There are several myths surrounding the safety and effectiveness of trabecular micro-bypass procedures. There is also, however, evidence supporting the benefits of these devices, especially as an early intervention strategy for mild to moderate glaucoma. This article challenges common misconceptions and further establishes the need for devices such as the iStent inject® W (Glaukos).
FIVE COMMON MYTHS
Myth No. 1: iStent inject W is minimally effective. A pivotal randomized controlled trial of 505 patients dispelled doubts about the device’s efficacy.1 Patients were randomized to undergo cataract surgery alone or in combination with the iStent inject®. All endpoints were met, including at least a 20% reduction in unmedicated diurnal IOP and a mean reduction in unmedicated diurnal IOP (Figure 1). More than 75% of patients in the iStent® group were medication-free and attained the primary endpoint of more than 20% reduction in any medicated IOP. The mean IOP reduction from baseline was 7 mm Hg, which was a statistically significant difference versus the cataract surgery only group. Additionally, the IOP was consistent across all time points (6, 12, and 24 months) without additional medication. When performed as a standalone procedure, iStent can decrease IOP by up to 8 mm Hg.2

Figure 1. IOP reduction was sustained through 24 months.
A study by Ahmed et al showed the iStent inject had long-term safety,3 and sustained effectiveness has been shown for up to 7 years.1,2,4
Myth No. 2: Patients do fine with eye drops. The two biggest challenges in preventing disease progression are late presentation and patient noncompliance with topical medication.5-10 According to Market Scope, worldwide glaucoma medical treatment is about 40 times more common than surgical treatment with MIGS. Yet glaucoma patients on prolonged medication often experience side effects such as inflammation and ocular surface disease. Quality of life is a critical factor in the decision-making process. The iStent inject is the first MIGS device to show significant durable Vision Related Quality of Life improvements from a pivotal trial by reducing ocular pain and improving general vision.1
Myth No. 3: iStent inject W doesn’t work when the baseline IOP is elevated. A post-hoc analysis of Samuelson et al’s pivotal trial demonstrated the device’s effectiveness of iStent inject even in patients with elevated baseline IOP exceeding 30 mm Hg11 with a mean diurnal IOP reduction of 9.8 mm Hg. In the study, patients were divided into three groups, those with low (< 25 mm Hg), mid (25 to 30 mm Hg), and high baseline (> 30 mm Hg) diurnal IOPs after washout (Figure 2).
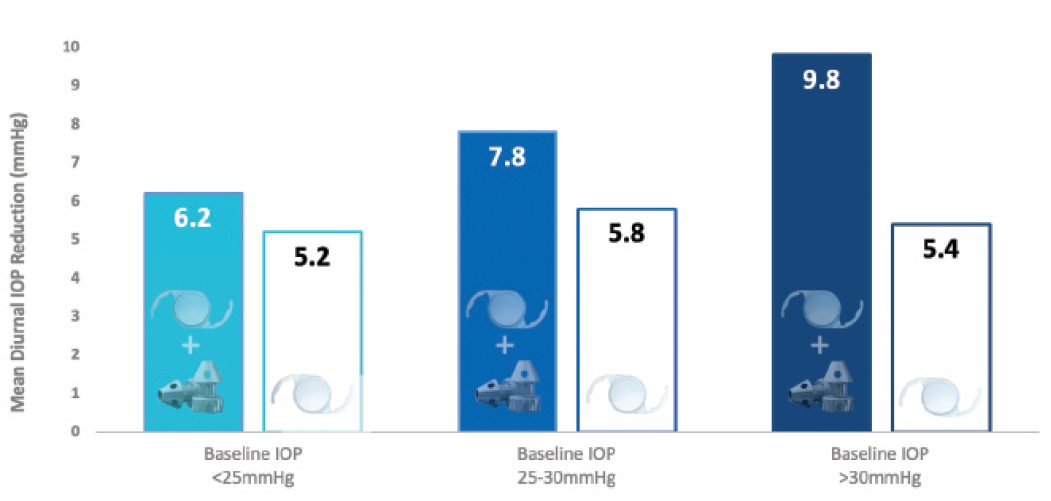
Figure 2. Pivotal data outcomes stratified by baseline IOP.
Myth No. 4: iStent inject W is not safe or well known. In the more than 300 published papers on iStent technologies with up to 8 years of follow-up, more than 20,000 eyes studied, and more than 1 million implanted devices worldwide, there have been no serious adverse events such as myopic shift, flat anterior chamber, choroidal hemorrhage, and corneal decompensation.1 Endothelial cell loss, a significant concern in other glaucoma surgeries, is with iStent inject nearly similar to phacoemulsification (6.3% and 9.4%, respectively, after 5 years).3
CONCLUSION
From its proven safety and efficacy in diverse patient populations to its ability to enhance quality of life, iStent inject® W offers many advantages to glaucoma patients and can be considered early in the disease state.
1. Samuelson TW, Singh IP, Williamson BK, et al. Quality of life in primary open-angle glaucoma and cataract: an analysis of VFQ-25 and OSDI from the iStent inject® pivotal trial. Am J Ophthalmol. 2021;229:220-229.
2. Healey PR, Clement CI, Kerr NM, Tilden D, Aghajanian. Standalone iStent trabecular micro-bypass glaucoma surgery: a systematic review and meta-analysis. J Glaucoma 2021;30(7):606-620.
3. Ahmed IIK, Sheybani A, De Francesco T, et al. Long-term endothelial safety profile with iStent inject in patients with open-angle glaucoma. Am J Ophthalmol. 2023;252:17-25.
4. Ziaei H, Au L. Manchester iStent study: long-term 7-year outcomes. Eye (London). 2021;35(8):2277-2282.
5. Boodhna, Trishal, and David P. Crabb. “Disease severity in newly diagnosed glaucoma patients with visual field loss: trends from more than a decade of data.” Ophthalmic and Physiological Optics 35.2 (2015): 225-230.
6. Nordstrom BL, Friedman DS, Mozaffari E, Quigley HA, Walker AM. Persistence and adherence with topical glaucoma therapy.” Am J Ophthalmol. 2005;140(4):598-606.
7. Fechtner RD, Godfrey DG, Budenz D, Stewart JA, Steward WC, Jasek MC. Prevalence of ocular surface complaints in patients with glaucoma using topical intraocular pressure-lowering medications. Cornea 2010;29(6): 618-621.
8. Li G, Akpek EK, Ahmad S. Glaucoma and ocular surface disease: more than meets the eye. Clinical Ophthalmology. 2022:3641-3649.
9. Skalicky SE, Goldberg G, McCluskey P. Ocular surface disease and quality of life in patients with glaucoma. Am J Ophthalmol. 2012;153(1):1-9.
10. Tsai JC, McClure CA, Ramos SE, Schlundt DG, Pichert JW. Compliance barriers in glaucoma: a systematic classification. J Glaucoma. 2003;12(5):393-398.
11. Singh IP, et al. Treatment success across different levels of preoperative disease burden: stratified two-year outcomes from the pivotal trial of iStent inject® trabecular micro-bypass in primary open-angle glaucoma and cataract. Clin Ophthalmol. 2021;15:3231.
INDICATION FOR USE: The iStent inject® W, is intended to reduce intraocular pressure safely and effectively in patients diagnosed with primary open-angle glaucoma, pseudo-exfoliative glaucoma or pigmentary glaucoma. The iStent inject® W, can deliver two (2) stents on a single pass, through a single incision. The implant is designed to stent open a passage through the trabecular meshwork to allow for an increase in the facility of outflow and a subsequent reduction in intraocular pressure. The device is safe and effective when implanted in combination with cataract surgery in those subjects who require intraocular pressure reduction and/or would benefit from glaucoma medication reduction. The device may also be implanted in patients who continue to have elevated intraocular pressure despite prior treatment with glaucoma medications and conventional glaucoma surgery. CONTRAINDICATIONS: The iStent inject® W System is contraindicated under the following circumstances or conditions: • In eyes with primary angle closure glaucoma, or secondary angle-closure glaucoma, including neovascular glaucoma, because the device would not be expected to work in such situations. • In patients with retrobulbar tumor, thyroid eye disease, Sturge-Weber Syndrome or any other type of condition that may cause elevated episcleral venous pressure. WARNINGS/PRECAUTIONS: • For prescription use only. • This device has not been studied in patients with uveitic glaucoma. • Do not use the device if the Tyvek® lid has been opened or the packaging appears damaged. In such cases, the sterility of the device may be compromised. • Due to the sharpness of certain injector components (i.e. the insertion sleeve and trocar), care should be exercised to grasp the injector body. Dispose of device in a sharps container. • iStent inject® W is MR-Conditional; see MRI Information below. • Physician training is required prior to use of the iStent inject® W System. • Do not re-use the stent(s) or injector, as this may result in infection and/or intraocular inflammation, as well as occurrence of potential postoperative adverse events as shown below under “Potential Complications.” • There are no known compatibility issues with the iStent inject® W and other intraoperative devices. (e.g., viscoelastics) or glaucoma medications. • Unused product & packaging may be disposed of in accordance with facility procedures. Implanted medical devices and contaminated products must be disposed of as medical waste. • The surgeon should monitor the patient postoperatively for proper maintenance of intraocular pressure. If intraocular pressure is not adequately maintained after surgery, the surgeon should consider an appropriate treatment regimen to reduce intraocular pressure. • Patients should be informed that placement of the stents, without concomitant cataract surgery in phakic patients, can enhance the formation or progression of cataract. ADVERSE EVENTS: Please refer to Directions For Use for additional adverse event information. CAUTION: Please reference the Directions For Use labelling for a complete list of contraindications, warnings and adverse events.
© 2024 Glaukos Corporation. Glaukos, iStent®, iStent inject® and iStent inject® W are registered trademarks of Glaukos Corporation. PM-EU-0284


