Since the introduction of refractive lenticule extraction for the correction of myopia and myopic astigmatism in 2011,1,2 significant improvements have been made on the surgical and technological aspects of the procedure. Lenticule extraction has gained significant momentum, claiming its place as a mainstream refractive surgery procedure. Currently, the lenticule extraction landscape is being shaped by four different manufacturers that are developing dedicated femtosecond laser modules for the procedure. Corneal lenticule extraction for advanced refractive correction (CLEAR) is a new form of lenticule extraction performed with the Z8 femtosecond laser systems (Ziemer).
THE LASER
FEMTO LDV Z8 is a low-energy laser—working at the nanojoule level—with a solid track record of performing smooth flaps for femtosecond LASIK (femto-LASIK), customized tunnels for intrastromal corneal ring segments, deep anterior lamellar keratoplasty, Descemet-stripping endothelial keratoplasty, penetrating keratoplasty (PKP), arcuate keratectomy, and femtosecond laser-assisted cataract surgery (femto-phaco). The Z8’s intraoperative OCT ensures safety while performing the aforementioned procedures and allows the surgeon to view real-time corneal applanation that is generated by the system. Femto-phaco and PKP, however, use a liquid interface system with zero corneal compression to avoid mechanical deformation.
The CLEAR module of the Z8 builds on the advantages of the laser’s low energy source, ensuring smooth lenticule dissection.3 Additionally, the laser can center the treatment on the corneal vertex and adjust for cyclotorsion, all intraoperatively, thanks to the device’s corneal applanation feature. We have been using the Z8 for more than 4 years and have been performing CLEAR procedures for about 5 months at the time of this writing with great outcomes.
STUDY DESIGN
Patient population and examinations. This article reviews our 1- and 3-month retrospective data of the first 46 eyes treated with CLEAR. All eyes had myopia or myopic astigmatism and a preoperative corrected distance visual acuity (CDVA) of 20/25 or better. The mean age of patients was 26 ±5.94 years (range, 18–43 years) and the mean preoperative manifest refraction spherical equivalent (MRSE) and manifest cylinder were -3.78 ±1.53 D and -0.62 ±0.55 D, respectively. The planned optical zone was 6.5 ±0.13 mm. The complete preoperative data is represented in the Table.
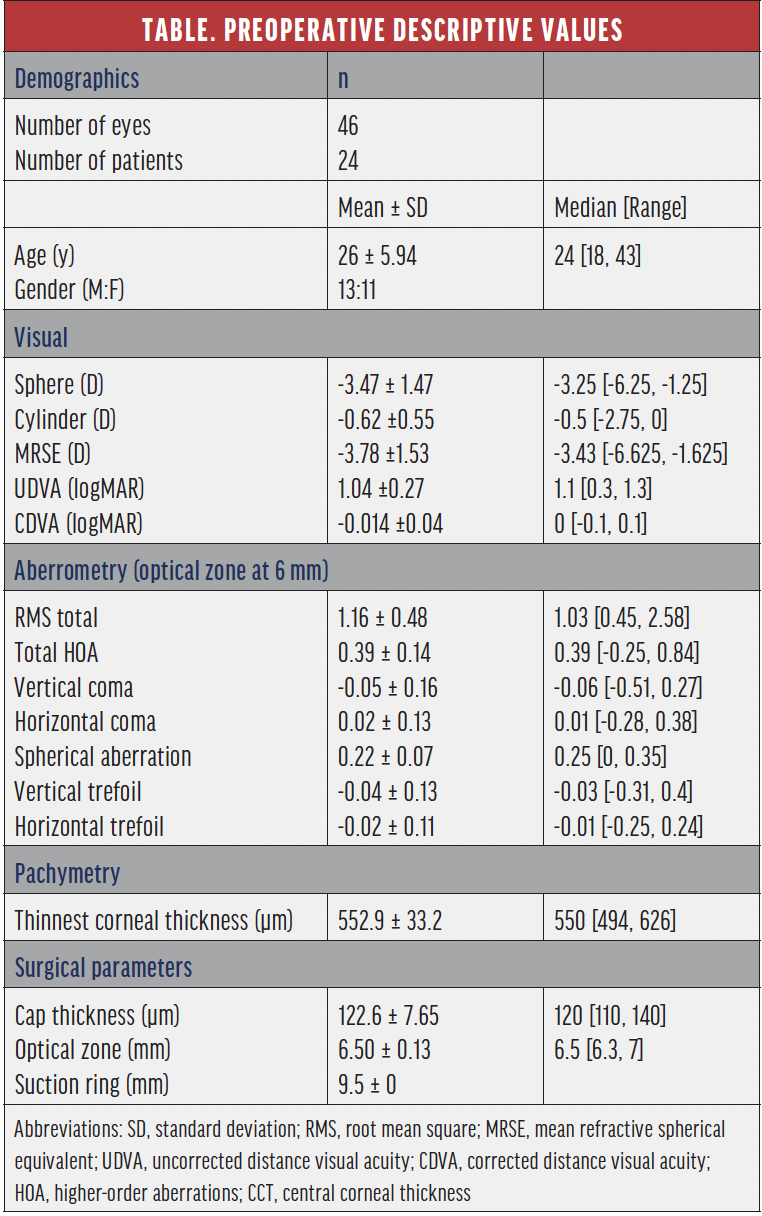
Uncorrected distance visual acuity (UDVA), corrected distance visual acuity (CDVA), objective and subjective refraction, topography, tomography, ocular aberrometry, and biomicroscopic examination were performed on all patients preoperatively and postoperatively at 1 day, 1 week, and 1 and 3 months.
Surgical technique. The procedure was performed under topical anesthesia with standard prepping and draping. Limbal markings were initially done at the slit lamp (Figures 1 and 2A) and were checked for accuracy using the toriCAM smartphone application (Graham Barrett) on iOS devices. Consequently, the limbal markings were extended under the microscope using a three-pronged instrument that acted as a crosshair (Awwad 3-Blade Marker for CLEAR, Epsilon Ophthalmic Surgical Instruments; Figures 2B and 2C). The instrument’s corneal markings were centered on the fixation light reflex, approximating the corneal vertex. Extension of preoperative markings onto the cornea allows them to be recognized by the automatic centration and cyclotorsion system of the Z8 (Figure 2D). Centration and cyclotorsion can be further readjusted manually by the surgeon if needed.
Figure 1. Limbal markings performed at the slit lamp.
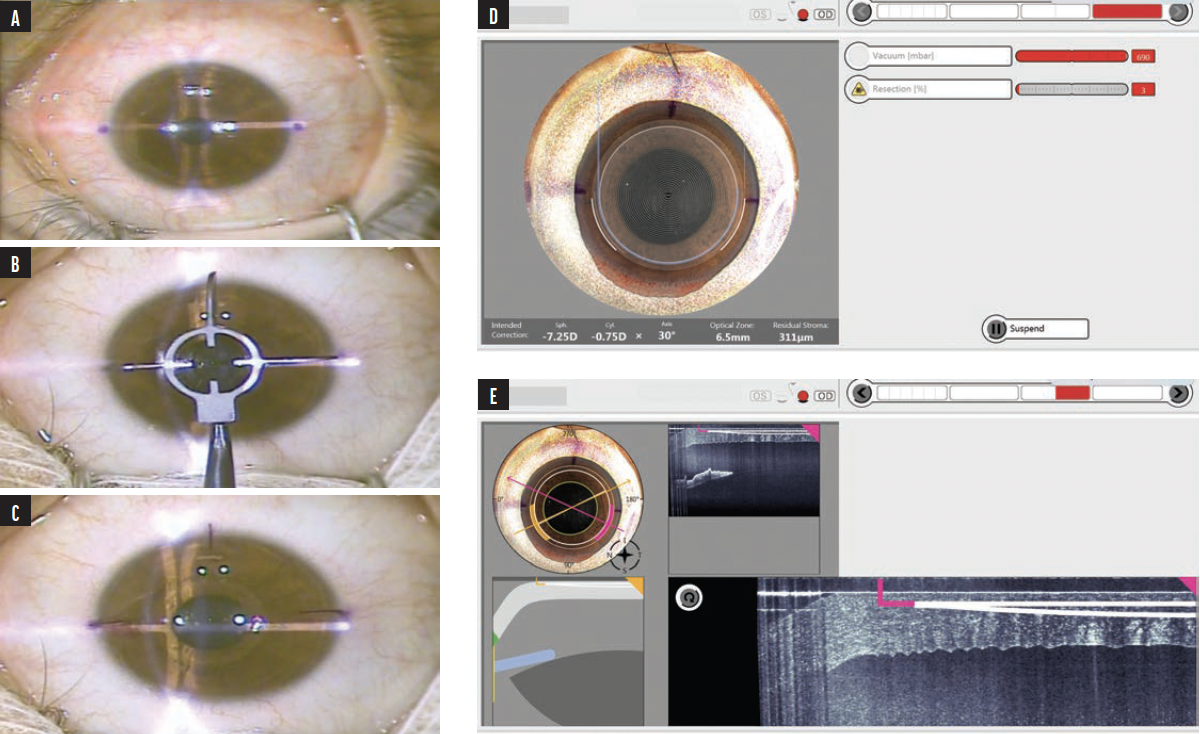
Figure 2. Intraoperative video snapshots. Two limbal dots are marked by gentian violet on the slit lamp (A). A three-pronged instrument is centered on the fixation light (ie, visual axis) and is meant to extend the two marking dots into the cornea (B). The three marked lines act as a crosshair on the light reflex, approximating the corneal vertex (C). Image from the Z8 display screen after eye docking. The laser software automatically recognizes the three marked lines and rotates and centers the treatment accordingly. There is room for the surgeon to recenter and control the location should the surgeon need further alignment (D). Image from the Z8 display screen showing an intraoperative OCT (E).
At the time of this writing, Ziemer presented a software patch on the Galilei that was fed to the Z8, allowing it to automatically detect the corneal vertex and compensate for cyclotorsion without any markings. The CLEAR software also allows adjustments to the number and size of the incisions as well as programming target refraction, optical zone, and energy settings.
STUDY OUTCOMES
Efficacy. At 1 month postoperatively, 93% of eyes had an UDVA of 20/20 or better, and 97% had an MRSE within ±0.50 D of the intended target (Figure 3A). At 3 months, 96% of eyes had an UDVA of 20/20 or better, and 100% had an MRSE within ±0.50 D (Figure 4). Figures 3B and 3C show the change in corrected distance visual acuity and attempted versus achieved spherical equivalent, respectively. Manifest cylinder changed from -0.63 ±0.56 D preoperatively to -0.16 ±0.26 D at 1 month (P < .001) and -0.2 ±0.30 D at 3 months (P = .049) postoperatively (Figure 3E). The correlation of attempted and achieved MRSE indicated good accuracy over the whole range of planned treatments with no obvious under- or overcorrection (Figure 3C). Figure 3F shows the stability of the spherical equivalent refraction.
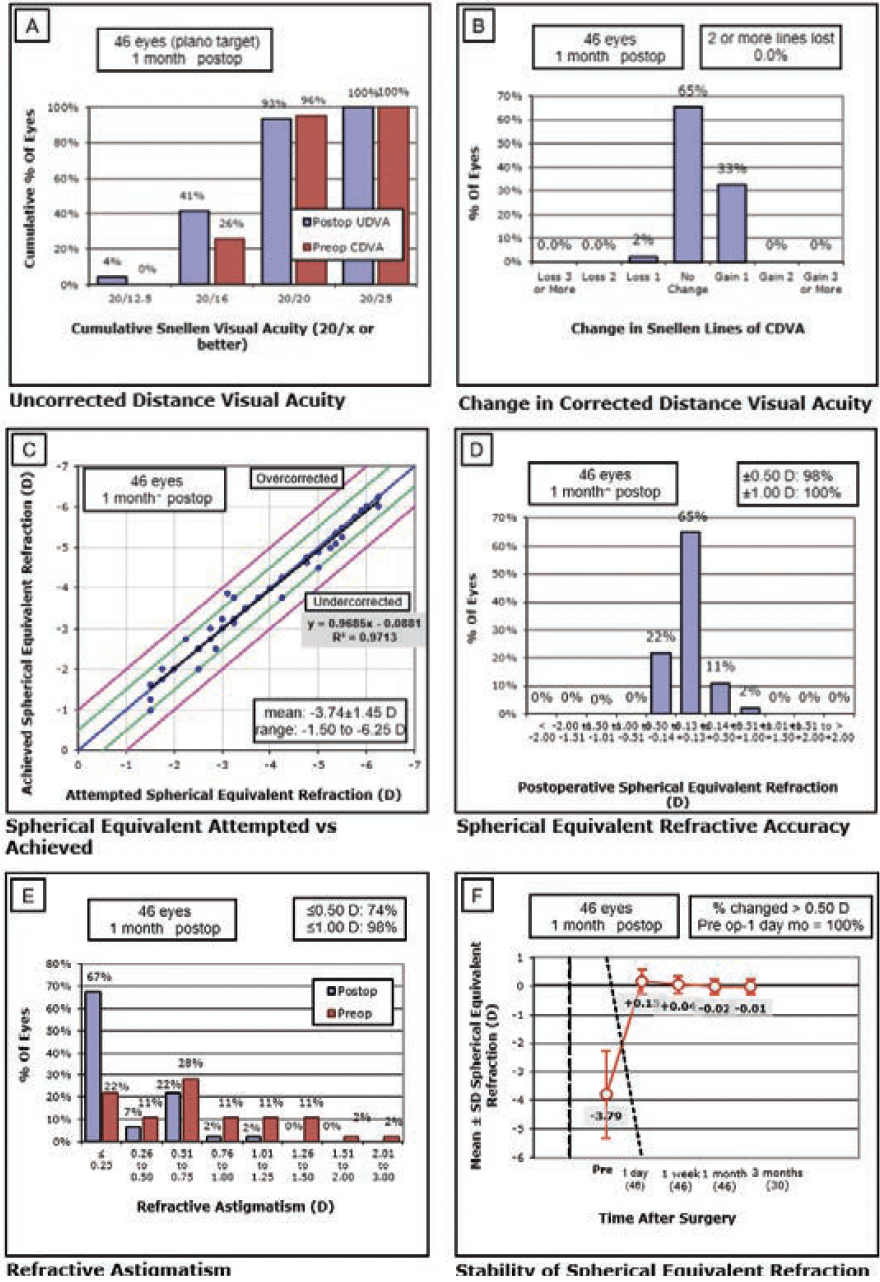
Figure 3. Six standard graphs for reporting refractive surgery showing the visual and refractive outcomes for 46 eyes treated with CLEAR.
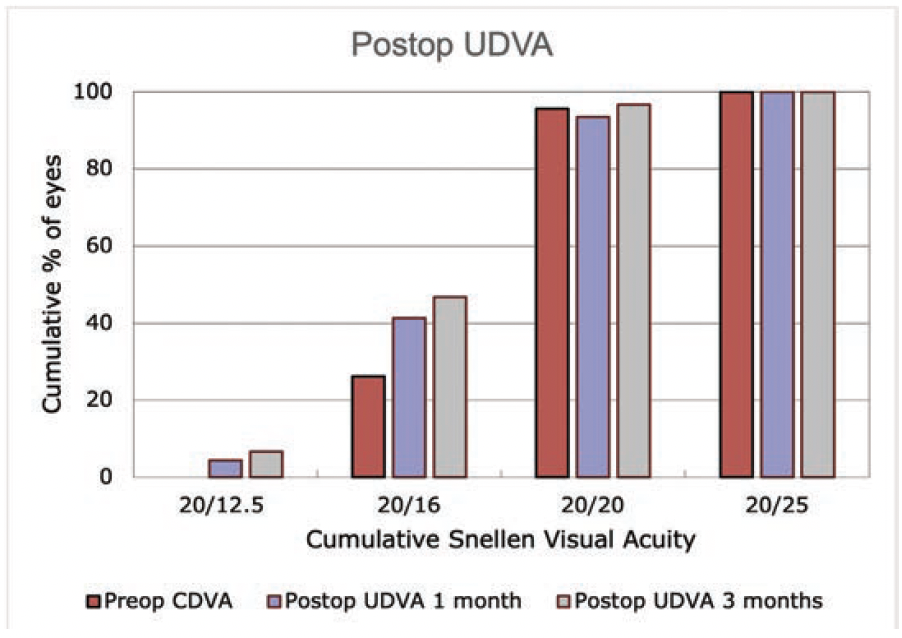
Figure 4. Postoperative UDVA at 1 and 3 months.
Aberrometry. Changes in total corneal aberrometry at a 6-mm optical zone are shown in Figure 5. Spherical aberration was almost unchanged from 0.22 ±0.07 µm preoperatively to 0.24 ±0.18 µm at 3 months postoperative (P = .013). Vertical coma also remained relatively stable while horizontal coma slightly increased from 0.03±0.13 µm to 0.13±0.19 µm after 1 month (P = .003). Total higher-order aberrations (HOAs) increased at 1 day but then decreased at 1 week and remained stable at 3 months.
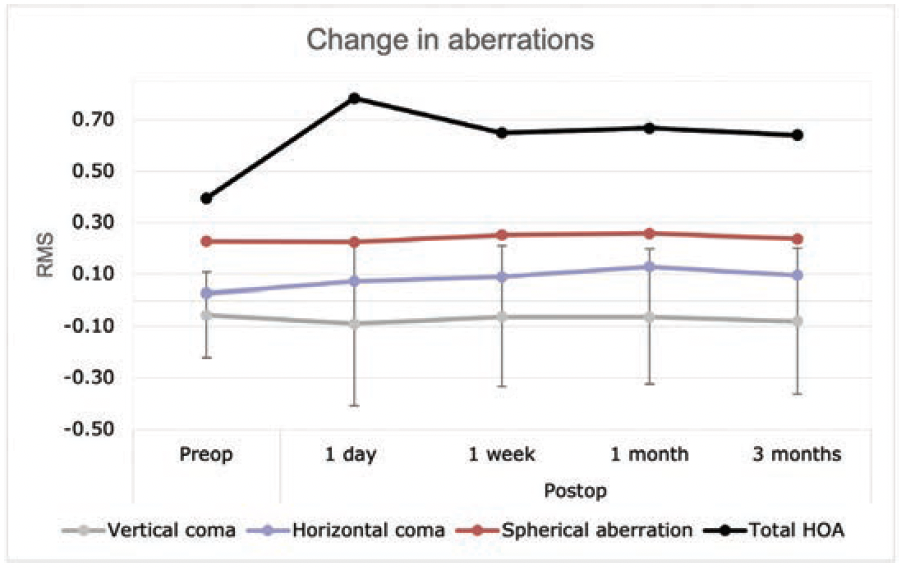
Figure 5. Total corneal HOA changes at different postoperative intervals.
Stability. As expected for lenticule extraction procedures, patients had a relatively slower visual recovery than what is customary for femto-LASIK, especially on day 1. However, visual results significantly improved over time. The percentage of eyes achieving an UDVA of 20/20 or better was 52.2% on day 1, 76.1% at 1 week, 93% at 1 month, and 96% at 3 months postoperatively. Mean MRSE changed from -3.79 ±1.53 D preoperatively to 0.04 ±0.29 D at 1 week postoperatively (P < .001). At 1 and 3 months, respectively, it further improved to -0.02 ±0.251 D (P < .001) and -0.01 ±0.26 D (P < .001).
Safety. Only one eye (3.3%) lost one line of CDVA at 3 months postoperatively. No patient experienced a loss of 2 lines or more (Figure 6). For CDVA, at 1 month, 33% gained 1 line and 65% experienced no change (Figure 3B). No aborted cases, no suction loss, and no cases of lenticule tears were encountered.
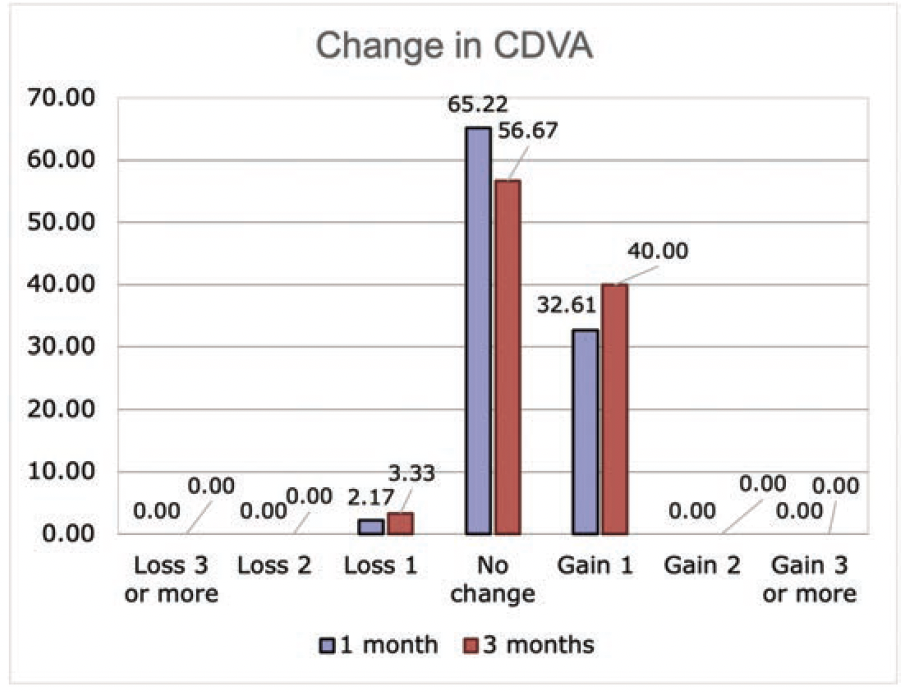
Figure 6. Lines of CDVA gained or lost at 1 and 3 months postoperatively.
DISCUSSION
Given that the sample study was the first series of CLEAR procedures performed by one of us (STA), there was a significant improvement in UDVA, MRSE, and refractive astigmatism and an excellent aberrometry profile postoperatively. The mild increase in coma mirrors the results of other studies4,5 and is possibly due to manual centration. This can be mitigated currently by using a large optical zone. In the near future, the Z8 will provide automated centration and cyclotorsion technique.
We believe the reason for the minimal induction of coma was the adoption of the three-pronged marker to aid in accurate treatment centration and cyclotorsion correction. The minimal induction of spherical aberration underlines the large functional optical zones achieved.
Previous studies of the aberrations after femto-LASIK and corneal wavefront-guided PRK have shown that HOAs, notably spherical aberration, increase postoperatively.6 This pattern, however, has been shown to be better with lenticule extraction procedures.7-12 Maintaining a large functional optical zone can therefore curb the increase in HOAs and spherical aberration postoperatively.
The relatively slower recovery observed after lenticule extraction compared to flap-based procedures can be in part attributed to the undulations in the Bowman layer created by lenticule dissection and the time needed for flattening and repositioning of the cap until visual results are achieved. Microdistortions in the Bowman layer have been reported in various procedures of lenticule extraction.13 The low energy profile of the Z8 is key to providing the optimal visual recovery.
CONCLUSION
CLEAR is a great lenticule extraction procedure that offers safe, efficient, and stable results. The results reported in this article represent our initial experience with the first series of eyes. We are confident that further refinement of the energy profiles and additional surgeon experience will enhance the procedure’s results.
CLEAR induces less spherical aberration. This is ideal in eyes with moderate to high myopia. The procedure also ensures the integrity of the nerve plexus, which is important to preserve the ocular surface while having less biomechanical impact on the cornea compared to flap-based procedures.
The FEMTO LDV Z8 is CE marked and FDA cleared. Lenticule extraction application is CE marked but not yet FDA cleared for use in the United States. For other countries, availability may be restricted due to regulatory requirements. Please contact Ziemer for details.
1. Sekundo W, Kunert KS, Blum M. Small incision corneal refractive surgery using the small incision lenticule extraction (SMILE) procedure for the correction of myopia and myopic astigmatism: results of a 6 month prospective study. Br J Ophthalmol. 2011;95(3):335-339.
2. Shah R, Shah S, Sengupta S. Results of small incision lenticule extraction: all-in-one femtosecond laser refractive surgery. J Cataract Refract Surg. 2011;37(1):127-137.
3. Izquierdo LJ, Sossa D, Ben-Shaul O, Henriquez MA. Corneal lenticule extraction assisted by a low-energy femtosecond laser. J Cataract Refract Surg. 2020;46(9):1217-1221.
4. Sekundo W, Gertnere J, Bertelmann T, Solomatin I. One-year refractive results, contrast sensitivity, high-order aberrations and complications after myopic small-incision lenticule extraction (ReLEx SMILE). Graefes Arch Clin Exp Ophthalmol. 2014;252(5):837-843.
5. Reinstein DZ, Archer TJ, Vida RS, Carp GI, Reinstein JFR, McAlinden C. Objective and subjective quality of vision after SMILE for high myopia and astigmatism. J Refract Surg. 2022;38(7):404-413.
6. Reinstein DZ, Carp GI, Archer TJ, et al. Long-term visual and refractive outcomes after LASIK for high myopia and astigmatism from -8.00 to -14.25 D. J Refract Surg. 2016;32(5):290-297.
7. Gertnere J, Solomatin I, Sekundo W. Refractive lenticule extraction (ReLEx flex) and wavefront-optimized femto-LASIK: comparison of contrast sensitivity and high-order aberrations at 1 year. Graefes Arch Clin Exp Ophthalmol. 2013;251(5):1437-1442.
8. Lin F, Xu Y, Yang Y. Comparison of the visual results after SMILE and femtosecond laser-assisted LASIK for myopia. J Refract Surg. 2014;30(4):248-254.
9. Ganesh S, Gupta R. Comparison of visual and refractive outcomes following femtosecond laser-assisted LASIK with SMILE in patients with myopia or myopic astigmatism. J Refract Surg. 2014;30(9):590-596.
10. Gyldenkerne A, Ivarsen A, Hjortdal JØ. Comparison of corneal shape changes and aberrations induced by FS-LASIK and SMILE for myopia. J Refract Surg. 2015;31(4):223-229.
11. Liu M, Chen Y, Wang D, et al. Clinical outcomes after SMILE and femtosecond laser-assisted LASIK for myopia and myopic astigmatism: a prospective randomized comparative study. Cornea. 2016;35(2):210-216.
12. Lee H, Yong Kang DS, Reinstein DZ, et al. Comparing corneal higher-order aberrations in corneal wavefront-guided transepithelial photorefractive keratectomy versus small-incision lenticule extraction. J Cataract Refract Surg. 2018;44(6):725-733.
13. Shroff R, Francis M, Pahuja N, Veeboy L, Shetty R, Sinha Roy A. Quantitative evaluation of microdistortions in Bowman’s layer and corneal deformation after small incision lenticule extraction. Trans Vis Sci Tech. 2016;5(5):12.



