Managing a Malpositioned Toric IOL
Intraoperative guidance can help to ensure rotation to the correct axis.
Toric IOLs are a versatile and robust solution for the correction of corneal astigmatism during cataract surgery. However, this benefit can vanish when the lens is not placed on the right axis of the eye or when it rotates after surgery.1-6 On average, a rotation of 3° reduces astigmatic correction by around 5%.
Our opinion is that, if a toric IOL is misplaced by 5° or more, further correction is required. Usually such correction can be achieved by IOL rotation based on one of the online toric IOL calculators, with the correct astigmatic axis marked either manually or with the assistance of a device such as the Callisto eye (Carl Zeiss Meditec). This secondary surgery should be conducted before the process of fibrosis begins in the lens capsule. Otherwise, IOL rotation can be difficult and can even lead to zonular damage.
In the case described here, we illustrate the impact of a minimal misplacement of a toric IOL and the positive effect of corrective rotation on the final refraction.
CASE REPORT
A 50-year-old woman presented to us after uneventful refractive lens exchange (RLE) with toric trifocal IOLs implanted bilaterally. Preoperative refraction in the patient’s right eye was -8.00 -3.00 x 168°. In December 2019, RLE was performed with implantation of an AT LISA tri toric 939MP (Carl Zeiss Meditec), with sphere power of 10.50 D and cylinder of 3.00 D, at 72° with manual marking of the axis.
At 6 weeks postoperative, refraction appeared to be +0.75 -1.25 x 17°, and the patient’s corrected distance visual acuity (CDVA) was 0.1 logMAR, uncorrected distance visual acuity (UDVA) was 0.3 logMAR, uncorrected near visual acuity at 40 cm was 0.3 logMAR, and uncorrected intermediate visual acuity at 70 cm was 0.4 logMAR. The IOL was found to be on the 55° axis, in contrast with the intended axis of 72°. The patient complained of uncorrected double vision and a high level of dysphotopsia.
Ten weeks after implantation, IOL rotation was performed. In surgery, the clear corneal paracenteses were reopened, and the anterior chamber was stabilized with a 1% hyaluronic acid OVD. The anterior capsule was then slowly loosened from the anterior optic, and the haptics were freed using the same OVD on a Sauter hydrodissection cannula.
The correct axis was defined intraoperatively using the Callisto eye (Figures 1 and 2) and based on the recommendation of the Toric Results Analyzer of Berdahl and Hardten. The IOL was rotated 12° counterclockwise from the measured position to reach the 67° axis. After the IOL was rotated to the intended axis, the position was confirmed using the Callisto eye. The OVD was then extracted using bimanual irrigation and aspiration, and the paracenteses were hydrated with balanced saline solution.
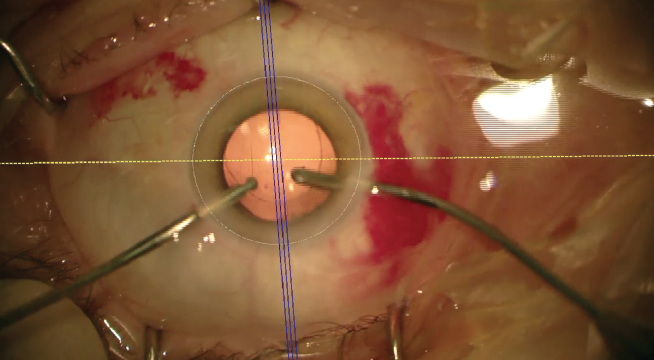
Figure 1. Adjusting the reference image to intraoperative view with Callisto eye.
Courtesy of Florian T.A. Kretz, MD, FEBO
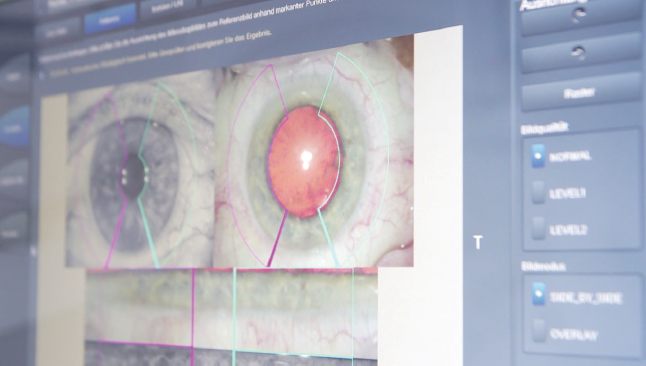
Figure 2. Intraoperative IOL alignment of the AT LISA tri toric with Callisto eye.
One day after the rotation procedure, the refraction in the right eye was +0.25 -0.75 x 21°, and UDVA improved to 0.1 logMAR with a CDVA of 0.0 logMAR. One week later, UDVA had improved to 0.0 logMAR and CDVA to -0.1 logMAR with a residual refraction of +0.25 -0.50 x 44°.
As the patient had already noticed significant improvement at day 1 and no more double vision or dysphotopsia, no further treatment seemed necessary.
DISCUSSION
Miscalculation of toric IOL marking points or refractive error, misplacement of the IOL during or after surgery, and pre- or postoperative dry eye disease can lead to residual astigmatic error. Some of these factors are not predictable, and even accurate preoperative calculations can result in residual astigmatism after cataract surgery.
In general, we suggest secondary rotation of a toric IOL misaligned by 5° or more. Misalignment of less than that amount is difficult to correct, and the influence of opening the wound itself could cause a shift of axis.
Another key factor is the timing of IOL rotation. Especially in cases of early rotation, the capsular bag may still be too large, and the IOL could rotate again. We prefer to perform rotation at around 6 to 15 weeks after the primary surgery, so that the capsule has shrunk already but fibrosis is minimal.
By following these guidelines, we have experienced success in correcting misaligned toric IOLs. We hope these pointers are helpful for readers who encounter this bothersome postoperative complication of toric IOL implantation.
1. Kahl KL. IOL rotation can treat residual astigmatism after toric lens implantation. Ocular Surgery News. March 10, 2015.
2. Hahn U, Krummenauer F, Schmickler S, Koch J. Rotation of a toric intraocular lens with and without capsular tension ring: data from a multicenter non-inferiority randomized clinical trial (RCT). BMC Ophthalmol. 2019;19(1):143.
3. Roach L. Toric IOLs: four options for addressing residual astigmatism. EyeNet. April 2012.
4. Feng S, Jung HC. Surgical tips for toric lens rotation . American Academy of Ophthalmology. January 8, 2019. https://www.aao.org/clinical-video/surgical-tips-toric-lens-rotation. Accessed March 18, 2020.
5. Hillman L. Toric IOL rotated—now what? EyeWorld. October 2018.
6. Buznego C, Kontos M, Raviv T, Whitman J, Whitsett JC. Postoperative rotation of a toric IOL. CRST. 2012;8;17-18.
Management of Dislocated Presbyopia-Correcting IOLs
Strategies may be chosen based on the particulars of the case.
As innovation surrounding cataract and refractive surgery evolves, we are afforded the opportunity not only to improve but also to enhance our patients’ vision. The results achieved with presbyopia-correcting IOLs have steadily improved over time, and at the same time potential side effects have decreased.
That being said, most cataract surgeons understand that not every patient is an ideal candidate for a presbyopia-correcting IOL.1,2 Certain personality traits, career profiles, and anatomic findings can predispose patients to a less-than-successful surgical result.1 Screening patients carefully for findings such as zonular laxity and phacodonesis and providing comprehensive explanations of the risks, benefits, and alternatives associated with presbyopia-correcting IOLs can help surgeons to produce happy postoperative patients.
Still, there will be cases—fortunately not commonly—when a patient may experience dislocation of a presbyopia-correcting IOL.2-6 Early causes of dislocation include complications during surgery (eg, vitreous loss, zonular loss, zonular damage) and IOL malpositioning. Late causes of dislocation can include trauma, pseudoexfoliation, capsular contraction, and subsequent posterior segment surgical procedures. There have been case reports of IOL decentration after intravitreal injection, and IOL tilt can occur after vitrectomy as well.6
HAVE A PLAN
Because centration is crucial to the performance of presbyopia-correcting IOLs, it is important not only to recognize decentration when it happens but also to have a plan for management.2-5,7,8 It is also crucial to check for signs of decentration before performing Nd:YAG capsulotomy; the posterior capsular opening can make subsequent repositioning or exchange surgeries more difficult.2
Management strategies for a decentered presbyopia-correcting IOL can be chosen based on the severity, location, and nature of the decentration. Mild decentration can be managed by returning to the OR, meticulously polishing the capsule to remove lens epithelial cells, and placing a capsular tension ring (CTR). All of these can be done through a small incision.5,8
When a CTR is used, it can be placed into the capsular bag at any time after creation of the capsulorhexis, generally at the first sign of zonular weakness. In most cases, the CTR will provide satisfactory stability, but in severe instances the capsule may remain loose or decentered. In this event, a capsular tension segment with or without scleral fixation of the capsular bag–IOL complex may be necessary.
If the IOL is partially subluxed out of the bag (eg, one haptic in and one out, one haptic in and both the optic and the other haptic out), surgical repositioning is required. One method is to viscodissect the bag open using a cannula or bent 30-gauge needle attached to a vial of dispersive OVD. Once the bag is open, a cannula or Sinskey hook can be used to gently rotate and center the IOL into the appropriate position.3,8
Anterior capsular contraction can cause the IOL to shift, creating astigmatism and a change in vision.2,3,8 With capsular phimosis and a small rhexis, the IOL can not only tilt, causing astigmatism, but can move posteriorly in the eye, inducing hyperopia. Management can include Nd:YAG laser lysis of the anterior capsular rim to release contracture, which may return the IOL to the correct position.2,8
If the decentered lens is in the sulcus and the capsular bag is compromised, the lens can be fixated to the iris or sclera. Multiple techniques to accomplish this goal have been described, including the commonly used modified Siepser9 and McCannel10 techniques. The glued IOL11 and the Yamane intrascleral haptic fixation12 (Figure 3) techniques are excellent options for scleral fixation and centration of three-piece IOLs.
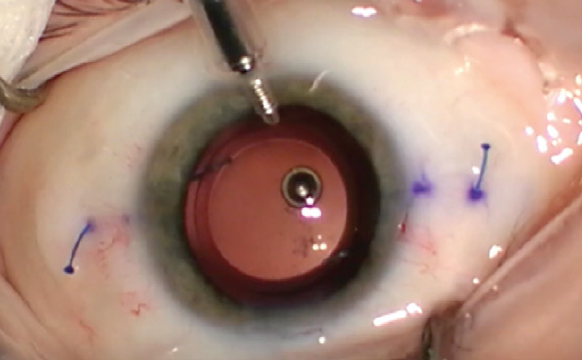
Figure 3. The Yamane technique obviates the need for glue or sutures.
Courtesy of Kamran Riaz, MD
All iris and scleral fixation methods require care. Because they involve haptic manipulation, it is important to consider the type of IOL and the length of time it has been in the eye. A haptic breaking during manipulation for iris or scleral fixation can turn a relatively straightforward procedure into a complex, much longer surgery and even risk loss of the IOL into the posterior segment, among other complications.
Finally, if there has been a complete dislocation of the IOL into the vitreous cavity or onto the retina, assistance from a retinal surgeon is necessary to bring the IOL anterior for fixation or removal. Depending on the type of IOL, it may have to be removed and exchanged. A one-piece IOL cannot be fixated to the sclera, nor should it be placed in the sulcus due to risk of uveitis-glaucoma-hyphema syndrome, so it must be swapped for a three-piece or anterior chamber IOL.
COMFORT ZONE
For any of the scenarios described here, the surgeon should use whichever technique he or she is comfortable with. If sufficient previous experience is lacking, it is helpful to review a novel technique in person with another experienced surgeon before attempting it oneself. Performing advanced, complex surgery with an experienced assistant is priceless, and his or her presence can help save the day.
Prevention is always the optimal strategy to address decentration. In high-risk patients—those with severe pseudoexfoliation, history of trauma, or phacodonesis, for example—the consent process must include extensive discussion of the risks of surgery. Two important risks to include are the possibility that it will be impossible to implant the chosen IOL and that late complications include dislocation and its sequelae. Often, patients are happy that their surgeon has spent the chair time discussing their unique conditions with them, and they will be highly satisfied with any lens decided upon.
1. Rosenberg E, Nattis A, Donnenfeld ED, Barsam A. Refractive intraocular lenses – managing unhappy patients. In: Hovanesian J, ed. Refractive Cataract Surgery: Best Practices and Advanced Technology. Thorofare, NJ: Slack; 2017: 215-224.
2. Schena LB. Centration – the heart of IOL success. EyeNet Magazine. January 2007.
3. Decentered multifocal IOLs may be cause of worsened vision. Ocular Surgery News. April 10, 2015.
4. Packer M, Fine IH, Hoffman RS, Piers PA. Improved functional vision with a modified prolate intraocular lens. J Cataract Refract Surg. 2004;30(5):986-992.
5. Caceres V. Dislocation and decentration remain common reasons for IOL removal. Ophthalmology Times. August 8, 2019.
6. Degenring RF, Sauder G. Vitreous prolapse and IOL dislocation during intravitreal injection of triamcinolone acetonide. Graefes Arch Clin Exp Ophthmol. 2006;244(8):1043-1044.
7. Jafarinasab M, Feizi S, Baghi A, Ziaie H, Yaseri M. Aspheric versus spherical posterior chamber intraocular lenses. J Ophthalmic Vis Res. 2010;5(4):217-222.
8. Stephenson M. How to manage dislocated IOLs. Review of Ophthalmology. October 14, 2018.
9. Osher RH, Snyder ME, Cionni RJ. Modification of the Siepser slip-knot technique. J Cataract Refract Surg. 2005;31(6):1098-1100.
10. Wald KJ, Paccione JC, Gross NE, et al. Successful management of posteriorly dislocated PC-IOLs with the twenty-five gauge vitrectomy and modified McCannel suture technique. Invest Ophthalmol Vis Sci. 2007;48(13):2207.
11. Narang P, Agarwal A. Glued intrascleral haptic fixation of an intraocular lens. Indian J Ophthalmol. 2017;65(12):1370-1380.
12. Yamane S, Sato S, Maruyama-Inoue M, Kadonosono K. Flanged intrascleral intraocular lens fixation with double-needle technique. Ophthalmology. 2017;124(8):1136-1142.







