Over the past decade or more, the landscape of glaucoma management has changed profoundly. One of the biggest changes was the introduction of less invasive and traumatic procedures. The idea was that MIGS options could provide a procedural solution to the management of glaucoma at the milder end of the disease spectrum—before more invasive procedures such as trabeculectomy were indicated. The first of these devices was the iStent® (Glaukos), and it was clear from the randomized controlled studies carried out more than 10 years ago that the device had the ability to reduce both the IOP and the medication burden.1 What was less clear at the time was whether the device would have an effect on structural or functional endpoints, which is of course the ultimate goal in treating glaucoma.
A NEEDED SHIFT IN FOCUS
While IOP is an important factor in glaucoma management, several groups advise a shift in the focus of glaucoma clinical trials toward biomarkers that better represent disease stability, such as structural, functional, and composite endpoints.2 iStent technologies have well-documented IOP-reducing potential and favorable safety profiles, but no study concluded on their effect on the rates of functional progression in glaucoma.
In the first systematic review of the effect of iStent technologies on functional glaucoma progression, 15 studies were analyzed, encompassing a total of 1,115 eyes with a mean follow-up of 37.9 months (range, 12-96 months; personal data under review). At the end of the follow-up period, a weighted mean 26.6% IOP reduction was achieved (range, 15.2% to 42.3%).
iSTENT TECHNOLOGIES AND GLAUCOMA STABILITY
The rate of visual field progression over the entire cohort was -0.02 ±0.34 dBs per year, ranging from -0.01 ±0.42 dBs per year in combined procedures to -0.07 ±0.62 dBs per year in standalone iStent device implantations. To put these data into context, if a blind eye is considered to have a mean deviation between -25 and -30 dBs, it would take a healthy eye more than 1,000 years to become blind at this rate of progression. In comparison, large international cohorts of medically and surgically treated glaucoma exhibited mean progression rates ranging from -0.22 to -0.67 dBs per year (Figure).3-5
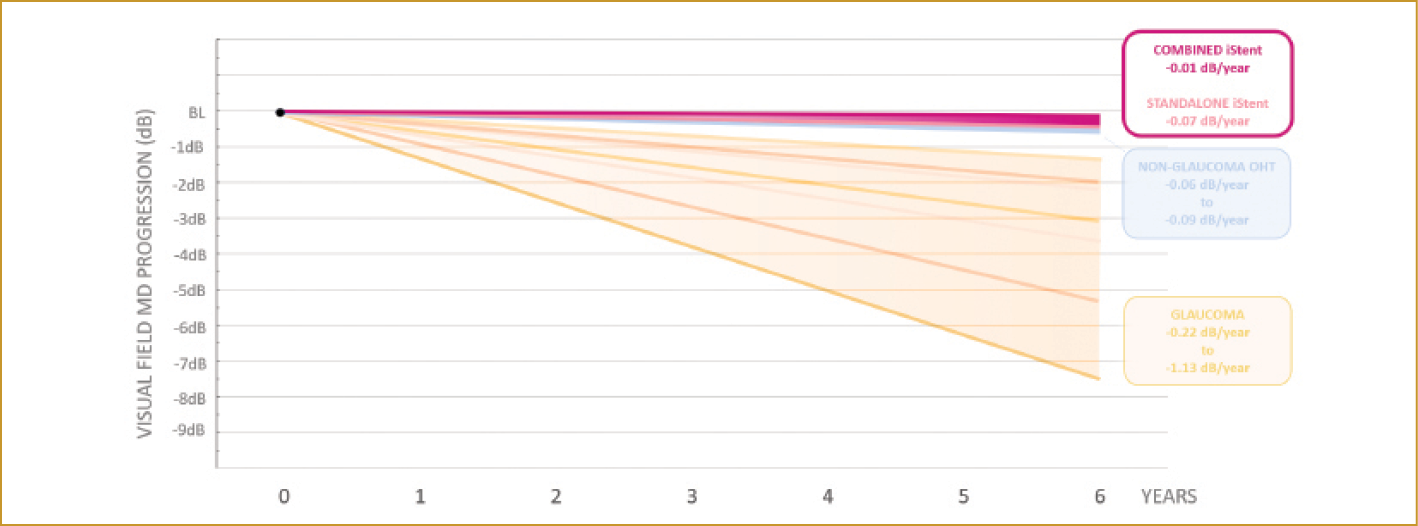
Figure. Observed visual field mean deviation progression following standalone and combined iStent technologies implantation (pink lines) compared to the rates of progression reported in the literature for ocular hypertension (blue lines) and treated glaucoma (yellow lines). Abbreviations: BL, baseline; MD, mean deviation; OHT, ocular hypertension.
CONCLUSION
iStent technologies have well-documented IOP-reducing potential and favorable safety profiles. In this meta-analysis of 1,115 eyes, the device achieved a mean rate of progression of -0.02 dBs per year with serial standard automated perimetry, which is similar to that reported in non-glaucomatous eyes and slower than that reported in medically treated glaucoma. While specifically designed and powered trials would be useful to confirm these results, the present findings suggest that early trabecular bypass surgery may be beneficial in stabilizing glaucoma progression.
1. Healey PR, Clement CI, Kerr NM, Tilden D, Aghajanian L. Standalone iStent trabecular micro-bypass glaucoma surgery: a systematic review and meta-analysis. J Glaucoma. 2021;30(7):606-620.
2. Weinreb RN, Ramulu P, Topouzis F, Park KH, Mansouri K, Lerner F. 2019. 11th Consensus Meeting: Glaucoma Surgery.
3. Heijl A, Bengtsson B, Hyman L, Leske MC. Natural History of Open-Angle Glaucoma. Ophthalmology. 2009;116(12):2271-6.
4. Heijl A, Buchholz P, Norrgren G, Bengtsson B. Rates of visual field progression in clinical glaucoma care. Acta Ophthalmologica. 2013;91(5):406-12.
5. Aptel F, Aryal-Charles N, Giraud JM, El Chehab H, Delbarre M, Chiquet C, et al. Progression of visual field in patients with primary open-angle glaucoma – ProgF study 1. Acta Ophthalmol. 2015;93(8):e615-20.
INDICATION FOR USE: The iStent inject® W, is intended to reduce intraocular pressure safely and effectively in patients diagnosed with primary open-angle glaucoma, pseudo-exfoliative glaucoma or pigmentary glaucoma. The iStent inject® W, can deliver two (2) stents on a single pass, through a single incision. The implant is designed to stent open a passage through the trabecular meshwork to allow for an increase in the facility of outflow and a subsequent reduction in intraocular pressure. The device is safe and effective when implanted in combination with cataract surgery in those subjects who require intraocular pressure reduction and/or would benefit from glaucoma medication reduction. The device may also be implanted in patients who continue to have elevated intraocular pressure despite prior treatment with glaucoma medications and conventional glaucoma surgery. CONTRAINDICATIONS: The iStent inject® W System is contraindicated under the following circumstances or conditions: • In eyes with primary angle closure glaucoma, or secondary angle-closure glaucoma, including neovascular glaucoma, because the device would not be expected to work in such situations. • In patients with retrobulbar tumor, thyroid eye disease, Sturge-Weber Syndrome or any other type of condition that may cause elevated episcleral venous pressure. WARNINGS/PRECAUTIONS: • For prescription use only. • This device has not been studied in patients with uveitic glaucoma. • Do not use the device if the Tyvek® lid has been opened or the packaging appears damaged. In such cases, the sterility of the device may be compromised. • Due to the sharpness of certain injector components (i.e. the insertion sleeve and trocar), care should be exercised to grasp the injector body. Dispose of device in a sharps container. • iStent inject® W is MR-Conditional; see MRI Information below. • Physician training is required prior to use of the iStent inject® W System. • Do not re-use the stent(s) or injector, as this may result in infection and/or intraocular inflammation, as well as occurrence of potential postoperative adverse events as shown below under “Potential Complications.” • There are no known compatibility issues with the iStent inject® W and other intraoperative devices. (e.g., viscoelastics) or glaucoma medications. • Unused product & packaging may be disposed of in accordance with facility procedures. Implanted medical devices and contaminated products must be disposed of as medical waste. • The surgeon should monitor the patient postoperatively for proper maintenance of intraocular pressure. If intraocular pressure is not adequately maintained after surgery, the surgeon should consider an appropriate treatment regimen to reduce intraocular pressure. • Patients should be informed that placement of the stents, without concomitant cataract surgery in phakic patients, can enhance the formation or progression of cataract. ADVERSE EVENTS: Please refer to Directions For Use for additional adverse event information. CAUTION: Please reference the Directions For Use labelling for a complete list of contraindications, warnings and adverse events.
© 2024 Glaukos Corporation. Glaukos, iStent®, iStent inject® and iStent inject® W are registered trademarks of Glaukos Corporation. PM-EU-0284

