Refractive surgery is booming. This is, in part, due to the negative effects of COVID-19 such as mask wear and increased computer use from working from home. In short, patients are motivated to improve their quality of life and now have the financial means to make their desires a reality. There is no doubt that refractive surgery is life-changing. It provides patients with the opportunity to achieve spectacle independence and a better quality of vision than they’ve ever experienced. With that said, it is important to understand the nuances of each procedure so that the right decision is made for every patient.
I have extensive experience with many refractive surgery procedures, including LASIK, PRK, phakic IOLs, and lenticule extraction. All have their place depending on patients’ needs and ocular anatomy, and we shouldn’t discredit one procedure over the other.
One important consideration when selecting the right procedure for patients is postoperative spherical aberration. The literature has shown that flap-based procedures are associated with an increase in corneal aberrations after surgery.1,2 Testing with low-contrast charts and with dilated pupils indicates that the overall image quality can degrade in response to the increased aberrations in the optical system.3,4 Corneal aberrations do not, however, describe the overall optical quality of the eye. This is for several reasons. First, the optical changes that are induced by refractive surgery occur on the cornea. Second, other parameters including lens position, lens thickness, the refractive index, axial length, and pupil centration are integral components of image formation. Nevertheless, the presence of excessive spherical aberration, for some patients, can lead to poor visual performance.
spherical aberrations
Lenticule extraction procedures induce only a minimal increase in spherical aberrations after surgery.5 So far what we have found with Corneal Lenticule Extraction for Advanced Refractive Correction (CLEAR) using the FEMTO LDV Z8 (Ziemer) is that there has been little change in the spherical aberration. More importantly, there has been minimal induction or change in vertical coma after surgery, which can result when the treatment is incorrectly centered on the visual axis. In my experience, it is easier to accomplish treatment centration with CLEAR. This device allows me to place the creation of the lenticule directly on the visual axis or the central corneal light reflex rather than on the center of the pupil, which is routine with other lasers.
Centration of the CLEAR treatment is simple. It is performed after the patient interface of the Z8 is docked. The lenticule is automatically centered, but it can be adjusted if needed. Perfect centration results in a smaller risk for the induction of vertical coma. Furthermore, the lenticule is rotated automatically, according to the marking of the horizontal axis, for cyclotorsion control. There is no risk of suction loss.
The ability to rotate the lenticule to achieve adequate torsional control gives me more confidence in the CLEAR procedure. The Z8 automatically detects the corneal markings. In the very near future, the centration and rotation will be automated with a link to the GALILEI (Ziemer) diagnostic system.
CLEAR ADVANTAGES
Lenticule extraction procedures require a very different skill set to LASIK. Surgeons must recognize the shape and the demarcation line of the lenticule, they must understand whether they are above or below the lenticule, and of course they must learn how to perform surgery through a keyhole incision. The CLEAR procedure boasts several advantages over other lenticule extraction procedures.
Guiding tunnels. One advantage of the CLEAR procedure is that two small tunnels can be created to guide the instruments into the correct lenticule plane. As an extended part of the incisions, one tunnel guides you to the anterior surface, and the other tunnel guides you to the posterior surface (Figure). The tunnel size can be customized to as small as 1.5 mm, and the position of the tunnel can be customized depending on the surgeon’s hand dominance. I typically create the first guiding tunnel at about the 11 o’clock position and the second at about the 1 or 2 o’clock position. The use of guiding tunnels also decreases the intrastromal manipulations and therefore reduces the amount of inflammation in the eye.
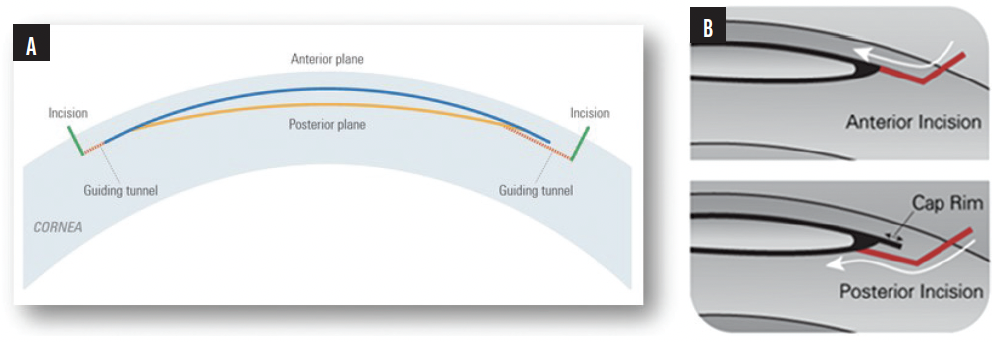
Figure. Schematics of the guiding tunnels to the anterior and posterior surfaces (A, B).
The use of two tunnels compared to one can also shorten the learning curve. I find that I no longer need to create two tunnels due to my level of experience with CLEAR; however, my colleagues sometimes still create two tunnels because the second acts as a safety net in case they run into problems during surgery.
Intraoperative OCT shows the positioning of the guiding tunnels in relation to the lenticule creation.
Less risk for formation of an opaque bubble layer. The venting tunnels of the CLEAR trajectory are created in such a way that gas can escape during the creation of the lenticule. This reduces the risk for an opaque bubble layer (OBL) to form. OBL can affect the dissection and therefore refractive outcome as well.6
A thinner lenticule. The shape of the CLEAR lenticule is slightly thinner at the edge compared to the lenticules that are created by other laser platforms. Because there is no sidecut, it is easy to navigate into the posterior plane to complete the dissection. If there is any problem, the guiding tunnels can be useful to determine which plane you are in.
Ease of centration. Centering the lenticule with the Z8 is easy. The eye is marked at 0º and 180º on the horizontal axis, followed by the center of the corneal light reflex. The latter is determined by where the patient fixates at the slit lamp.
I also check angle kappa with the Orbscan (Bausch + Lomb). If the angle kappa is large, the lenticule is decentered about 300 µm away from the center of the pupil toward the angle kappa intercept. If it is small, then the lenticule is centered on the visual axis.
After centration, cyclotorsion control is performed by automatically rotating the lenticule into position. The Z8’s software has a built-in feature to warn the surgeon if the lenticule is decentered or too close to the applanation zone.
In the near future, an exciting development should be available—automated centration and cyclotorsion. This will be achieved by linking the Z8 femtosecond laser to the GALILEI diagnostic system (Ziemer), allowing full automation of two of the key steps of the CLEAR procedure.
SURGERY ON A COLLEAGUE
In my early experience with CLEAR, the results have been encouraging. Here, I share one example of a case in which CLEAR was the best choice for my patient. It also highlights benefits of the CLEAR procedure in terms of inducing minimal spherical aberration and vertical coma.
In one of our ongoing clinical studies, one of my staff members participated in the study and is thrilled with her results in the CLEAR eye. She had moderate myopia and large eyes and presented with dry eye disease. She was prescribed omega-3 fatty acids, lubricants, and topical cyclosporine for 6 weeks before surgery. She also received punctal plugs to optimize the ocular surface before surgery. For any lenticule extraction procedure, it is crucial to make sure the cornea is well hydrated before surgery. Surgery was uncomplicated. At 3 months postoperative, her UDVA was 20/20+ OU. (To read the patient’s perspective, see the article on page 8.)
CONCLUSION
CLEAR has many advantages over other lenticule extraction procedures. For those who are just starting with the procedure, the use of two guiding tunnels to help ensure the instruments are in the correct plane is helpful, increasing the accuracy and ease of the procedure.
1. Oshika T, Klyce SD, Applegate RA, Howland HC, El Danasoury MA. Comparison of corneal wavefront aberrations after photorefractive keratectomy and laser in situ keratomileusis. Am J Ophthalmol. 1999;127:1–7.
2. Schwiegerling J, Snyder RW. Corneal ablation patters to correct for spherical aberration in photorefractive keratectomy. J Cataract Refract Surg. 2000;26:214–221.
3. Holladay JT, Dudeja DR, Chang J. Functional vision and corneal changes after laser in situ keratomileusis determined by contrast sensitivity, glare testing and corneal topography. J Cataract Refract Surg. 1999;25:663–669.
4. Verdon W, Bullimore M, Maloney RK. Visual performance after photorefractive keratectomy. A prospective study. Arch Ophthalmol. 1996;114:1465–1472.
5. Miraftab M, Hashemi H, Aghamirsalim M, Fayyaz S, Asgari S. Matched comparison of corneal higher order aberrations induced by SMILE to femtosecond assisted LASIK and to PRK in correcting moderate and high myopia: 3.00mm vs. 6.00mm. BMC Ophthalmol. 2021;21(1):216.
6. Son G, Lee J, Jang C, Choi KY, Cho BJ, Lim TH. Possible risk factors and clinical effects of opaque bubble layer in small incision lenticule extraction (SMILE). J Refract Surg. 2017;33(1):24-29.


