EDOF addition at near. It also filters ultraviolet and violet light, but it does not filter blue light. I consider it to be most similar to a trifocal lens. Compared with trifocal lenses, the Tecnis Synergy has a more natural range of near to intermediate vision and better mesopic near vision (Figure 18).
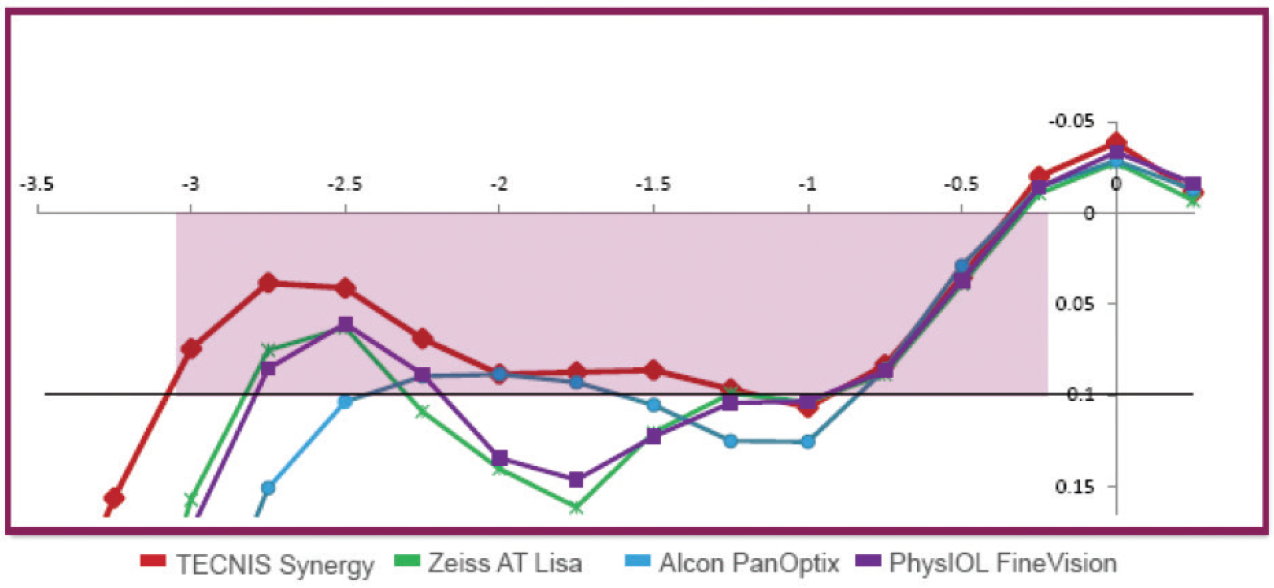
Figure 18. The Tecnis Synergy IOL offers a natural, continuous range of vision from near through intermediate, as well as high-quality distance vision (data on file with Johnson & Johnson Vision).
NEAR GAINS
The near point of the Synergy is a little closer than the near point of trifocal IOLs, at about 35 cm. From the near point through about 70 cm, the lens’ diffractive echelette optics provide a continuous range of near to intermediate vision that patients say feels natural. A range like this is ideal for working-age patients who wish to be able to look from their desk to their computer to a colleague a few feet away, or for those who are shopping and cooking and need to be able to see items on the grocery shelf or in their kitchen without constantly adjusting how far away they are from the objects.
The Synergy is relatively pupil independent, and, in my experience, patients report that they can still read in dim light. In fact, I have heard no complaints about near vision at all.
DISTANCE AND NIGHT VISION
The near-vision gains associated with the Synergy IOL have been achieved without sacrificing the excellent quality of distance vision that we have come to expect from the Tecnis platform. Like the other lenses in this IOL family, the material induces minimal chromatic aberration, and this is further reduced by the optic design, especially in dim light. When the pupil expands, the diffractive pattern corrects even more chromatic aberration, which explains the high-quality vision patients report in low light situations. I have also found that the lens reduces spherical aberration to near zero in most eyes.
Of course, because it has diffractive optics, the Synergy still has some issues with glare and halos at night, but these photic phenomena are generally well tolerated. My patients report that they are an acceptable trade-off for the quality and range of vision.
NO TORIC YET
The only other disadvantage of this lens is that it is currently not available in a toric version. In our refractive clinic, more than half of patients need correction of astigmatism, so this limits the number of patients who are good candidates for the Synergy lens. If a toric option becomes available, the Synergy will certainly be the most frequently implanted presbyopia-correcting IOL in my clinic.
Vivity
Natural vision at a range of distances provided by a novel optical technology.
Recently, the increasing number of advanced-technology IOLs available for the correction of presbyopia and astigmatism has changed my approach to patient management. In my practice, I need to understand a patient’s visual needs in order to provide him or her with the best surgical treatment.
Every decision requires consideration of the patient’s ocular health status, including the presence of regular astigmatism, the absence of pathologic conditions such as maculopathy or glaucoma, and a good tear film. For a long time, there wasn’t much problem choosing the right IOL because most IOLs were monofocal. They were not often associated with issues such as glare, low contrast, and other visual disturbances.
Now, the AcrySof IQ Vivity IOL (Alcon; Figure 19) is providing vision like that of a monofocal IOL, with clear visual acuity at a range of distances, without the drawbacks of existing multifocal IOL technologies. This innovation provides a newfound stability in IOL selection and new possibilities in visual correction.

Figure 19. The AcrySof IQ Vivity IOL.
Courtesy of Edoardo Ligabue, MD
NATURAL VISION
From my first experience with the Vivity, its distinguishing features have been its ease of use and a visual disturbance profile like that of a monofocal IOL. In my experience, patients with the Vivity have natural vision with a continuous extended focal range from distance to intermediate (66 cm) to functional near; in bright light, they can easily read up to J3, and they report high satisfaction.
The Vivity is pupil-independent and is not affected by angle kappa. Contrast sensitivity, distance visual acuity, glare, and night vision quality are comparable to the characteristics of a monofocal IOL.
The Vivity does not use diffractive or refractive technology to provide presbyopia correction. Instead, the lens uses Alcon’s novel X-Wave technology, which is completely different from the technologies used by all other IOLs in the market. The X-Wave technology creates an extended focal range by stretching and shifting the wavefront, as opposed to splitting the wavefront into multiple focal points as diffractive multifocal lenses do.
By using all available light, this IOL maintains the same vision quality as a monofocal IOL: excellent distance vision, intermediate sharp vision for active lifestyles, and good quality vision for close-up daily activities (Figure 20).1
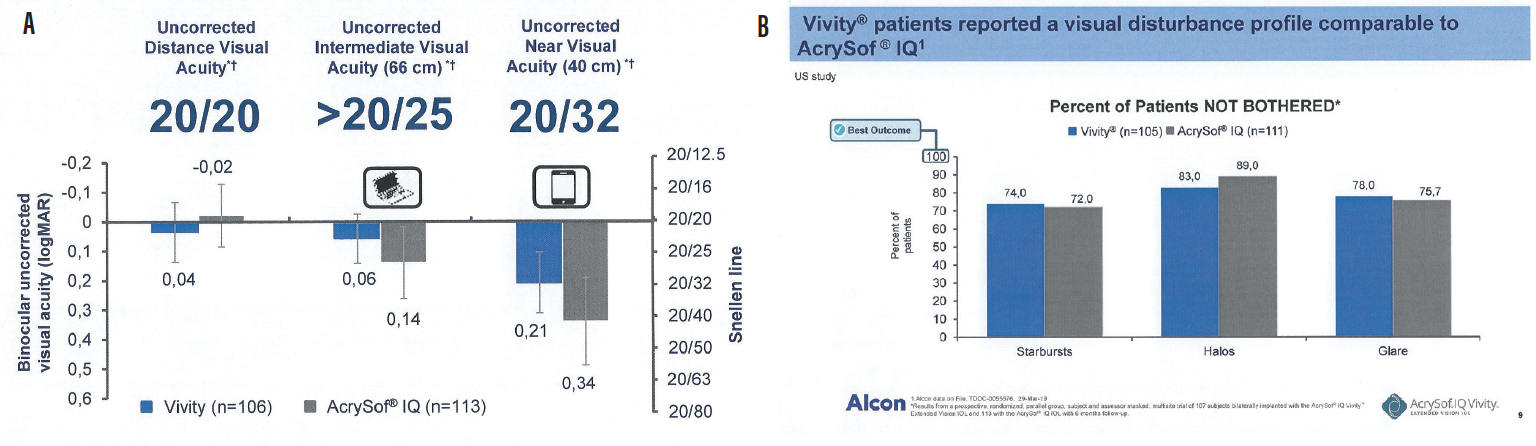
Figure 20. Uncorrected vision (A) and visual disturbance (B) results from a prospective, randomized, parallel-group, patient and assessor masked, multisite trial of 107 patients bilaterally implanted with the AcrySof IQ Vivity IOL and 113 with the AcrySof IQ IOL with 6-month follow-up. AcrySof IQ Vivity Alcon data on file (TDOC-0055576, March 29, 2019).
Courtesy of Alcon
CLINICAL EXPERIENCE
The results shown in Figure 20, from the first clinical studies of the AcrySof IQ Vivity, are in line with the results I have seen with my patients in my first 4 months implanting the Vivity.
When I propose a diffractive IOL to my patients, I always ask them if they can accept loss of a little bit of distance visual quality to gain independence from spectacles. But when I discuss the Vivity IOL, offering excellent distance visual quality with occasional spectacle use to read small text, the choice is unanimous: Quality of vision is patients’ chief preference.
The AcrySof IQ Vivity allows me to have no worries about refractive outcomes. In my experience, it is forgiving regarding biometric accuracy, it is not sensitive to tear film variations, and it does not impair visual acuity in patients with maculopathies or glaucoma. The lens provides stability in the capsular bag and is tolerant to slight decentration. The Vivity is also available in a toric IOL model, allowing patients to achieve astigmatism correction at the time of cataract surgery.
CONCLUSION
The Vivity is commercially available in selected European markets at present. Availability in additional countries will follow throughout 2020, according to Alcon. Based on my experience to date, the AcrySof IQ Vivity IOL will soon become the first IOL choice for most of my patients. It has changed the game by improving patients’ visual performance, making my job easier and more relaxing.
1. Data on file. Alcon.
xact Mono-EDoF
This lens acts like a true monofocal while providing a continuous focus from distance to intermediate.
The ongoing quest for the perfect presbyopia-correcting IOL has led to a series of compromises involving the trade-offs of multifocal dysphotopsia and reduced contrast sensitivity in exchange for spectacle independence. Although the benefits outweigh the risks for many patients, the relatively high cost of premium IOLs can exacerbate some patients’ dissatisfaction and lead to secondary surgery and IOL explantation.1
A new entry in the field of presbyopia correction seeks to minimize risk by delivering quality of vision and contrast sensitivity comparable to those of a monofocal IOL while providing an extended depth of focus with very low incidence of halo and glare compared with other diffractive EDOF lenses.
The xact Mono-EDOF IOL (Santen), a one-piece lens manufactured from a glistening-free hydrophobic acrylic material, meets international bench testing criteria for a monofocal IOL and has received the CE Mark as a monofocal IOL. Its optic, which incorporates four diffractive rings (Figure 21), demonstrates a single, broad through-focus modulation transfer function (MTF) peak. In comparison, traditional EDOF IOLs exhibit dual peaks.2 MTF measurements and retinal image modeling show that the xact IOL has a higher tolerance to tilt and decentration than other EDOF IOLs with at least 40% higher MTF. This directly affects the optical quality of the image delivered to the retina.3

Figure 21. The xact Mono-EDOF IOL.
Figures 21 and 22 courtesy of Santen
CLINICAL RESULTS
Clinical trials with the xact Mono-EDOF IOL have taken place at several sites in Europe, including clinics in Germany, the United Kingdom, France, and Portugal. The largest body of data to date has been provided by the International Vision Correction Research Centre at the University of Heidelberg, Germany.
In the Heidelberg study of the xact IOL, for which I am the medical monitor, 44 eyes of 25 patients (10 men and 15 women) have been followed for 9 months.4 The mean age of patients is 70.3 years, and their mean preoperative monocular BCVA was 0.33 ±0.16 logMAR (equivalent to 20/40-).
At 9 months, mean monocular BCVA improved to -0.05 ±0.13 logMAR (20/20+), and mean monocular UCVA was 0.05 ±0.19 logMAR (20/20-). Monocular DCIVA at 66 cm was 0.21 ±0.11 logMAR (20/30-), and monocular UIVA at 66 cm was 0.18 ±0.13 logMAR (20/30+).
With binocular testing, DCIVA was logMAR 0.09 ±0.13, and UIVA was logMAR 0.08 ±0.10 (20/25+).
Monocular and binocular defocus curves are shown in Figure 22. Binocular depth of focus at 20/30 is estimated to be 1.30 D. Of note, the defocus peak demonstrates a broad area within the range of ±0.50 D where visual acuity is better than 20/20, suggesting a relatively forgiving tolerance to normal variations in the accuracy of IOL power calculation. One would therefore expect binocular UDVA of 20/20 or better within the dioptric range of ±0.50 D postoperative manifest refraction spherical equivalent.
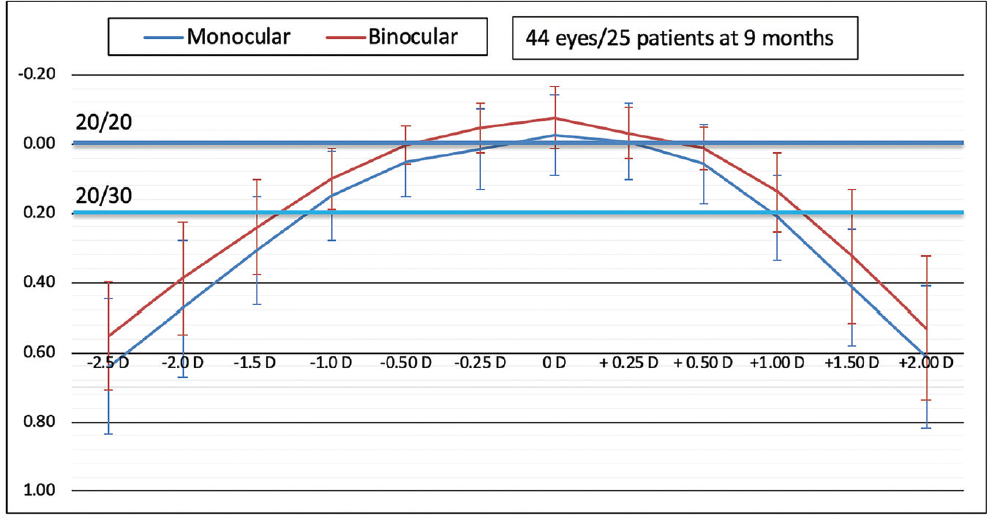
Figure 22. Defocus curves demonstrate 1.30 D binocular defocus at 20/30 and a broad peak within the range of ±0.50 D postoperative manifest refraction spherical equivalent (data on file with Santen).
Testing with the Halo and Glare Simulator (Eyeland Design Network) demonstrated only slight interference from dysphotopsia. Approximately 75% of patients reported little difficulty driving at night. Contrast sensitivity measured within the expected range for monofocal IOLs.
CONCLUSION
The xact Mono-EDOF IOL is an exciting entry in the field of presbyopia-correcting IOLs, providing quality of vision similar to that of a monofocal IOL with vision in the intermediate range like that of an EDOF IOL. Early clinical results suggest that this lens will be a valuable addition to cataract surgeons’ repertoire of lenses.
1. Packer M. Enhancements after premium IOL cataract surgery: tips, tricks, and outcomes. In Starr C, ed. Current Ophthalmology Reports. New York: Springer Science + Business Media; 2013.
2. Spalton DJ, Packer M, So Y, Tiwari N, Venkateswaran K. Optical and clinical comparison of a novel monofocal extended-depth-of-focus IOL and a conventional bifocal extended-depth-of-focus IOL. Paper presented at: ASCRS Annual Meeting; May 4, 2019; San Diego, CA.
3. Spalton DJ, So Y, Tiwari N, Venkateswaran K. The effect of tilt and decentration on a novel extended-depth-of-focus IOL compared to conventional extended-depth-of-focus IOLs. Paper presented at: ASCRS Annual Meeting; May 4, 2019; San Diego, CA.
4. Weindler J, Baur I, Son HS, et al. Prospektive klinische Studie mit einer neuen monofokalen Intraokularlinse mit erweiterter Tiefenschärfe. Paper presented at: Kongress der DGII; February 13, 2020; Mainz, Germany.
Triva-aA
HumanOptics received the CE Mark in February, granting approval in the European market for its trifocal Triva-aA IOL. According to the company, the optimized design of the Triva-aA enables patients to enjoy good vision at near, intermediate, and distance and is particularly suitable for performing screenwork including the use of tablets and smartphones.
The Triva-aA is produced with high-precision sub-nano resolution technology, requiring appropriate ultra-precision lathes. According to the company, this feature makes subsequent polishing superfluous.

















